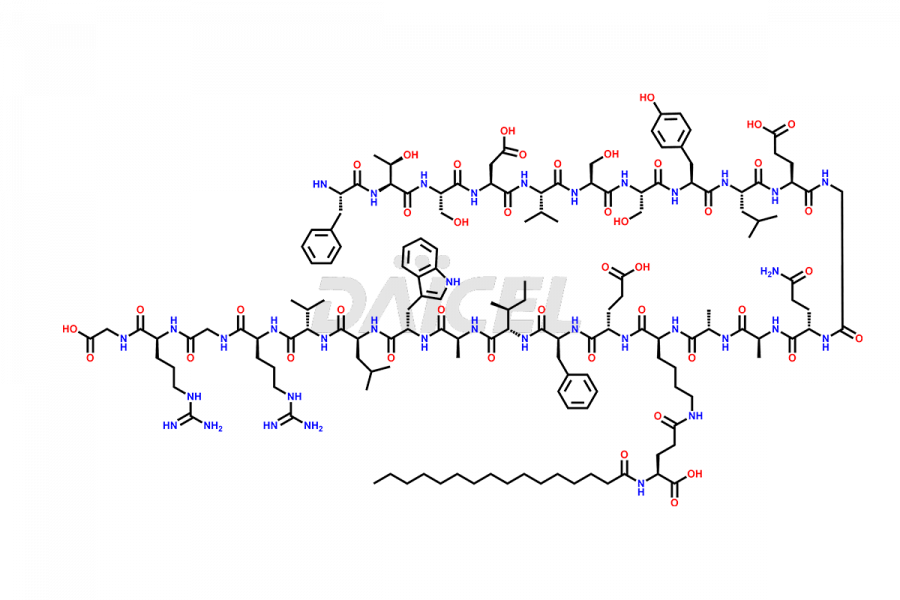
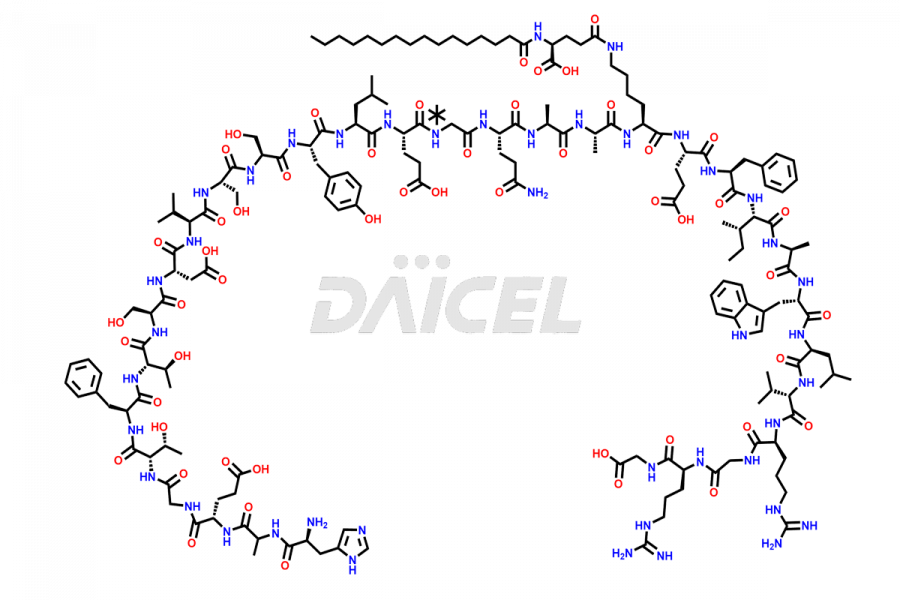
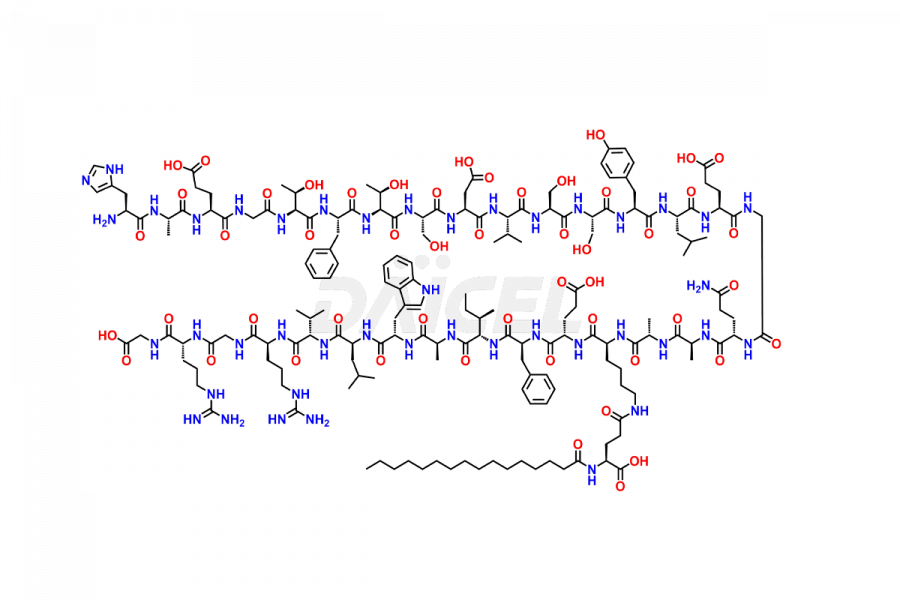
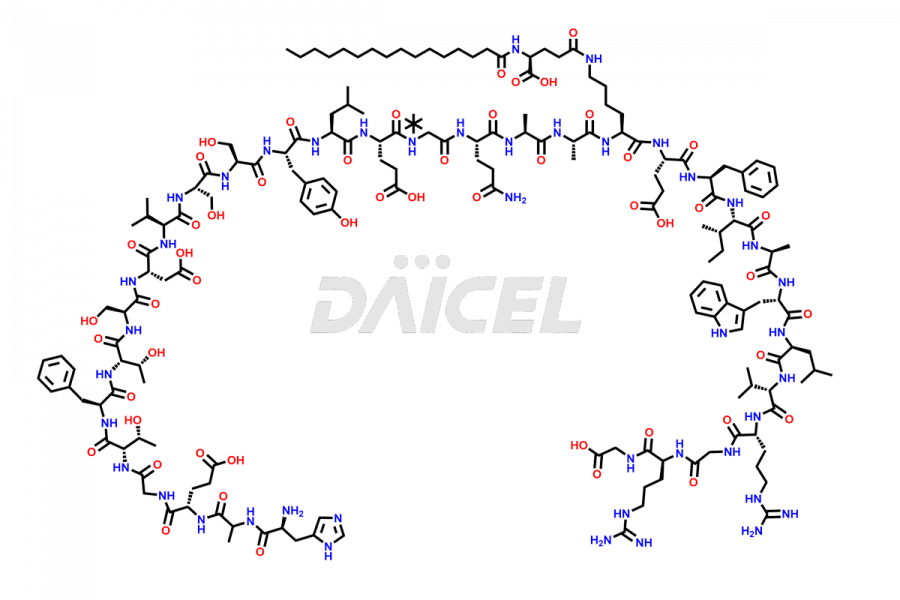
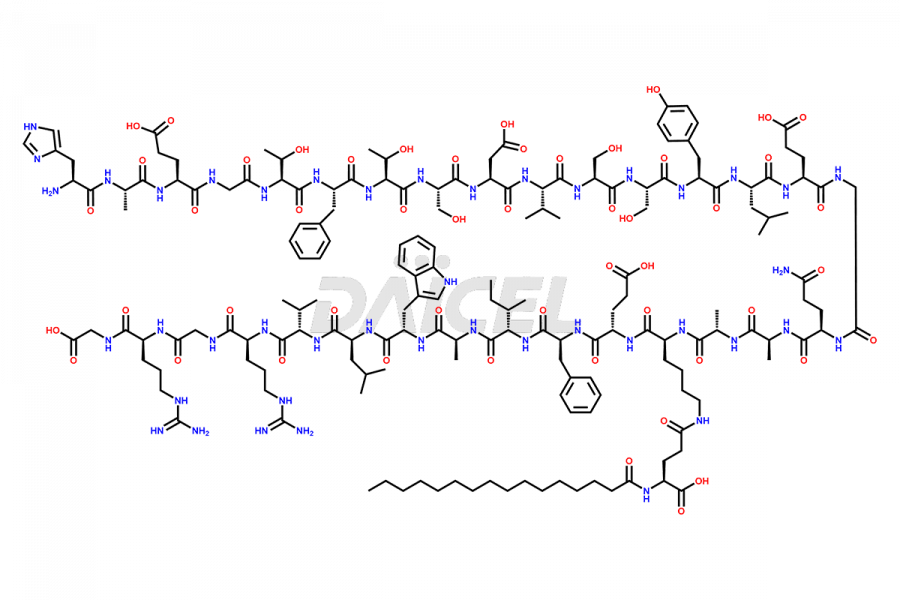
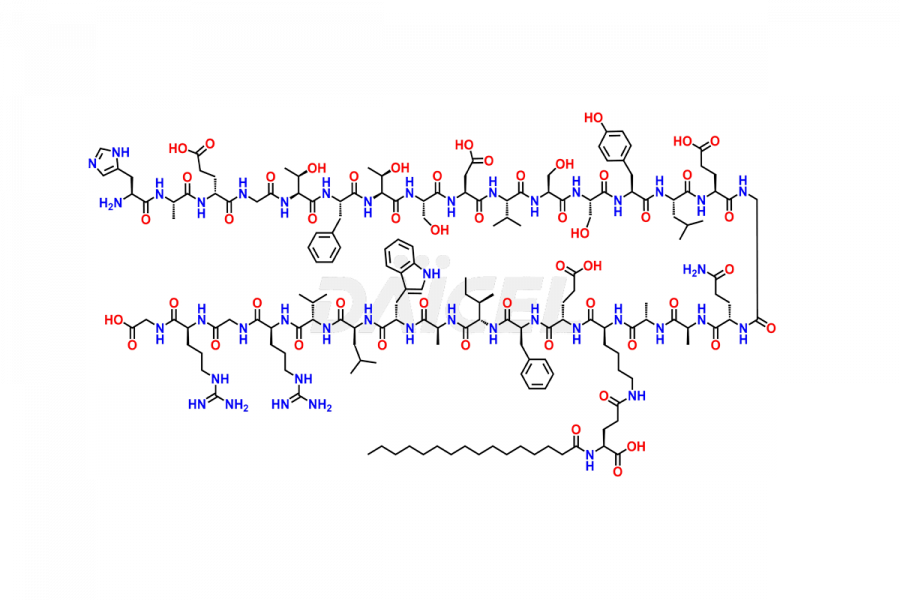
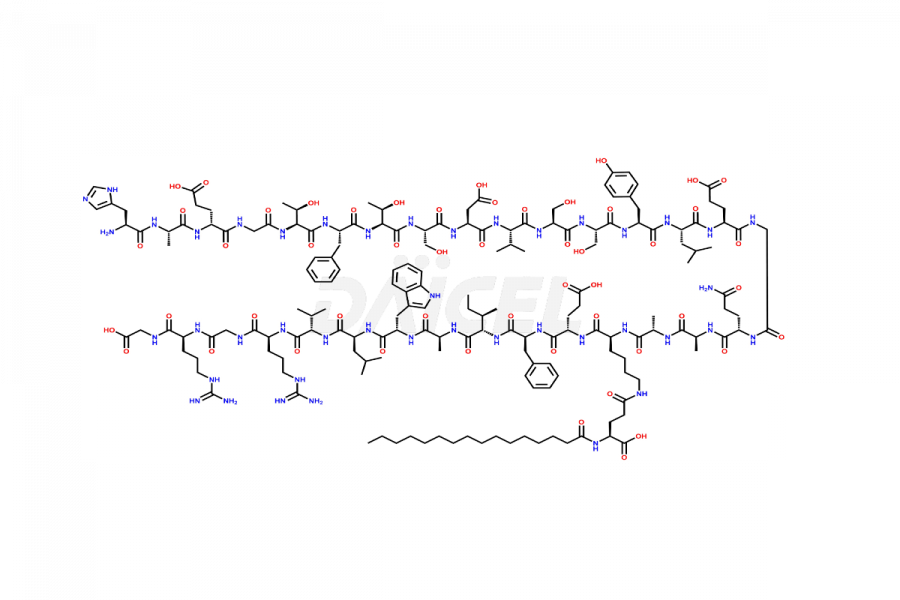
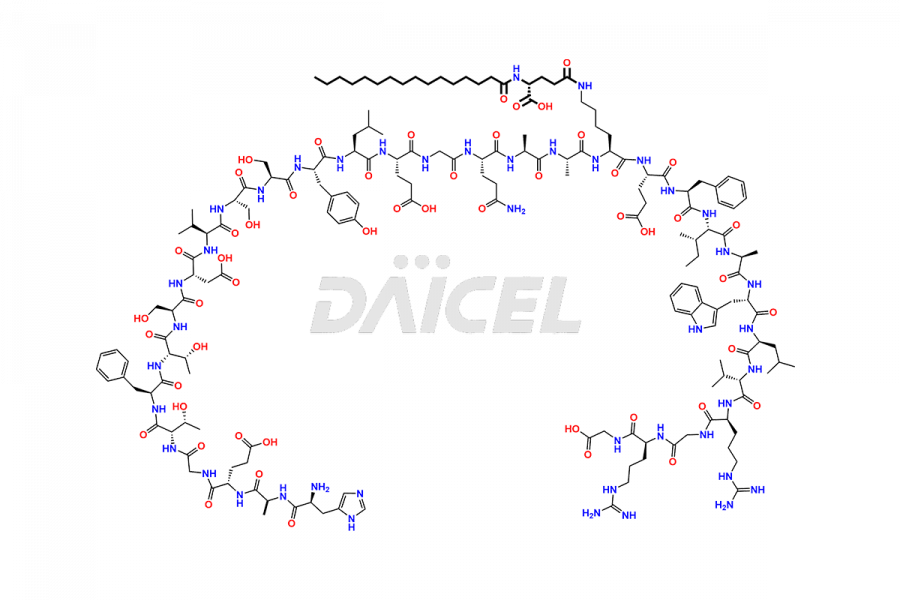
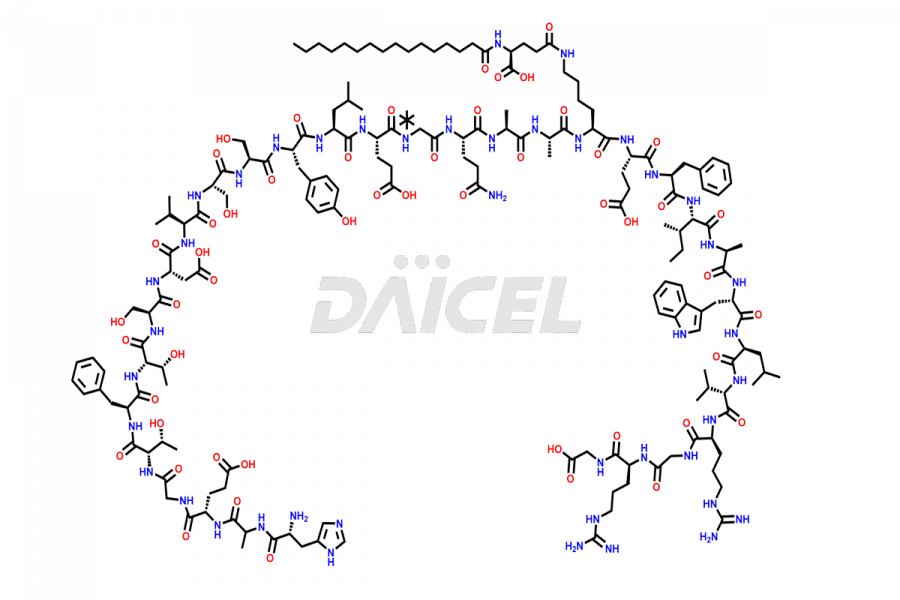
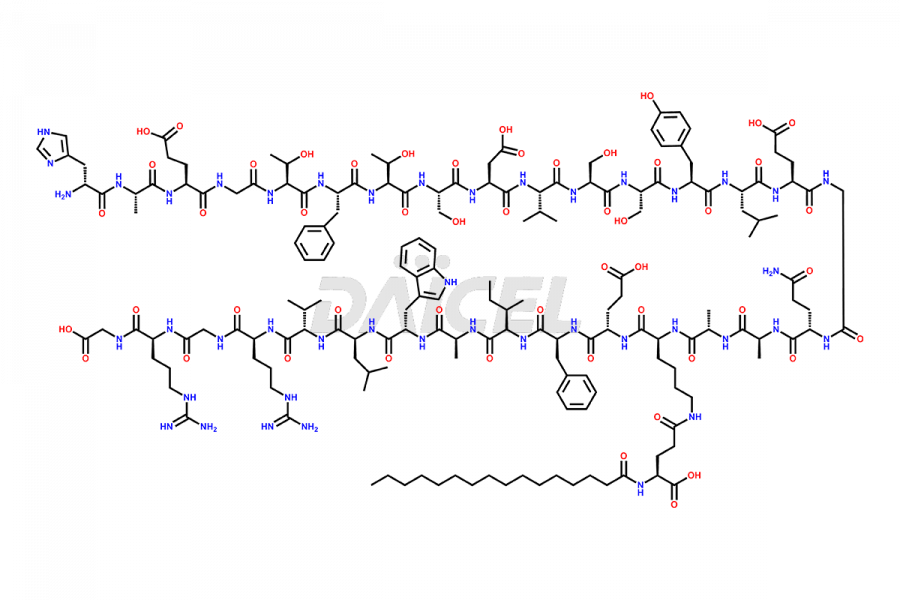
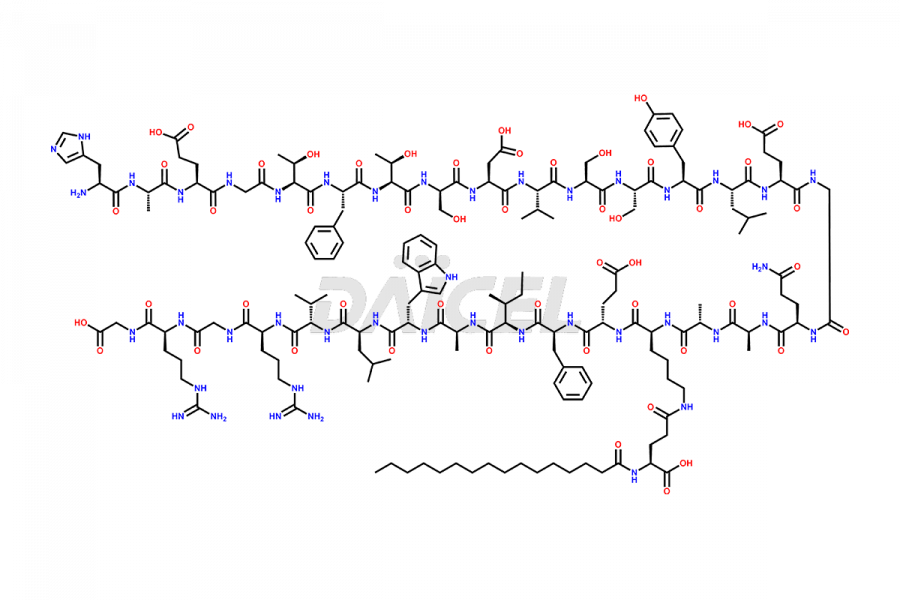
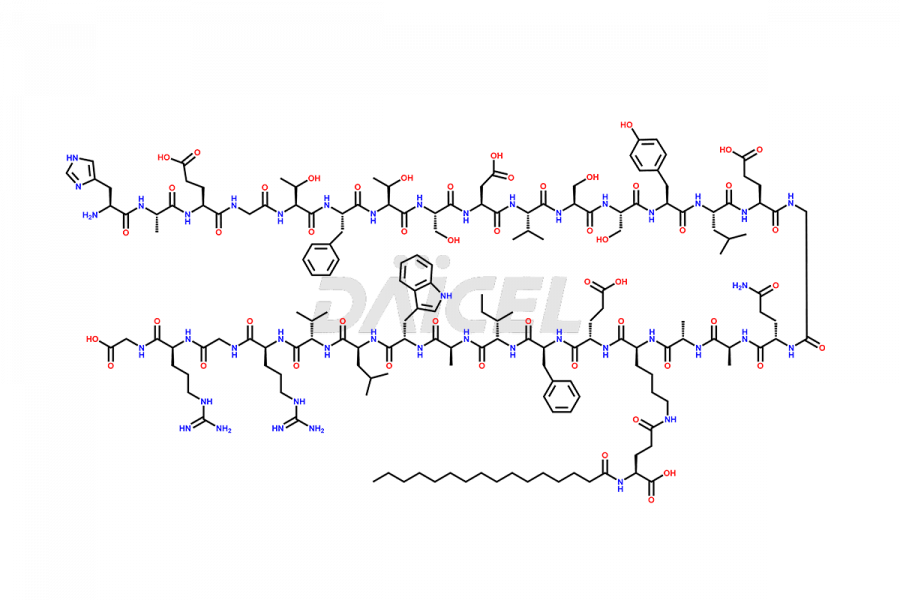
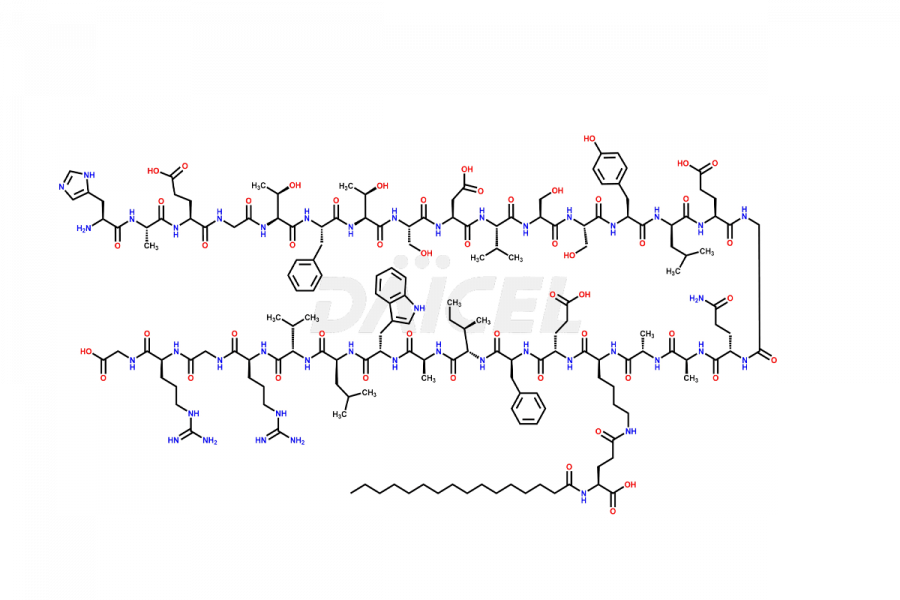
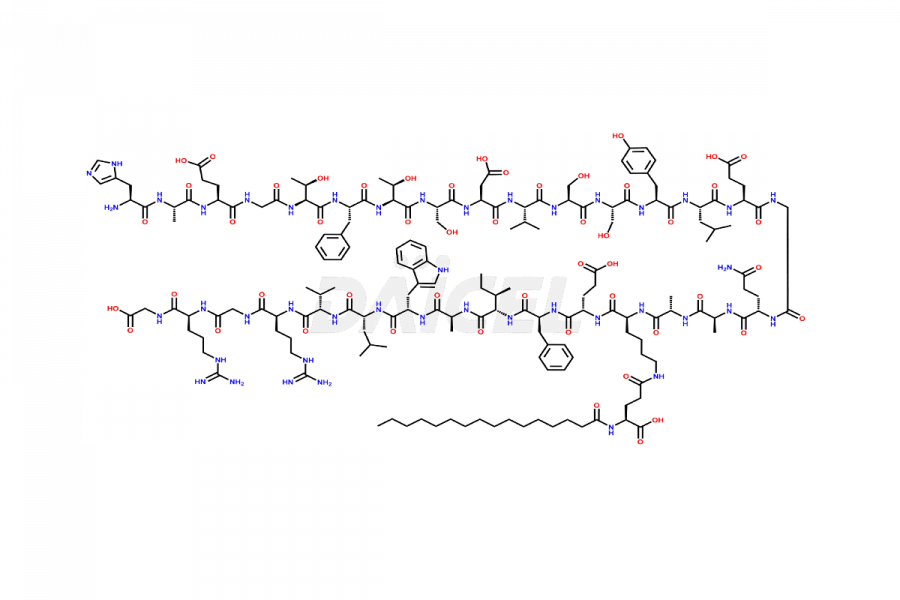
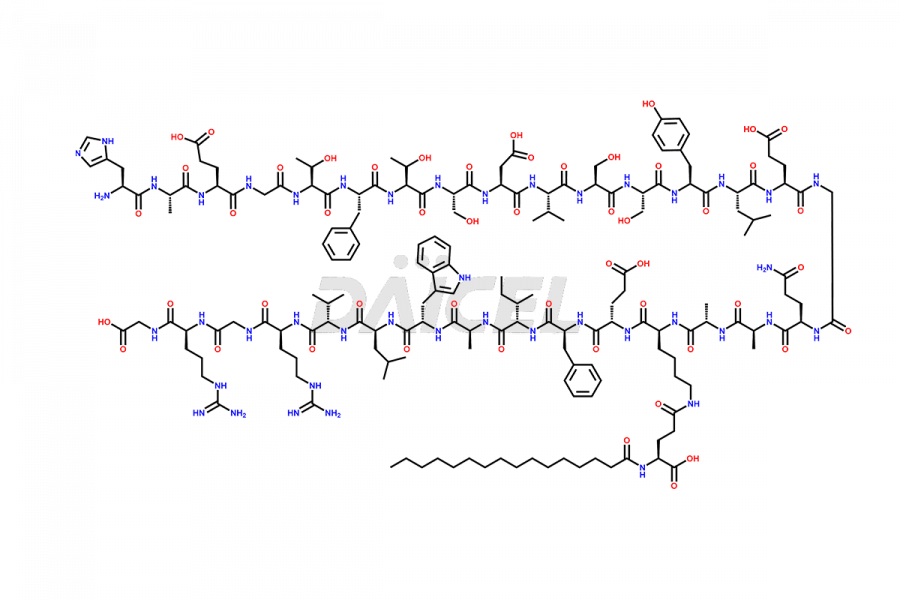
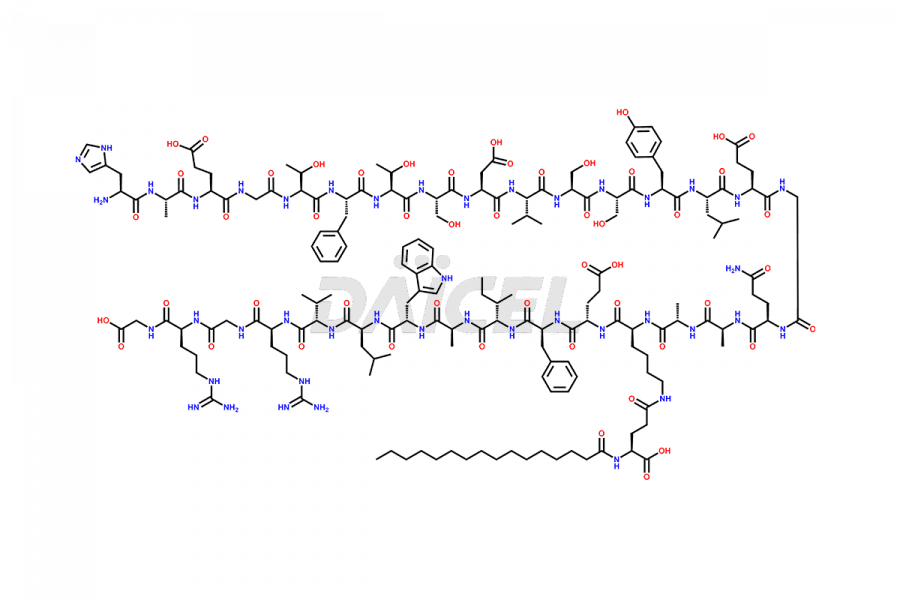
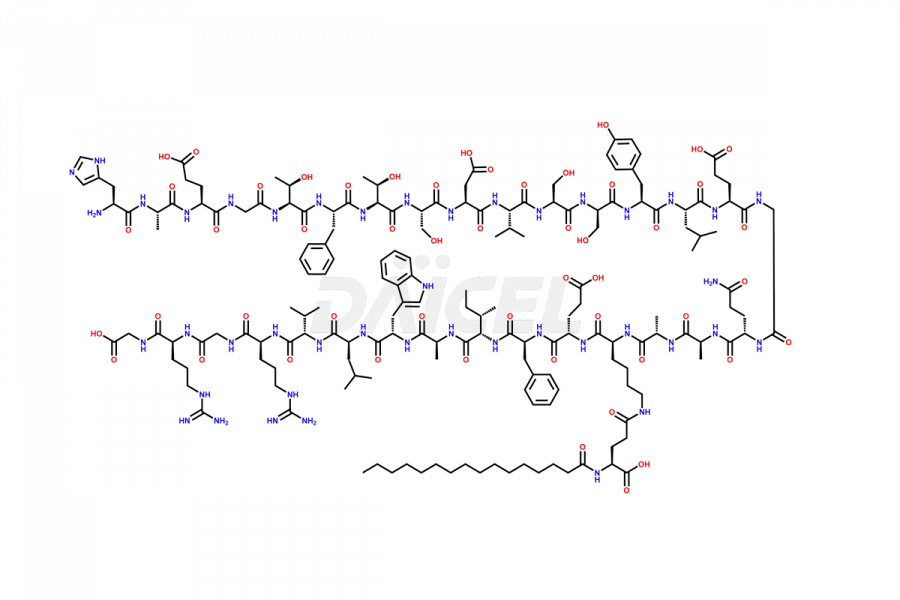
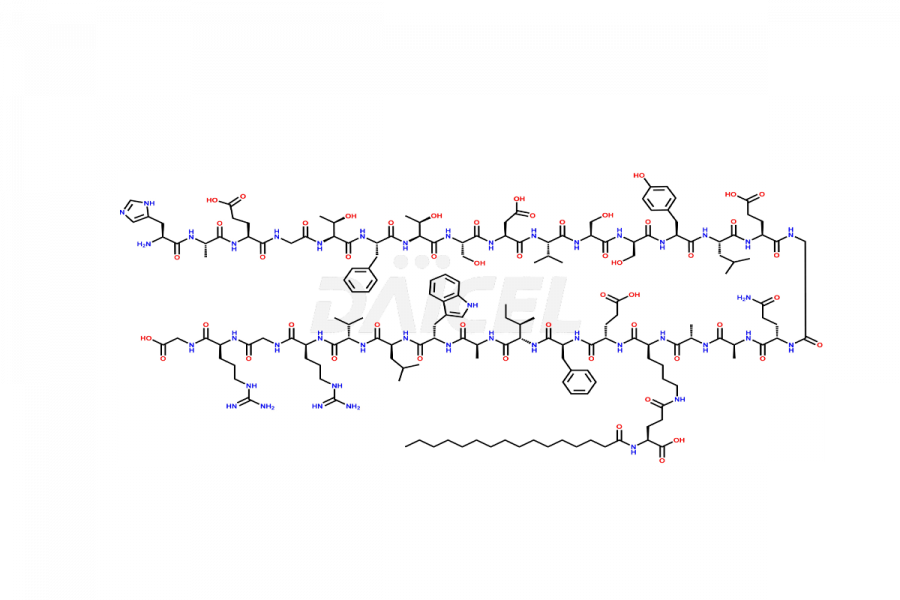
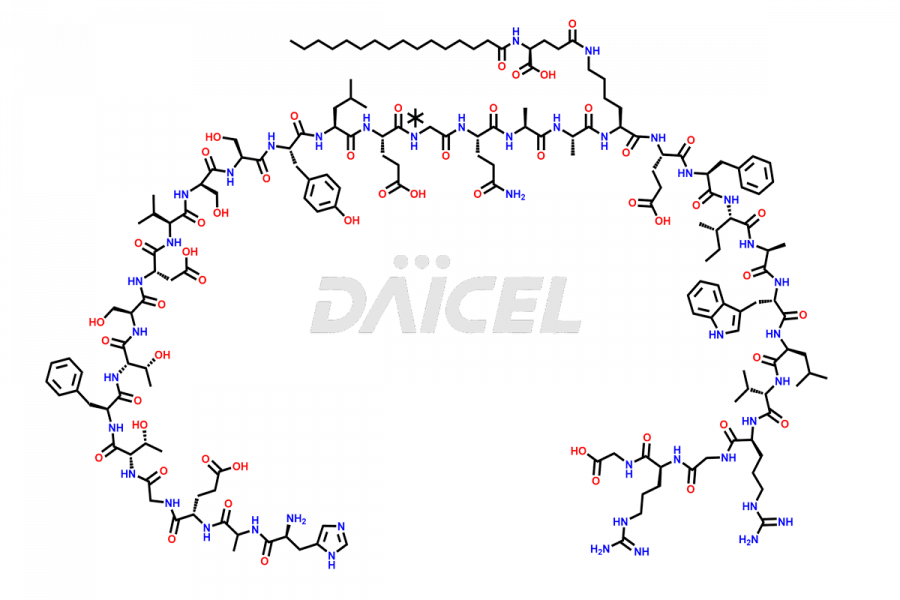
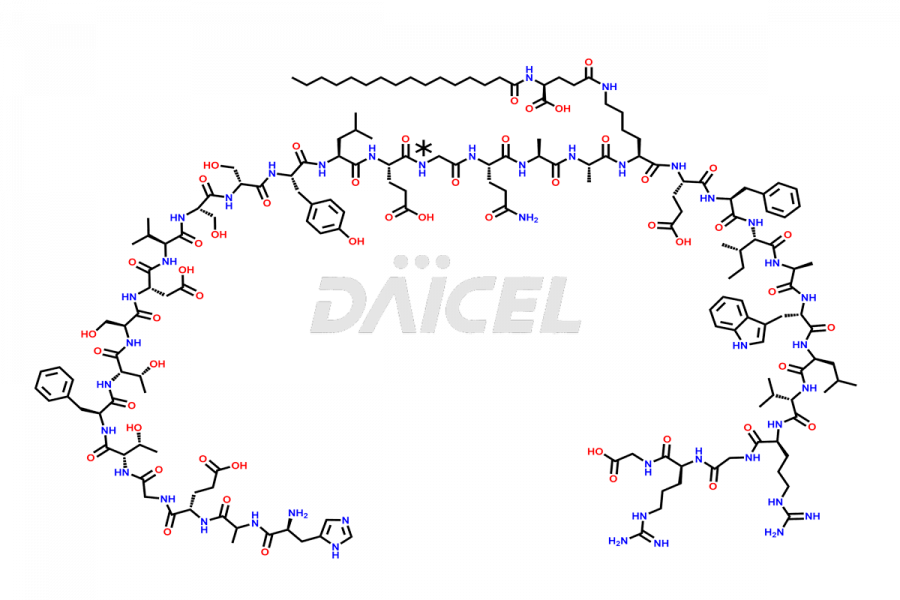
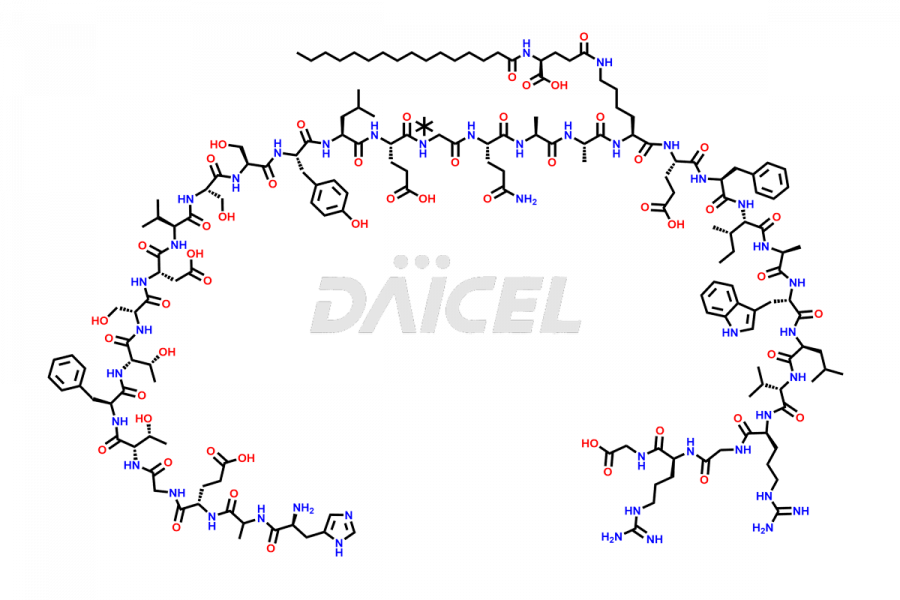
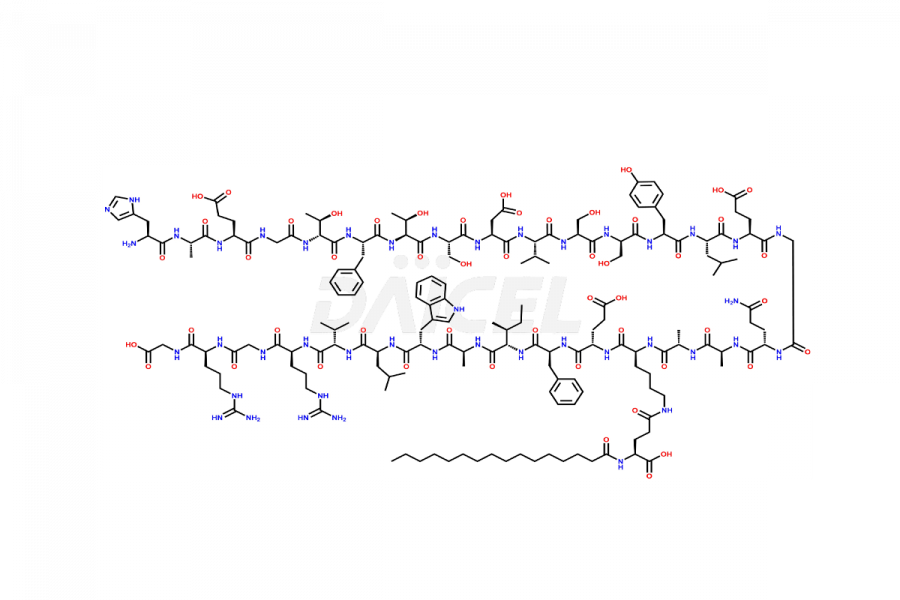
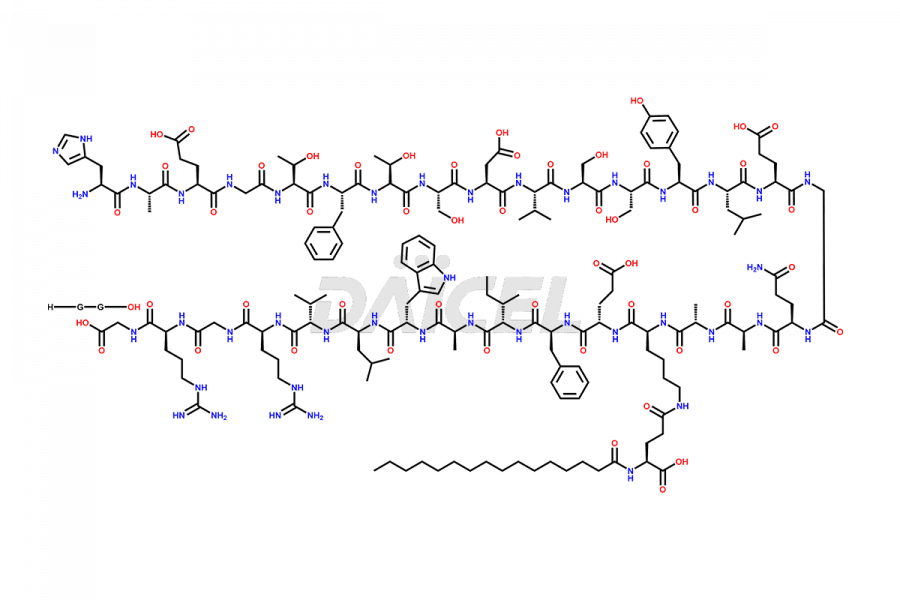
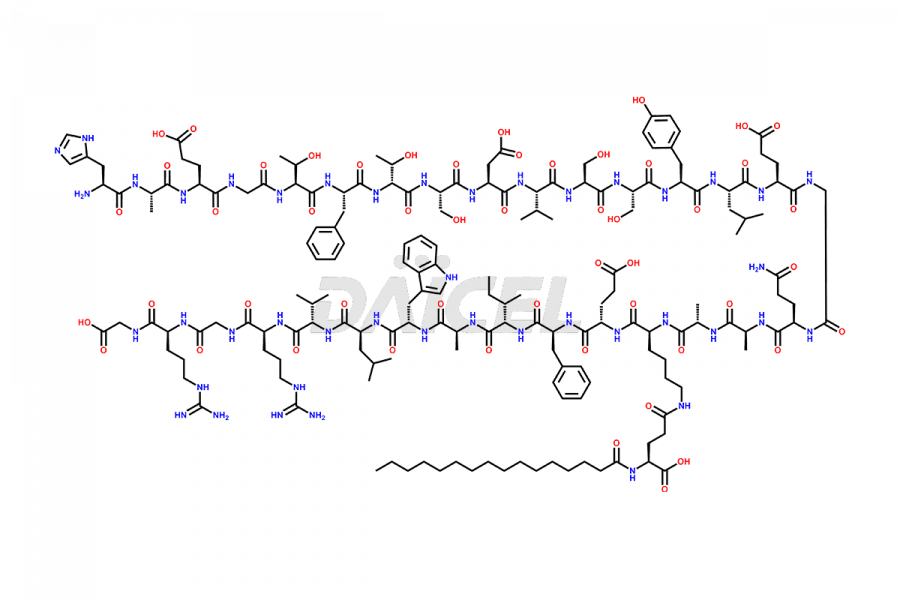
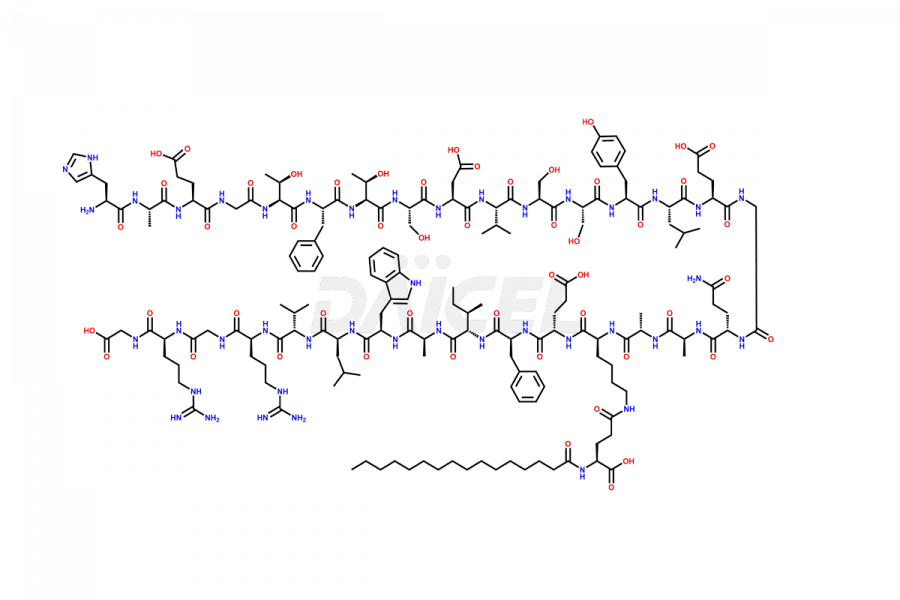
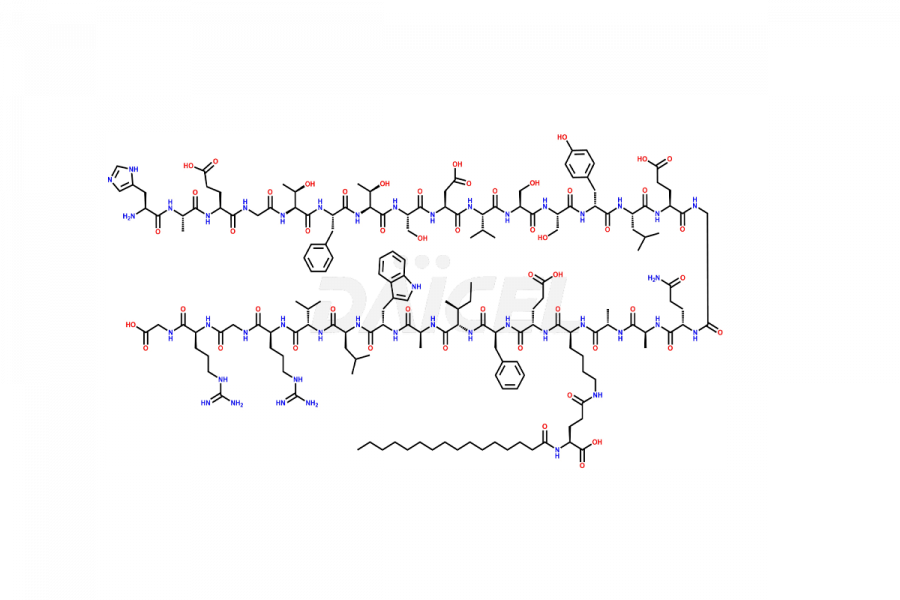
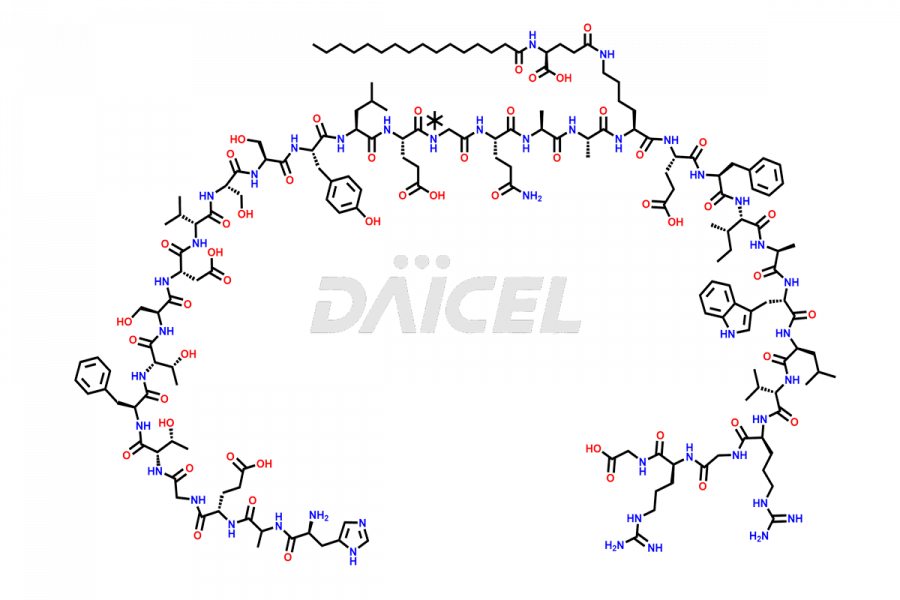
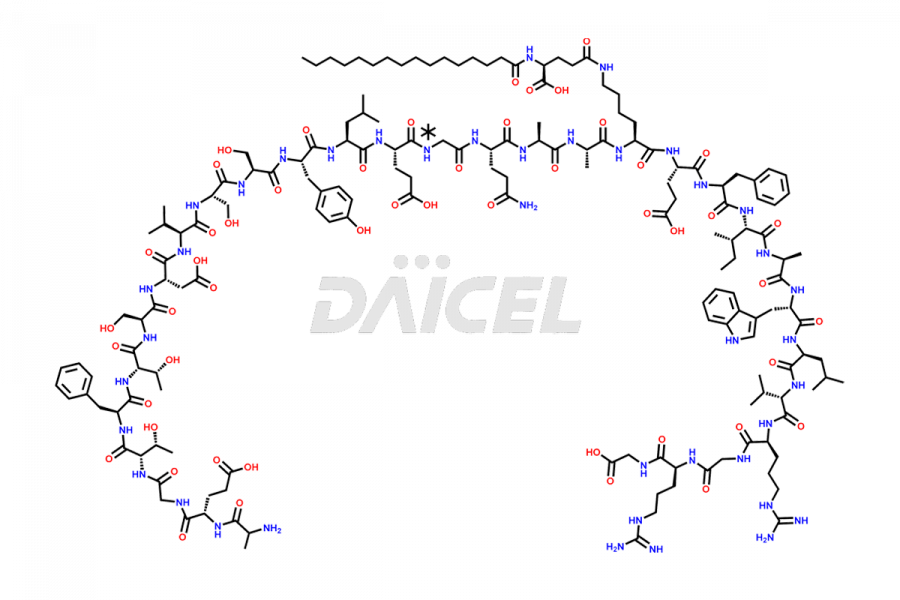
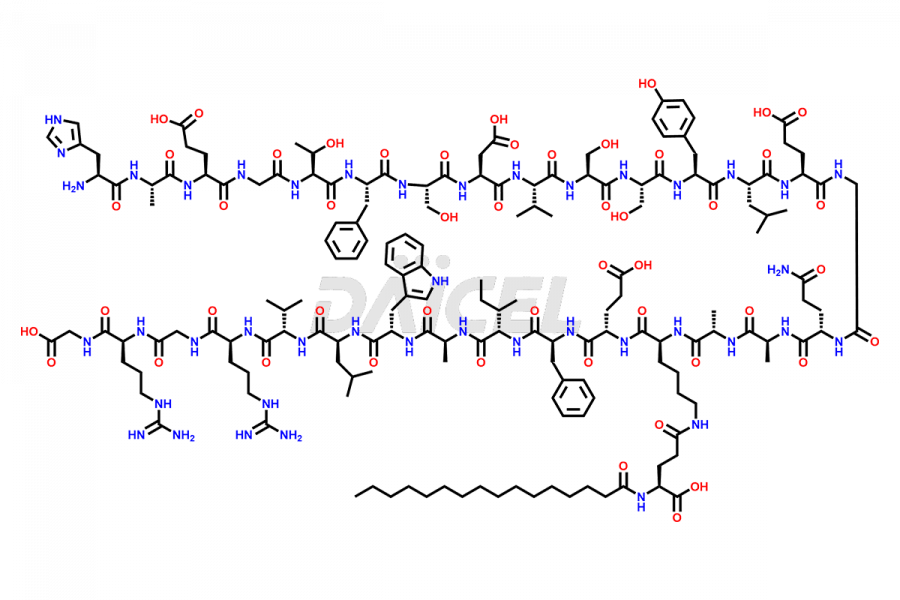
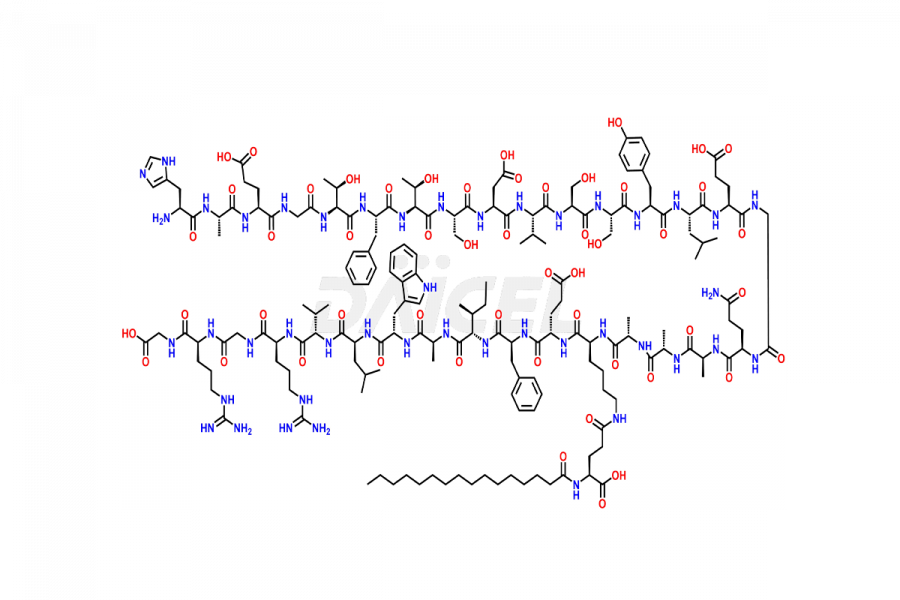
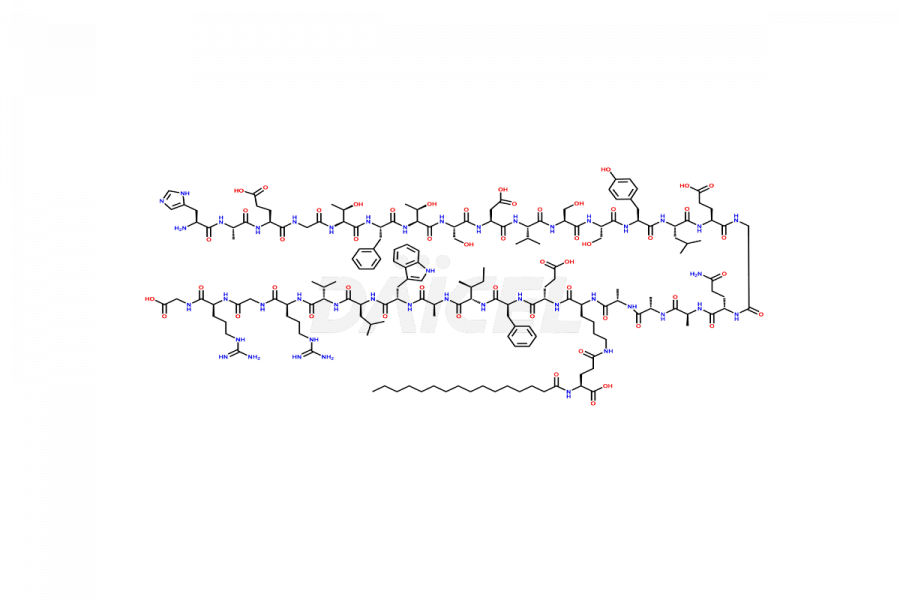
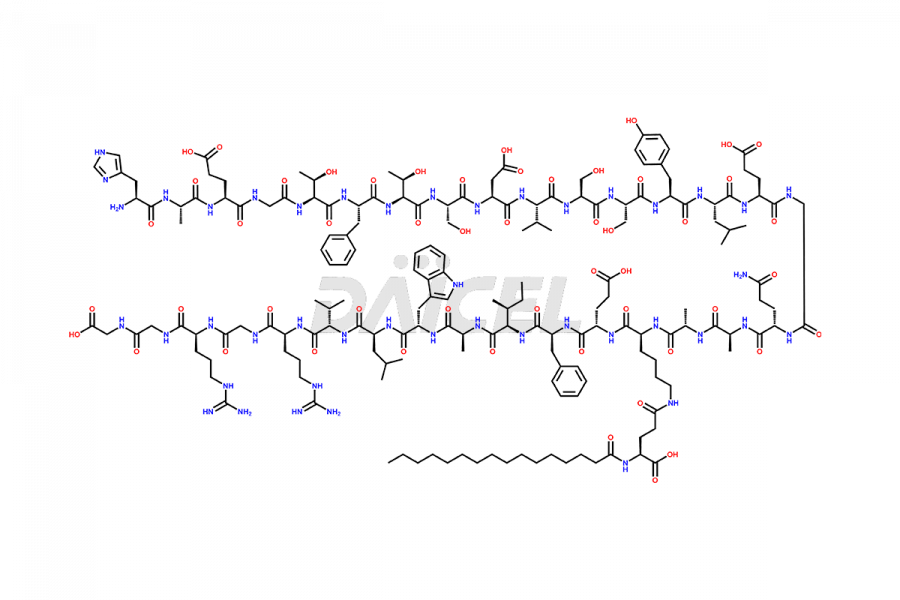
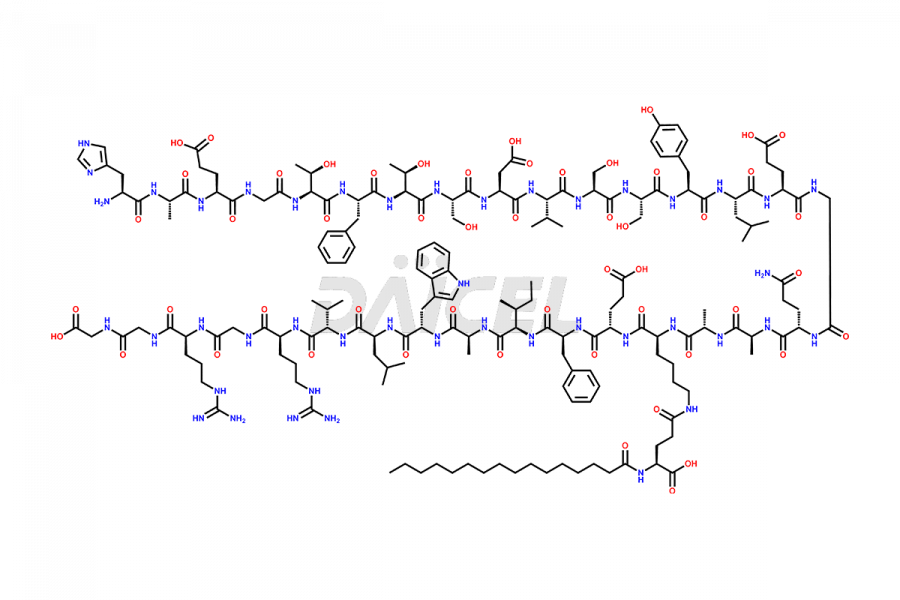
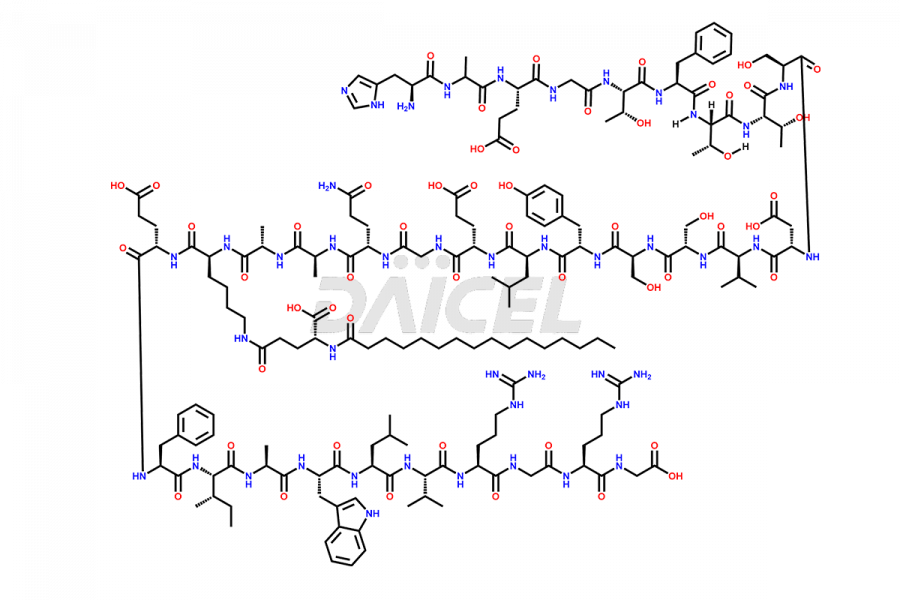
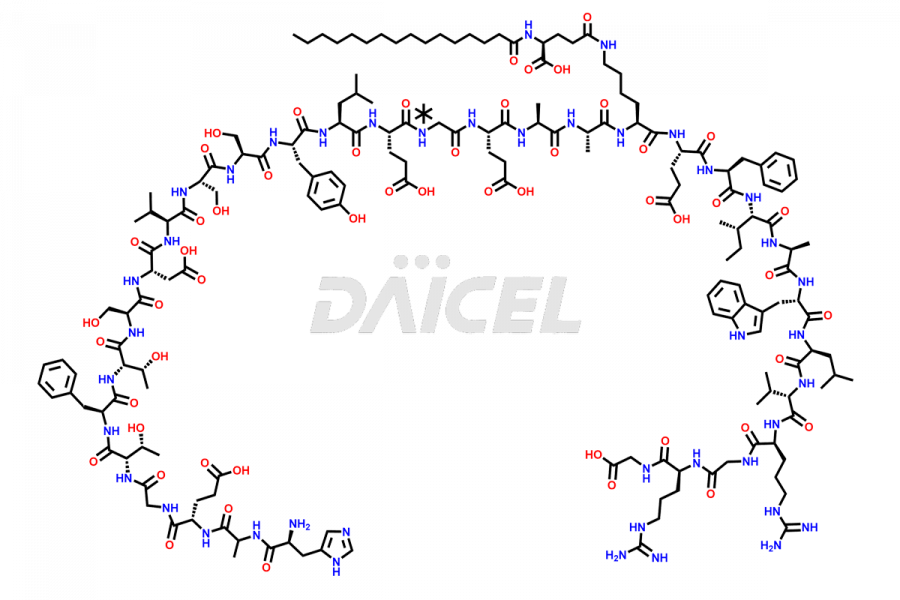
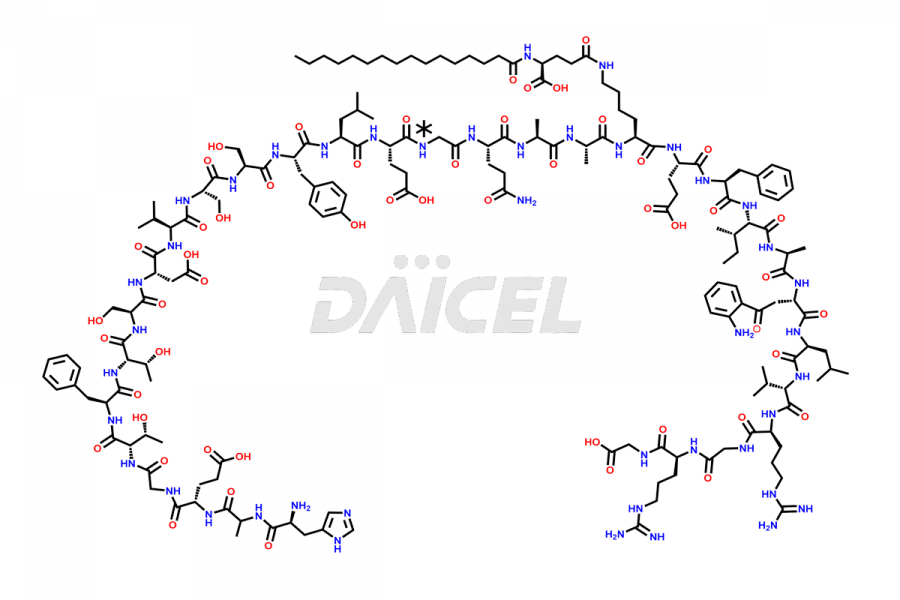
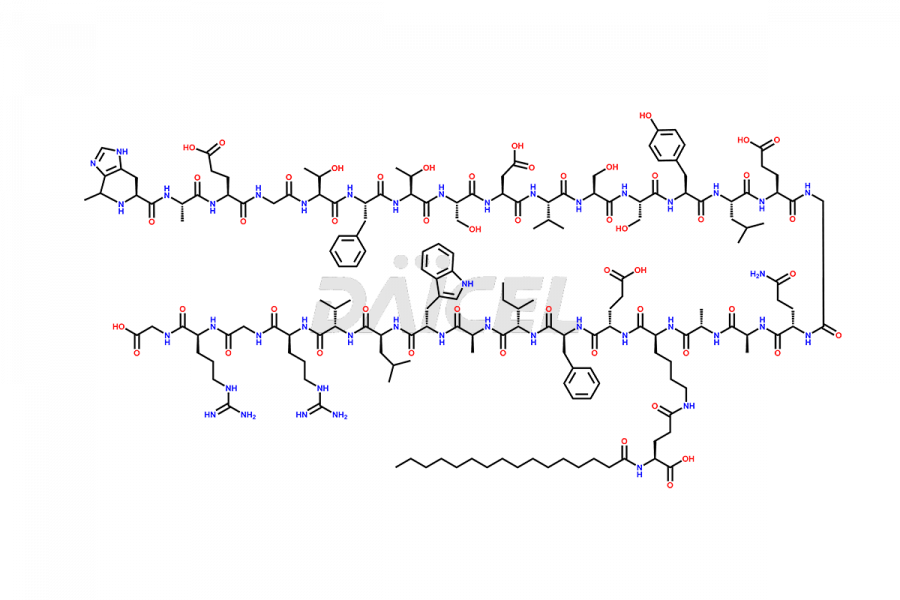
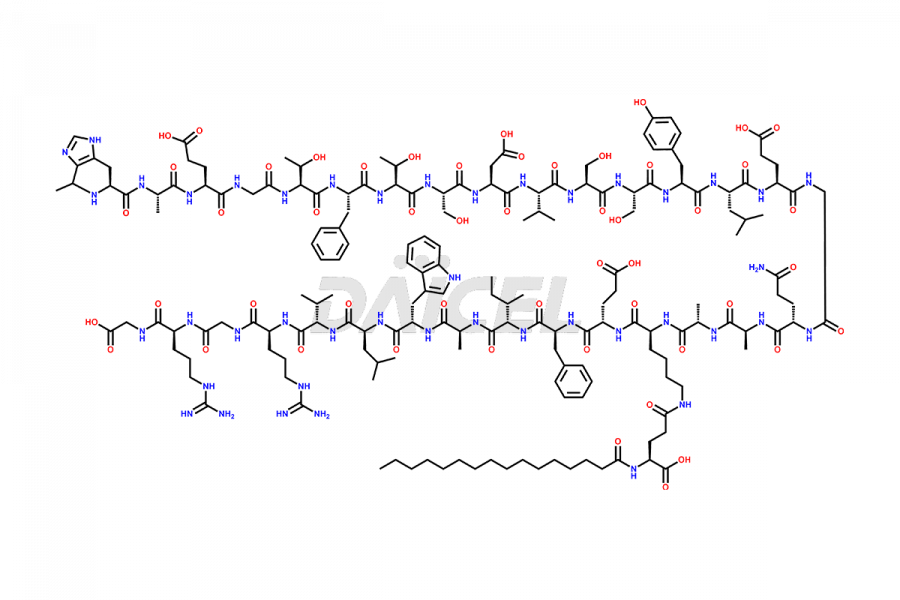
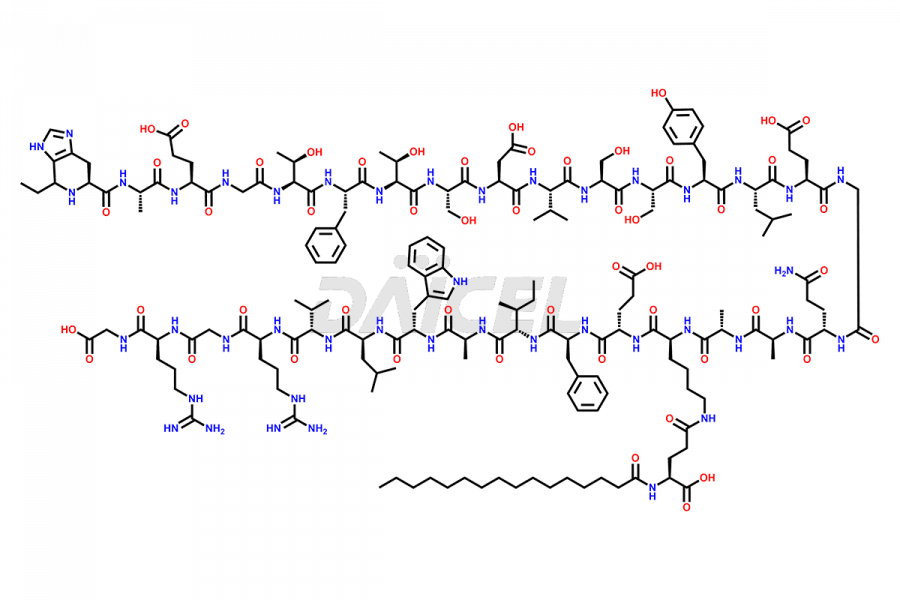
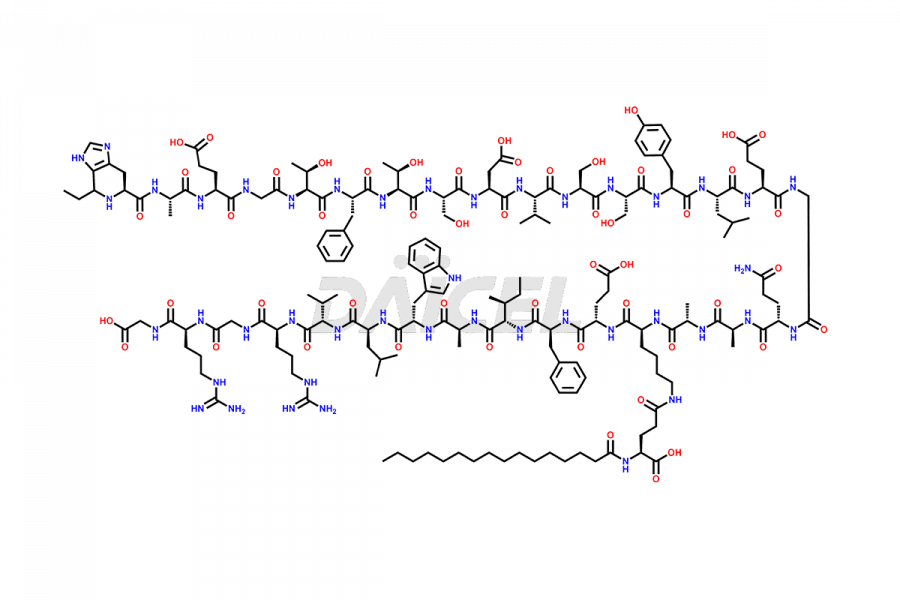
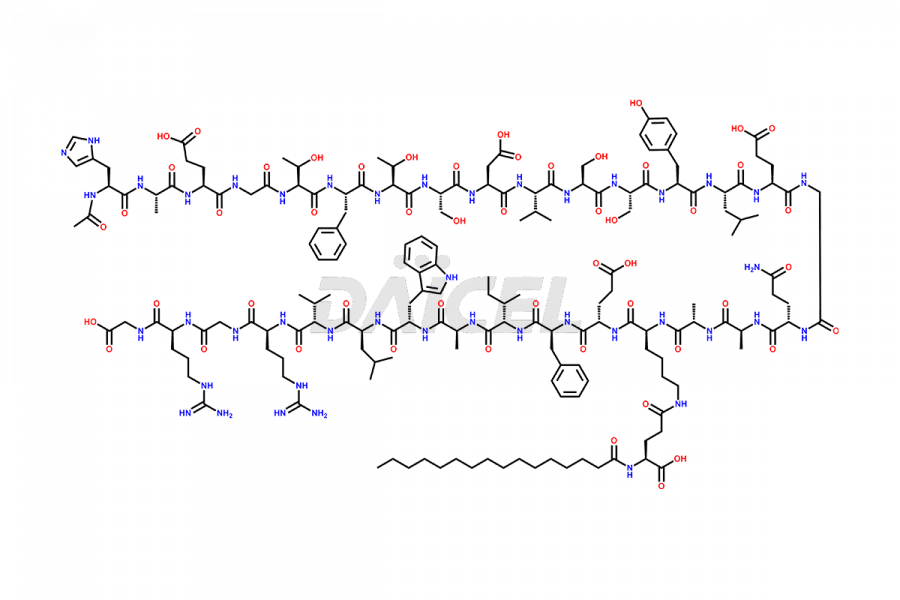
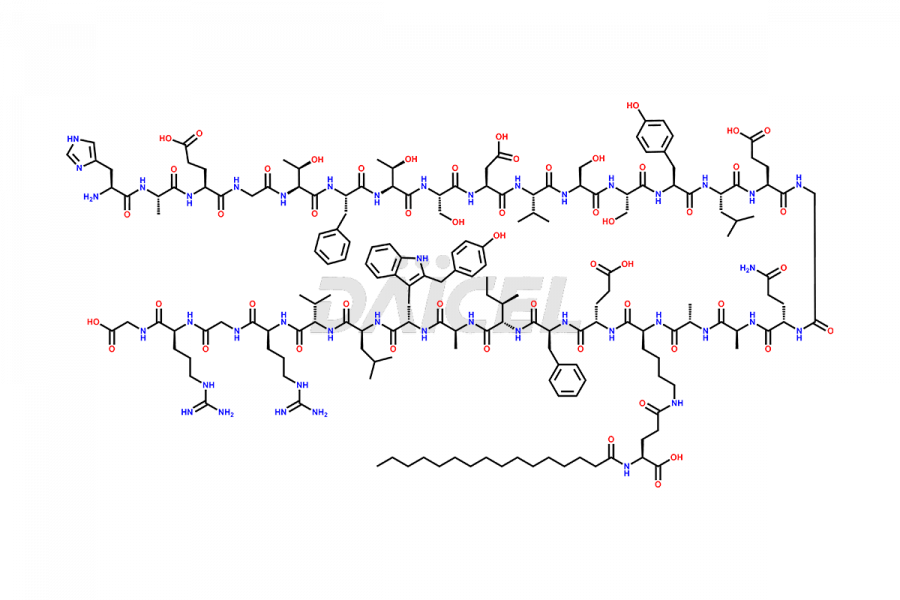
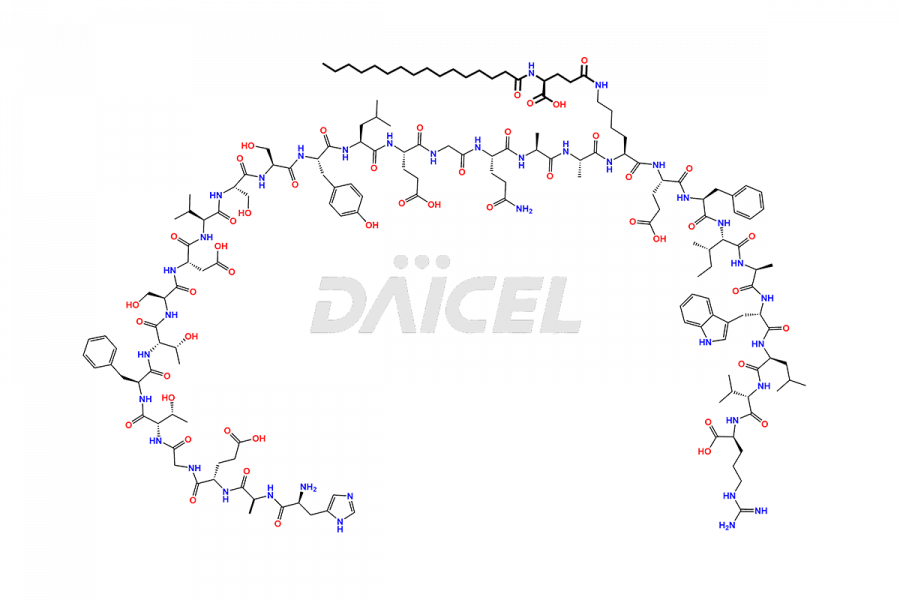
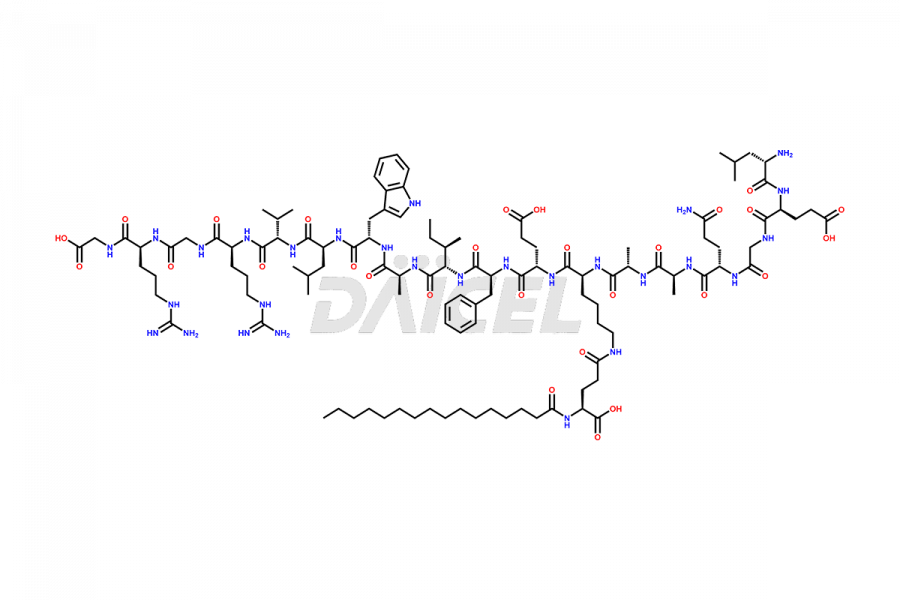
(6-31)-Liraglutide
- CAT Number DCTI-D-000156
- CAS NUMBER NA
- MOLECULAR FORMULA C152H236N36O43
- MOLECULAR WEIGHT 3255.77
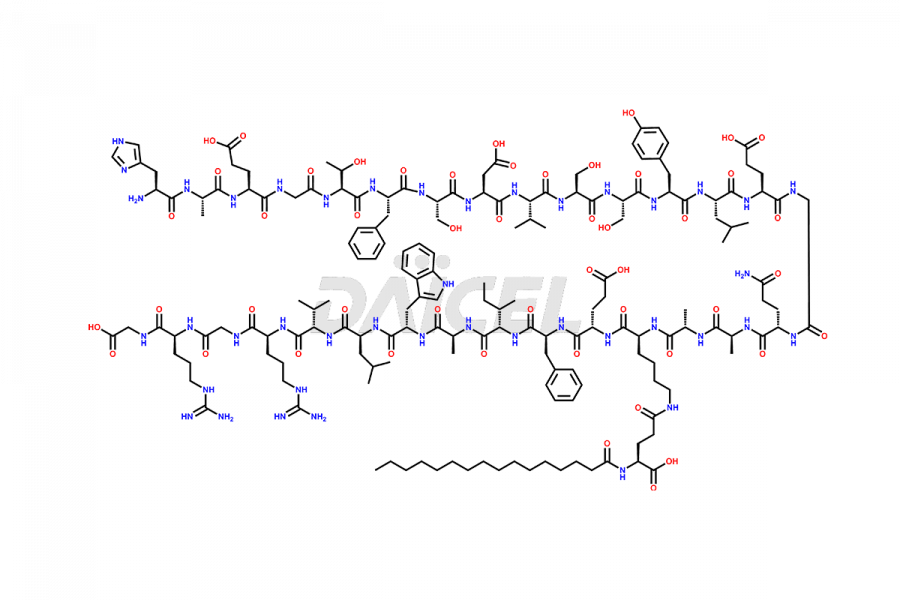
(9-31)-Liraglutide
- CAT Number DCTI-D-000157
- CAS NUMBER NA
- MOLECULAR FORMULA C136H215N33O38
- MOLECULAR WEIGHT 2920.41
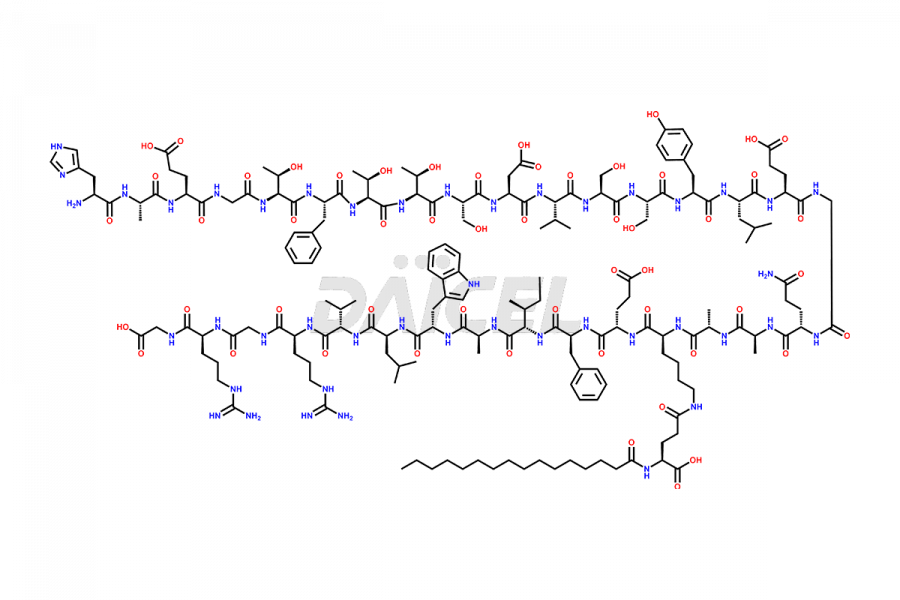
[1-28]-Liraglutide
- CAT Number DCTI-D-000258
- CAS Number NA
- Molecular Formula C162H247N37O48
- Molecular Weight 3480.97
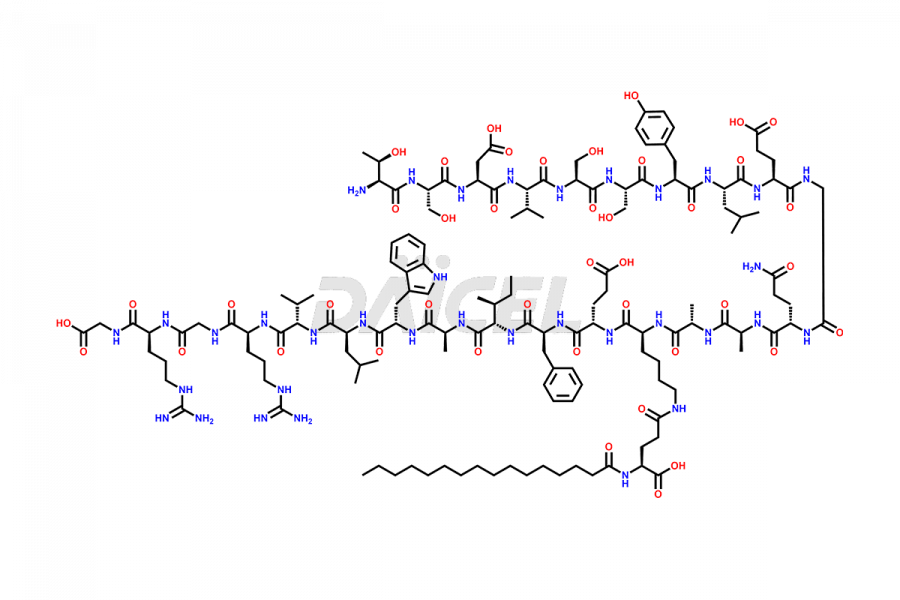
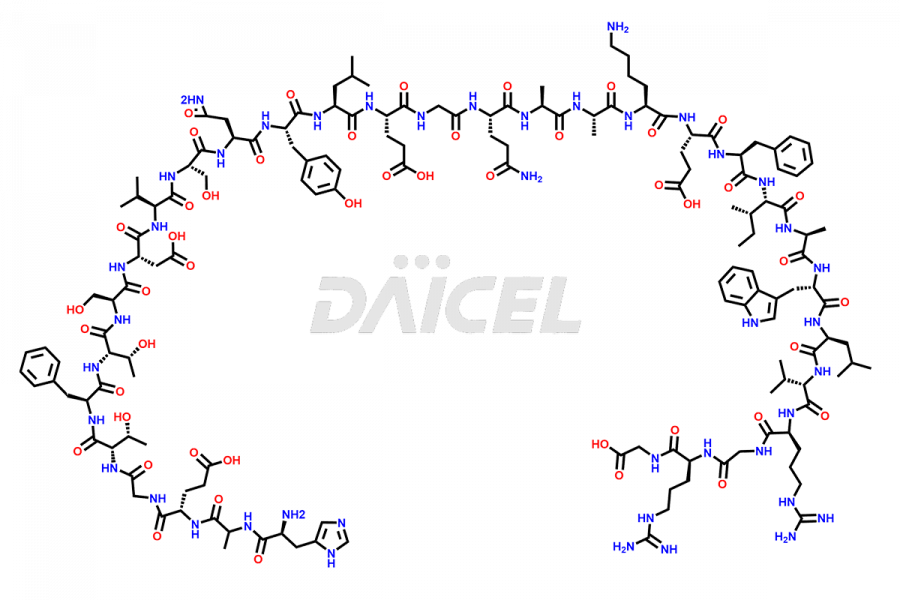
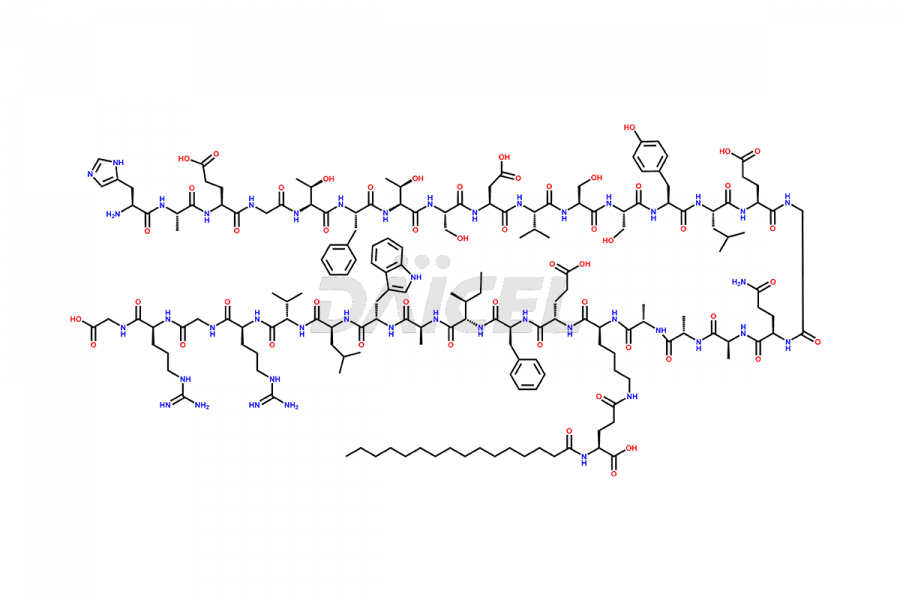
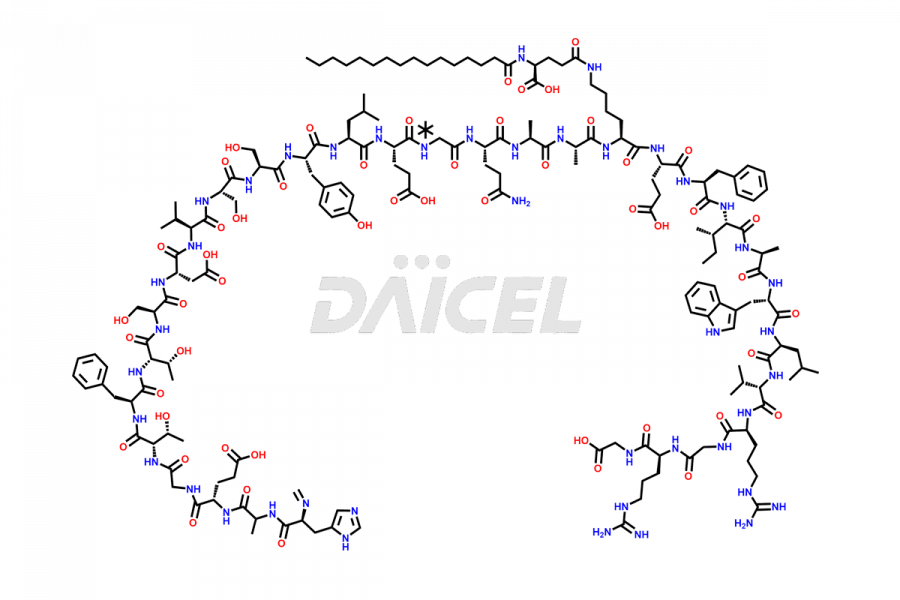
[10-31]-Liraglutide
- CAT Number DCTI-D-000479
- CAS NUMBER NA
- MOLECULAR FORMULA C132H210N32O35
- MOLECULAR WEIGHT 2805.32
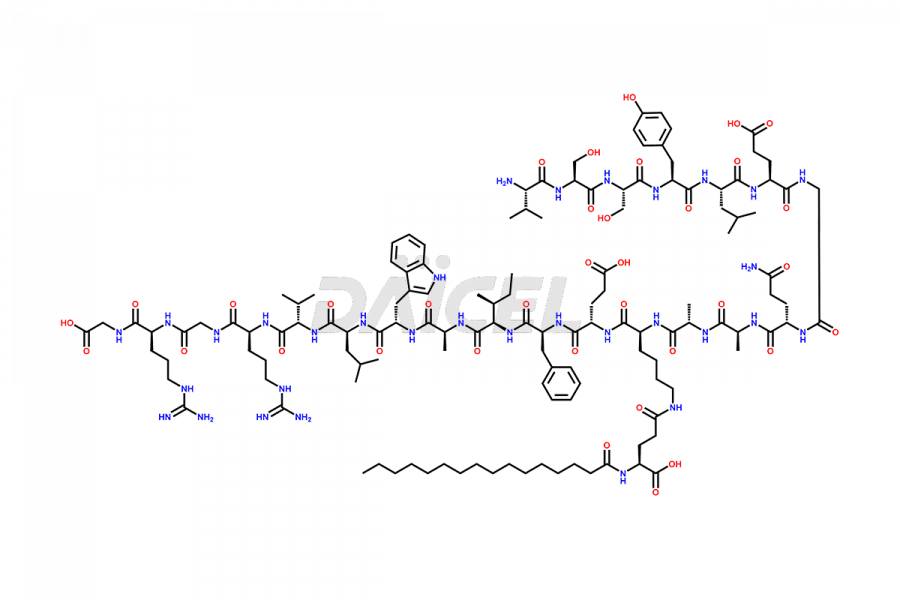
[10-31]-Liraglutide phosphate salt
- CAT Number DCTI-D-000537
- CAS Number NA
- Molecular Formula C132H210N32O35
- Molecular Weight 2805.32
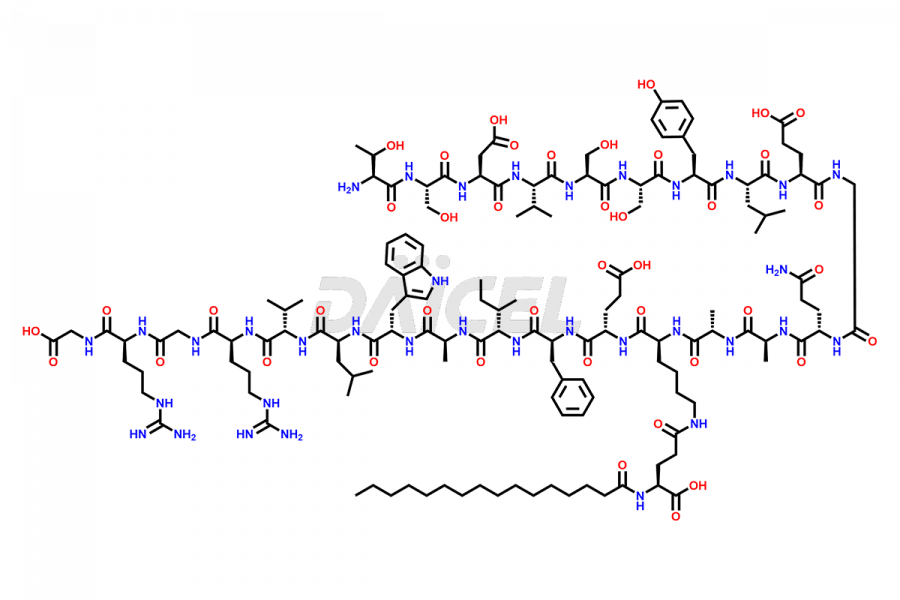
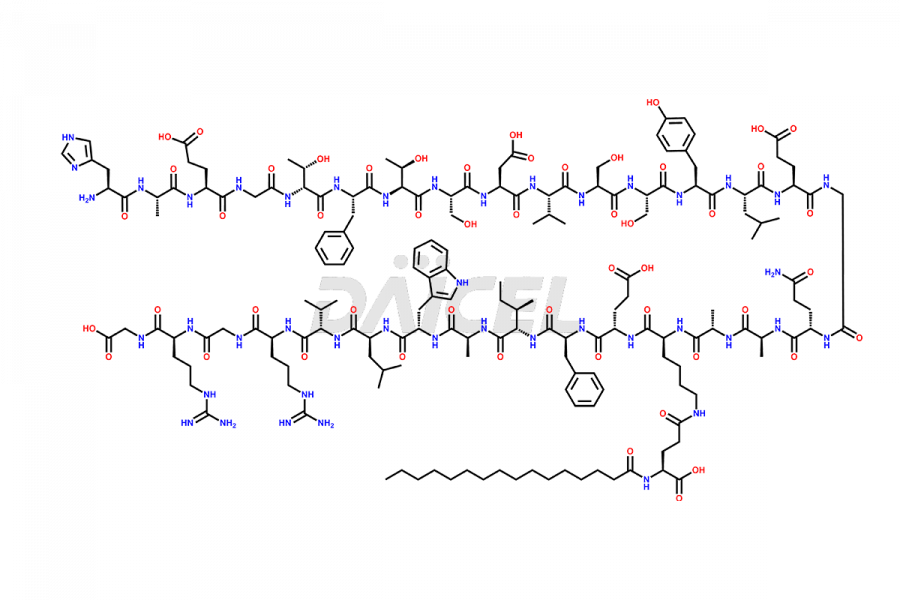
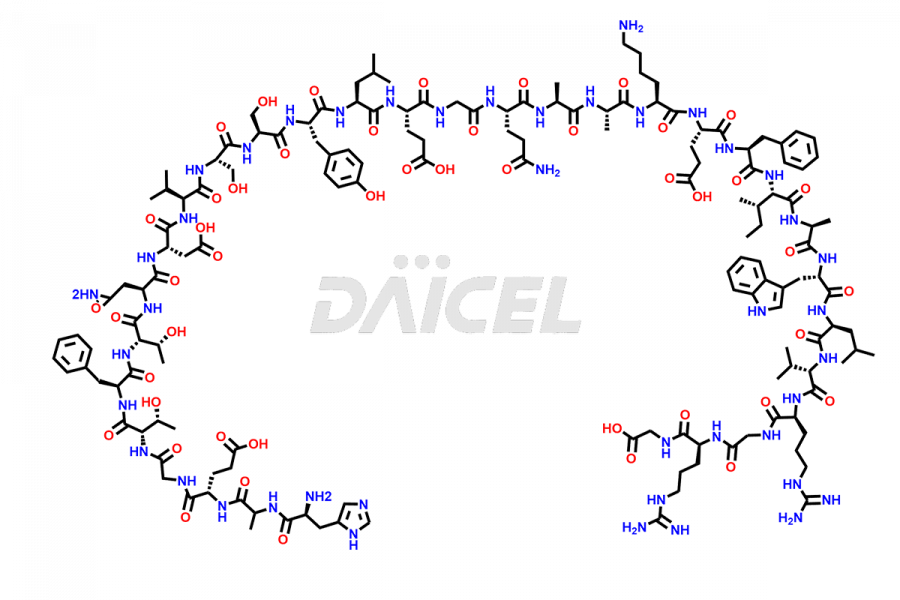
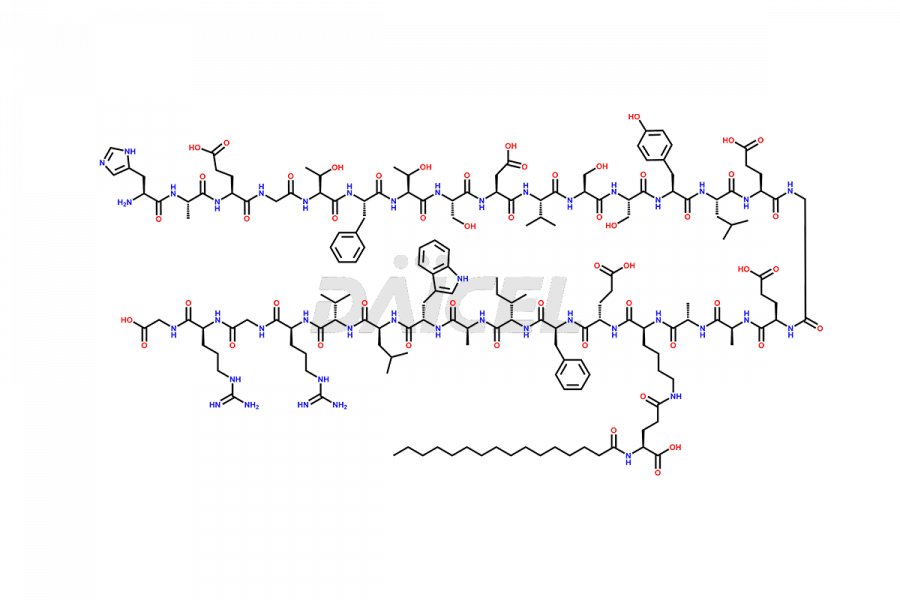
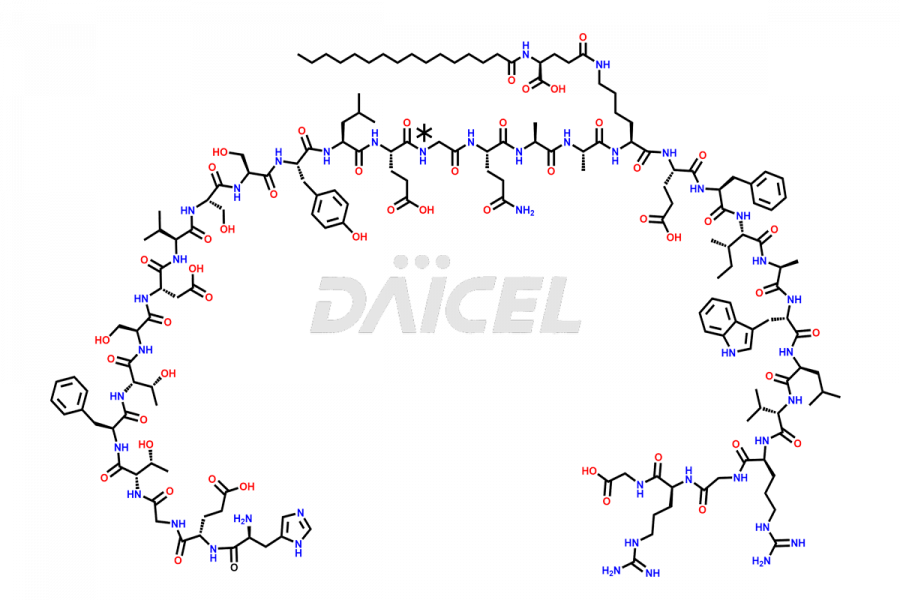
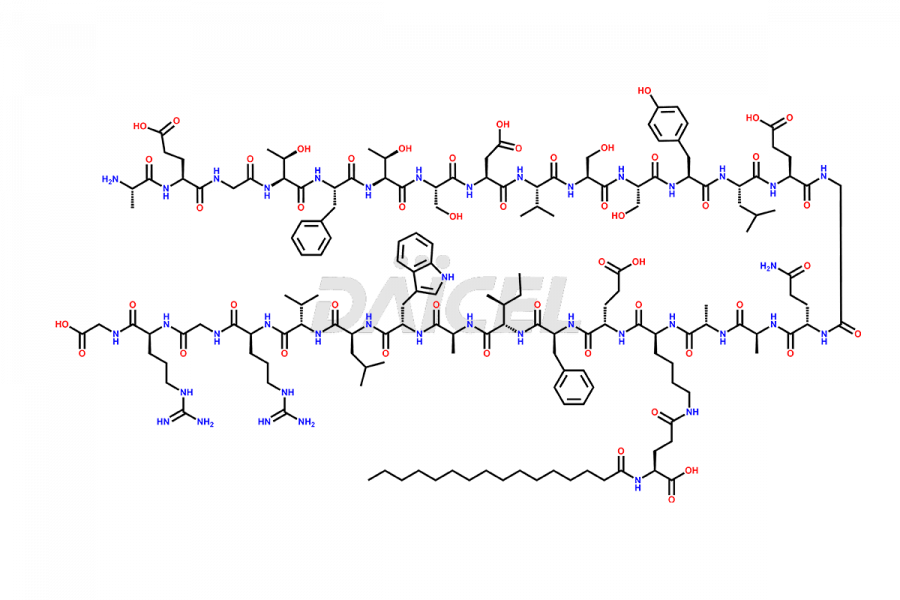
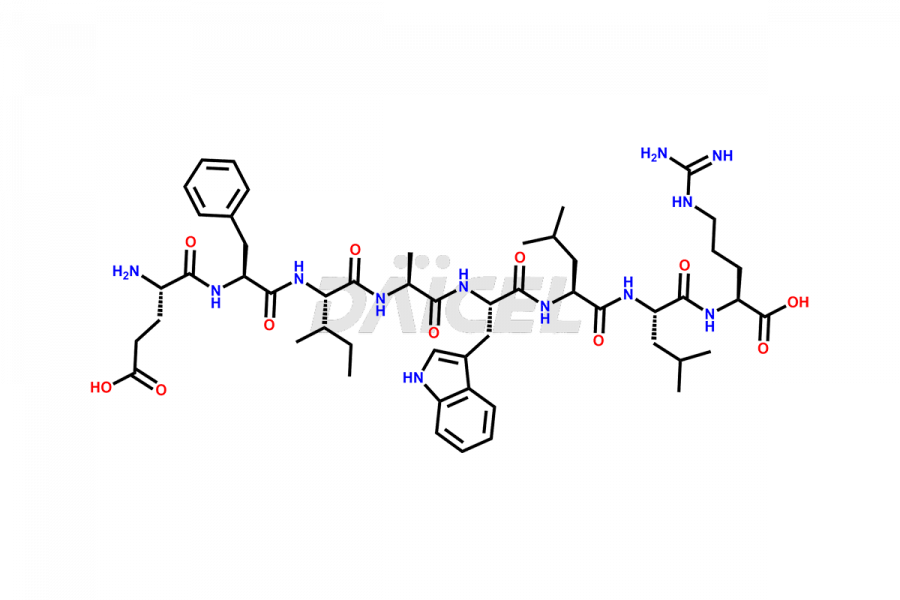
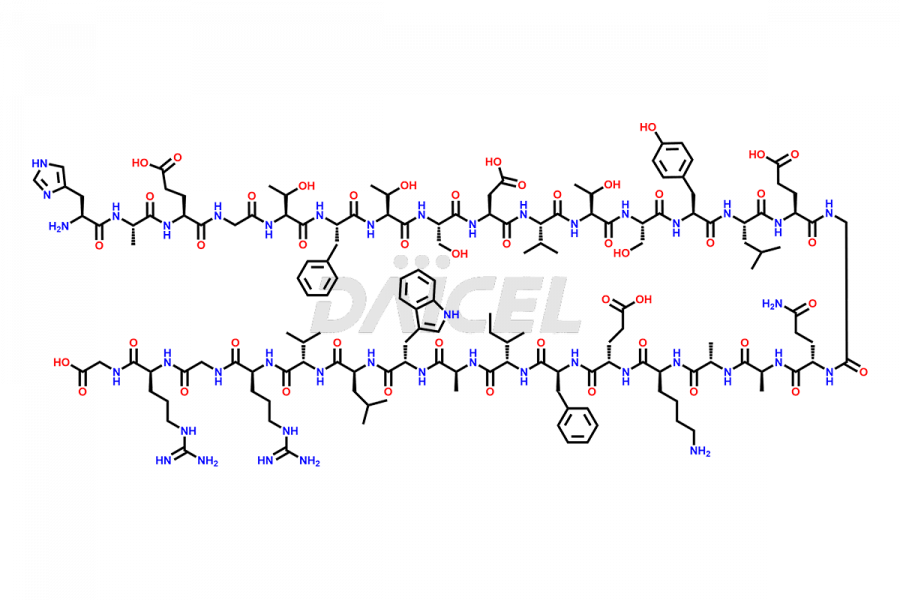
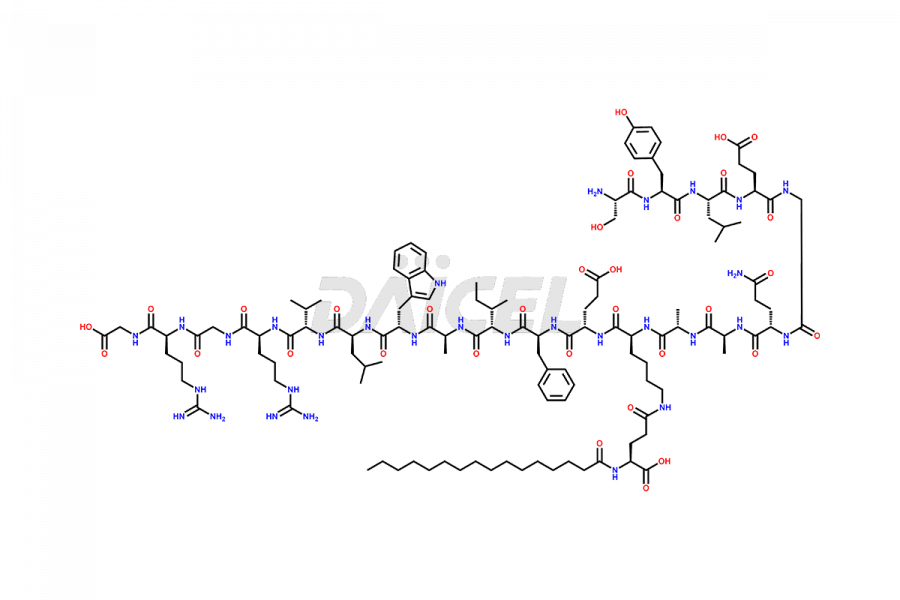
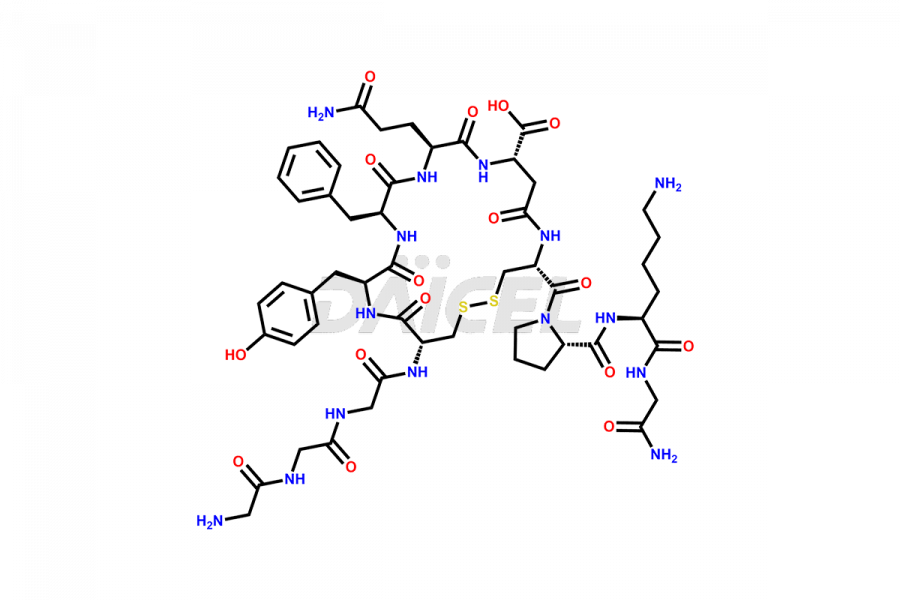
[11-31]-Liraglutide
- CAT Number DCTI-D-000420
- CAS NUMBER NA
- MOLECULAR FORMULA C127H201N31O34
- MOLECULAR WEIGHT 2706.19
[12-31]-Liraglutide
- CAT Number DCTI-D-000453
- CAS Number NA
- Molecular Formula C124H196N30O32
- Molecular Weight 2619.11
[12-31]-Liraglutide phosphate salt
- CAT Number DCTI-D-000530
- CAS Number NA
- Molecular Formula C124H196N30O32
- Molecular Weight 2619.11
[14-31]-Liraglutide
- CAT Number DCTI-D-000417
- CAS NUMBER NA
- MOLECULAR FORMULA C112H182N28O28
- MOLECULAR WEIGHT 2368.86
[14-31]-Liraglutide
- CAT Number DCTI-D-000417
- CAS Number NA
- Molecular Formula C112H182N28O28
- Molecular Weight 2368.86
[14-31]-Liraglutide (phosphate salt)
- CAT Number DCTI-D-000504
- CAS NUMBER NA
- MOLECULAR FORMULA C112H182N28O28
- MOLECULAR WEIGHT 2368.86
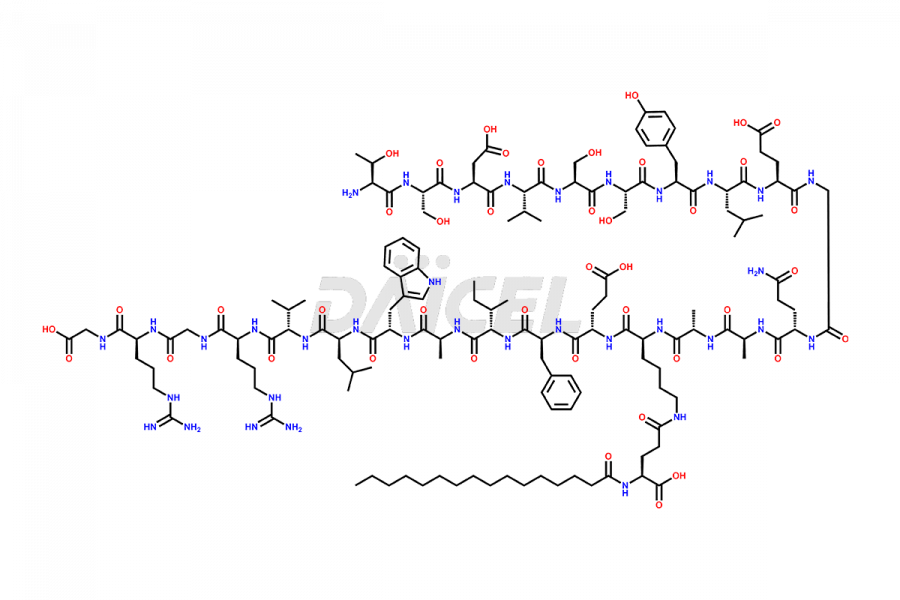
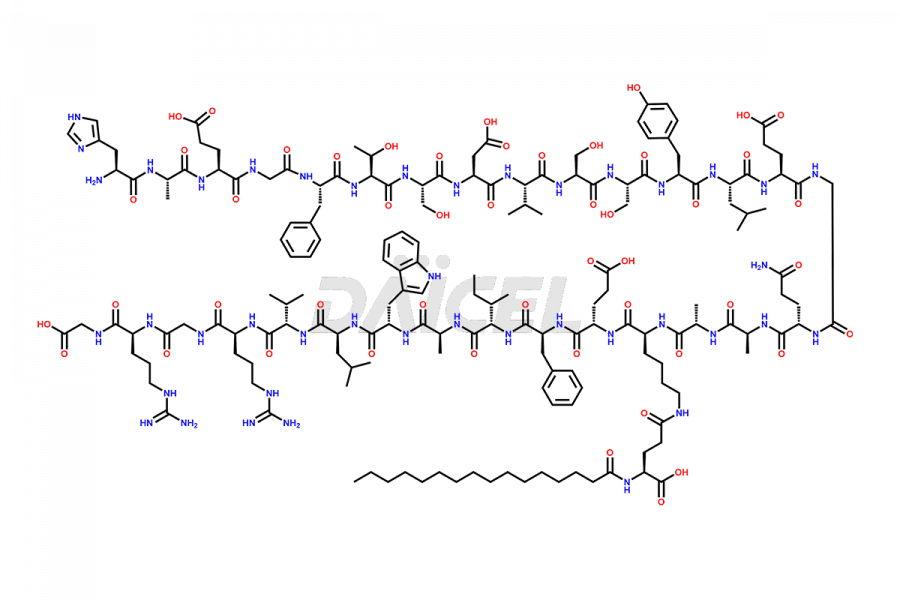
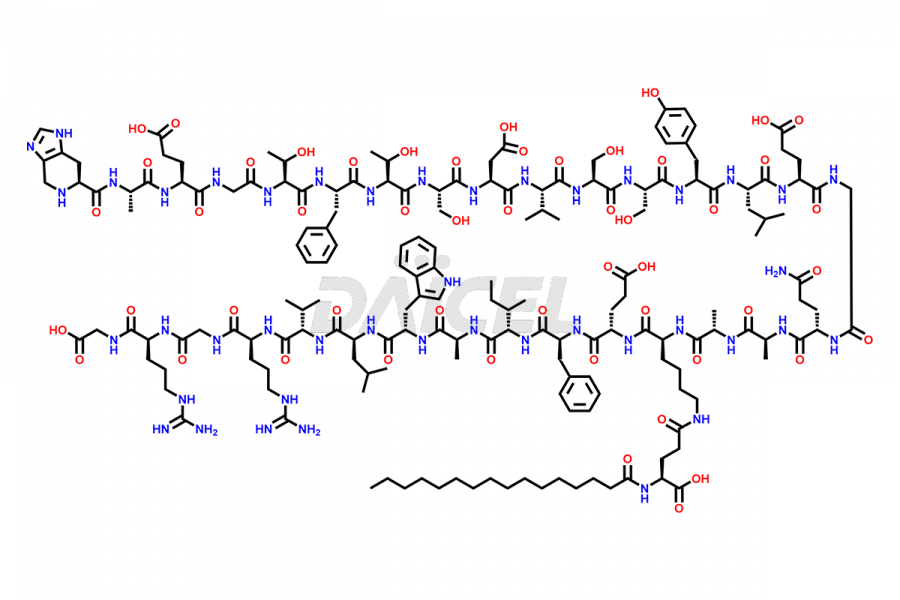
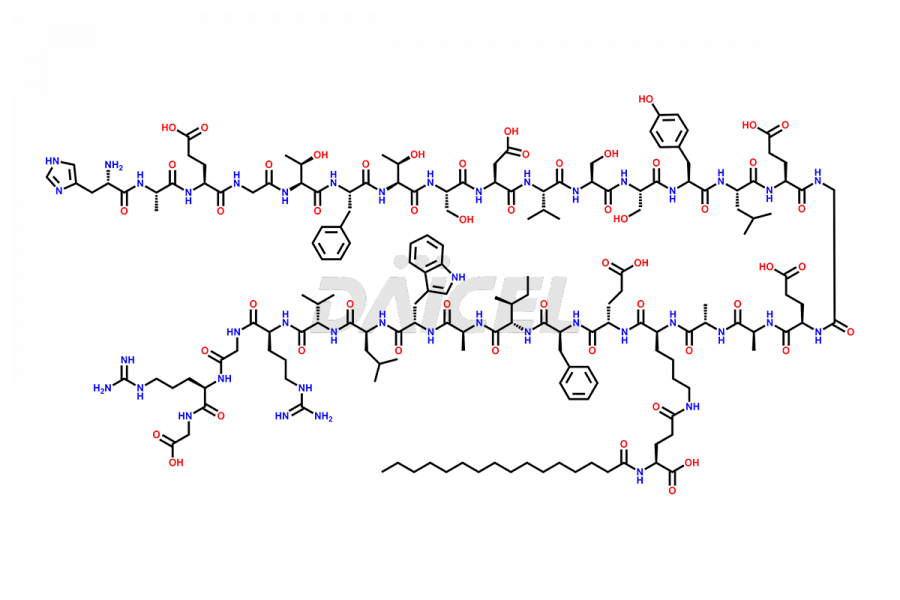
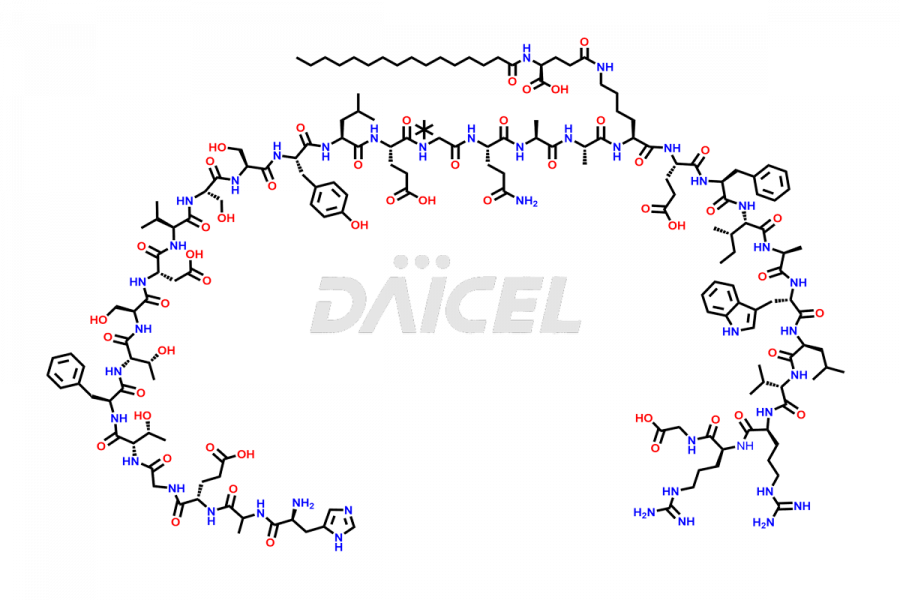
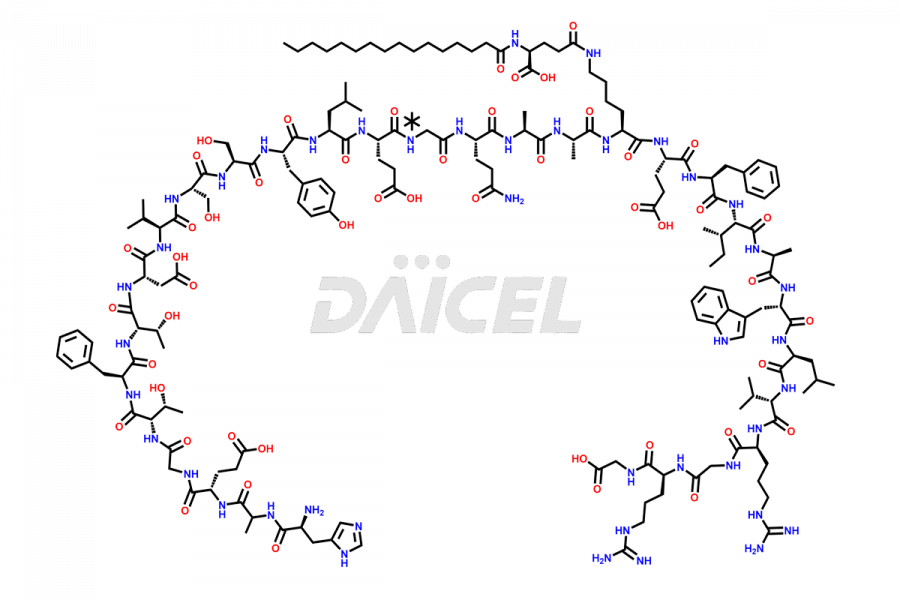
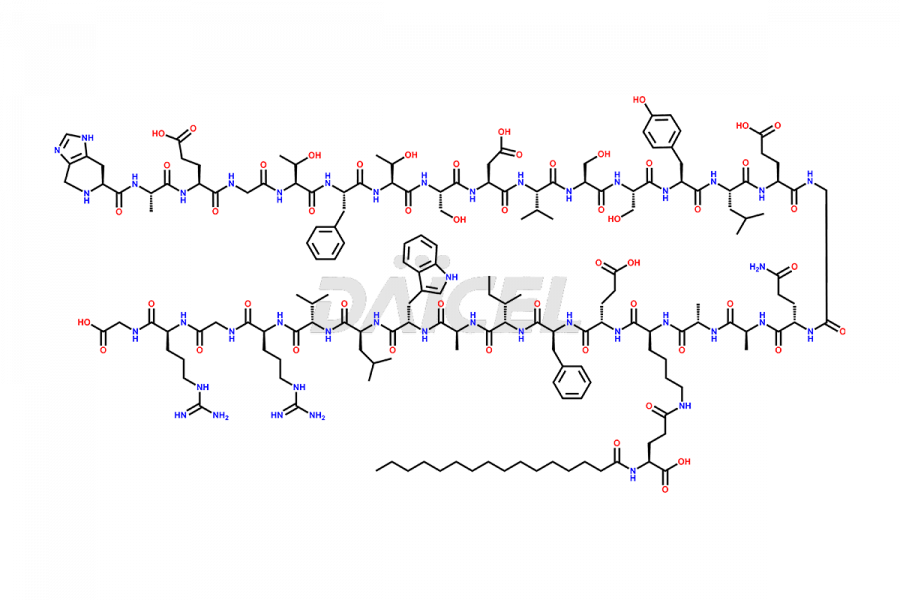
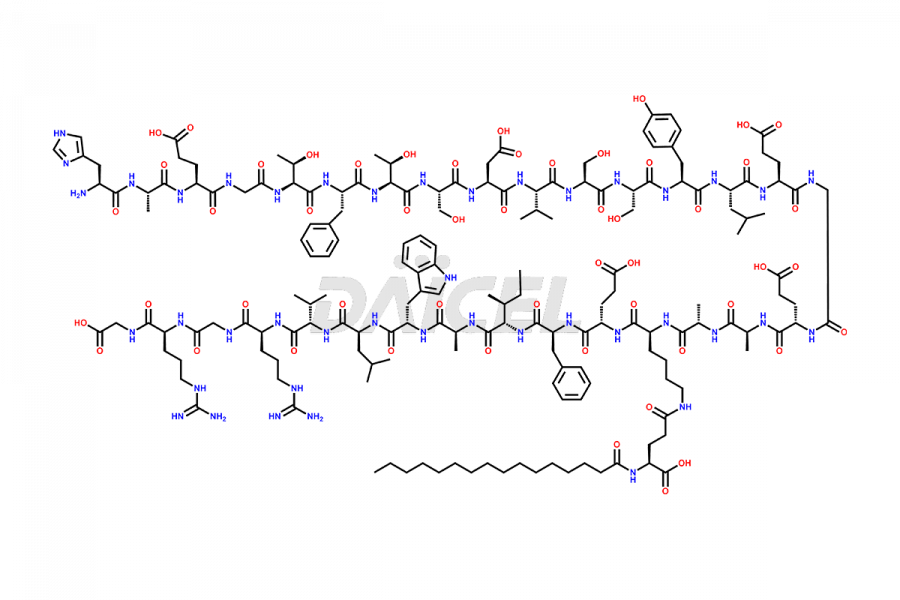
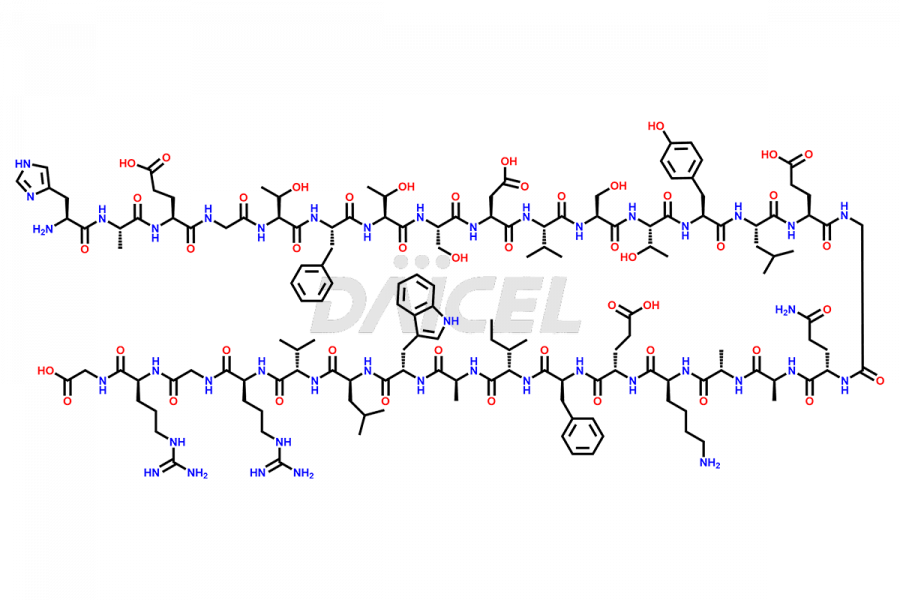
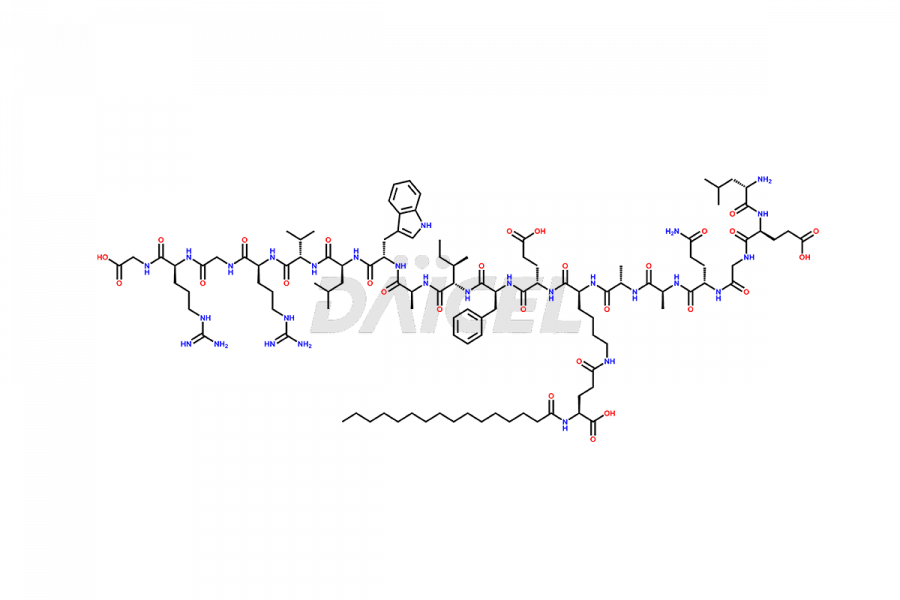
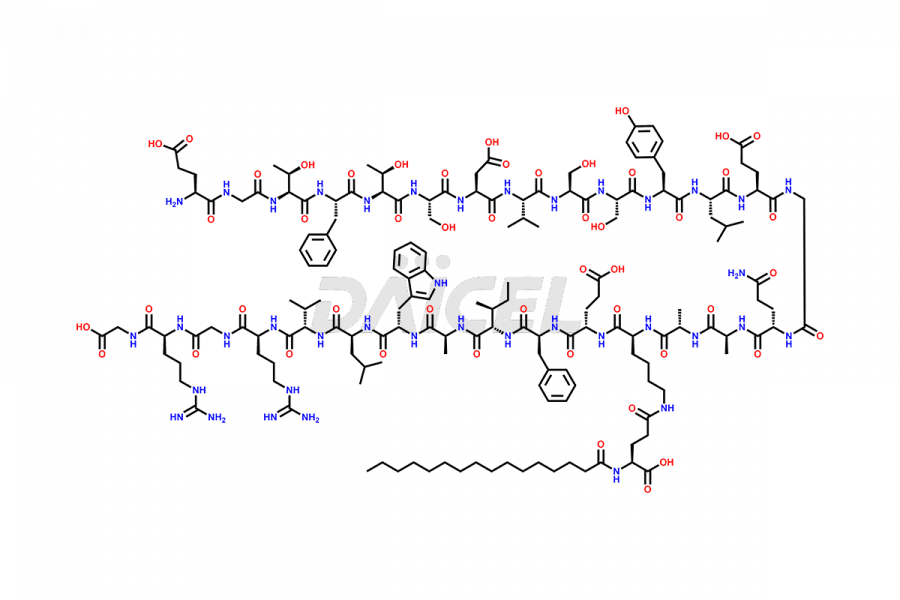
[3-31]-Liraglutide
- CAT Number DCTI-D-000225
- CAS NUMBER NA
- MOLECULAR FORMULA C163H253N39O49
- MOLECULAR WEIGHT 3543.04
[3-31]-Liraglutide Phosphate salt
- CAT Number DCTI-D-000538
- CAS Number NA
- Molecular Formula C163H253N39O49
- Molecular Weight 3543.04
[4-31]-Liraglutide
- CAT Number DCTI-D-000210
- CAS NUMBER NA
- MOLECULAR FORMULA C158H246N38O46
- MOLECULAR WEIGHT 3413.93
[4-31]-Liraglutide-Phosphate Salt
- CAT Number DCTI-D-000535
- CAS Number NA
- Molecular Formula C158H246N38O46
- Molecular Weight 3413.93
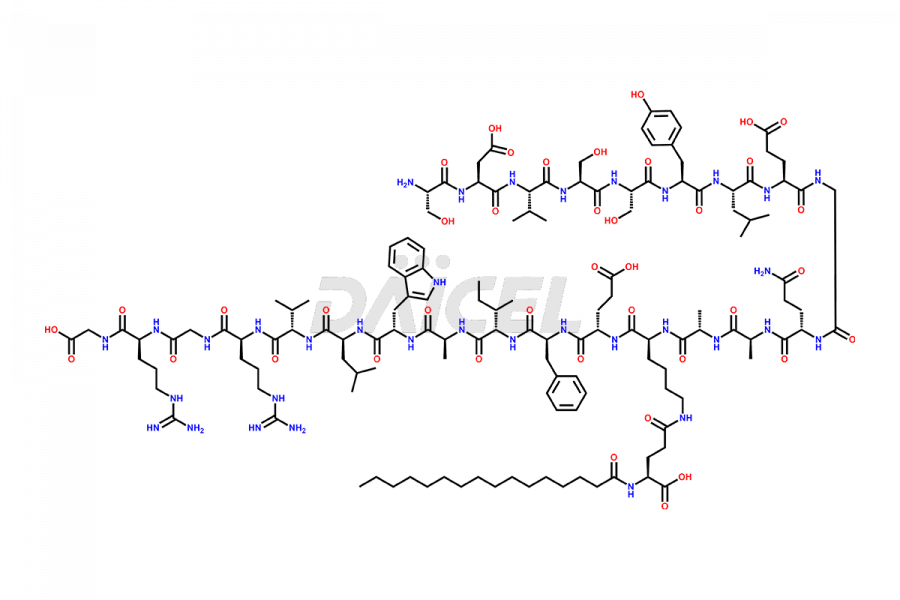
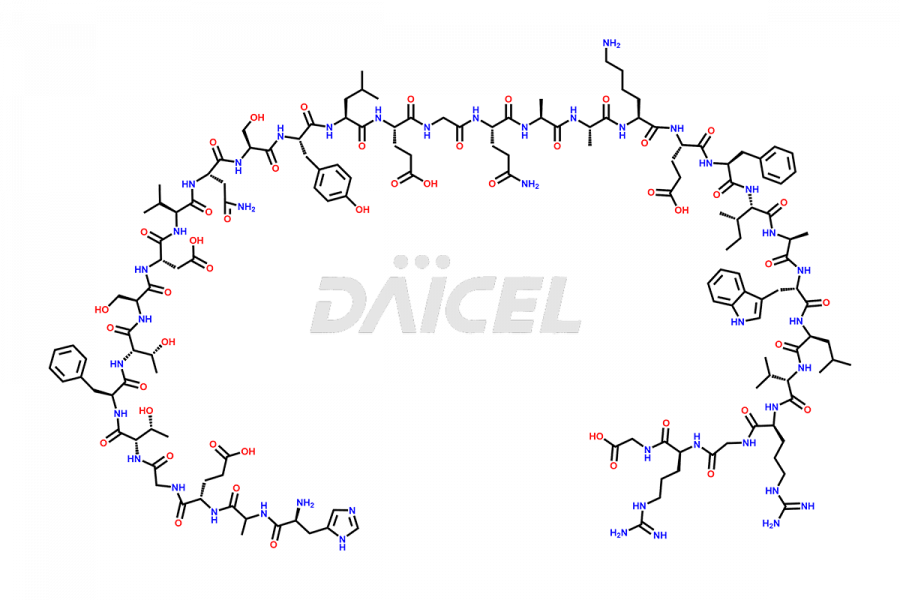
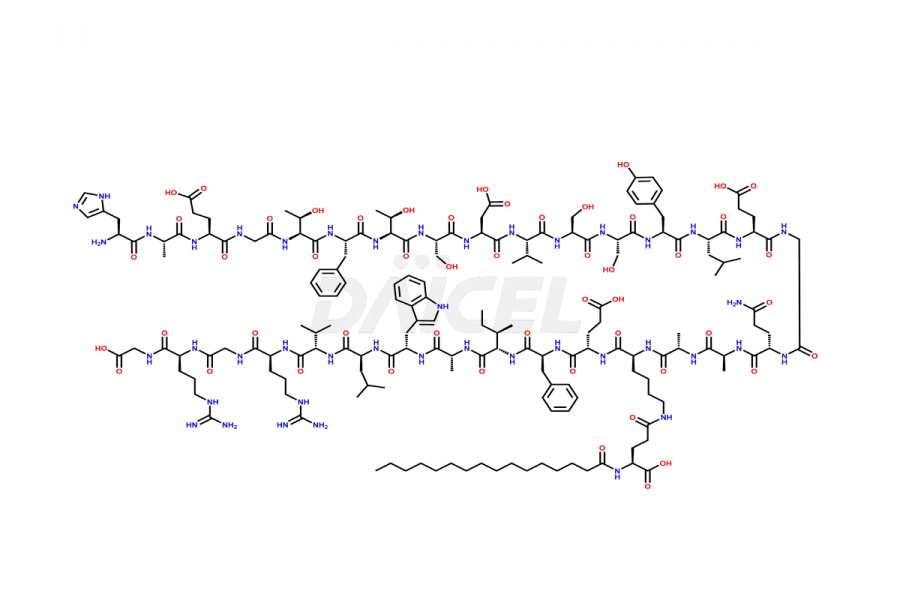
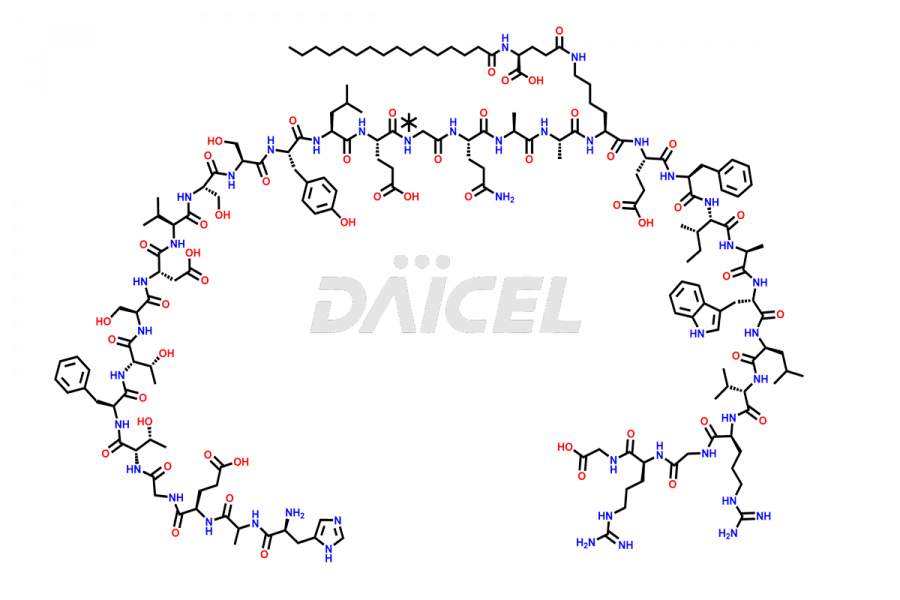
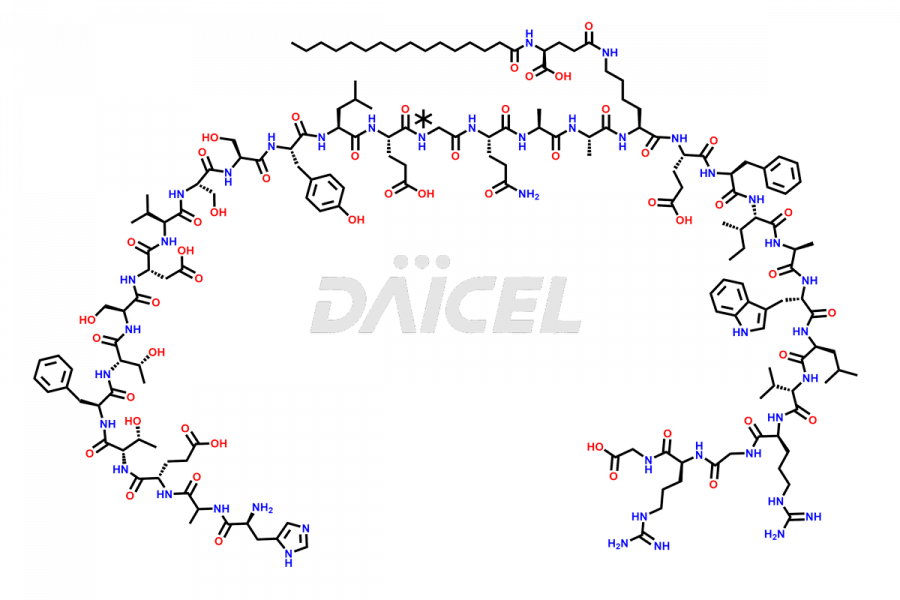
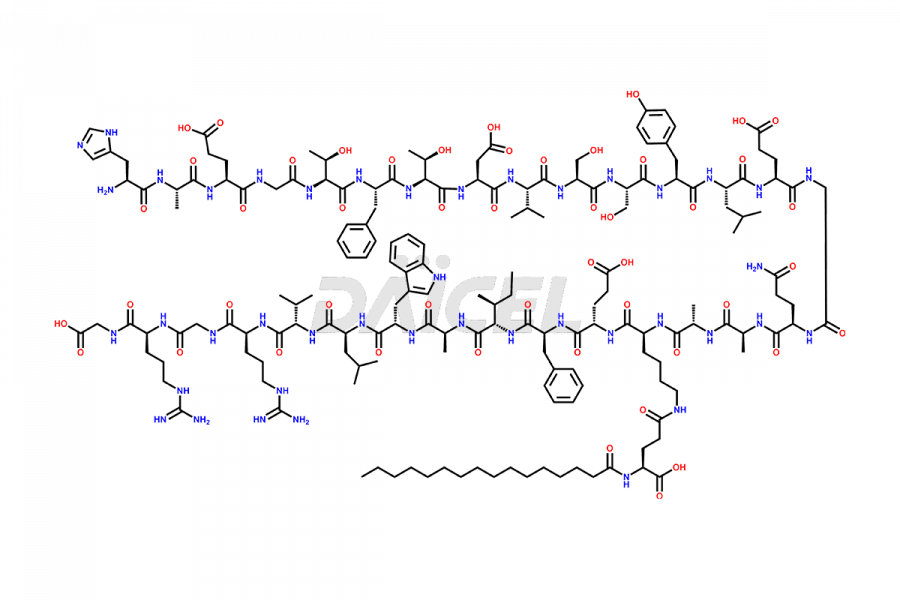
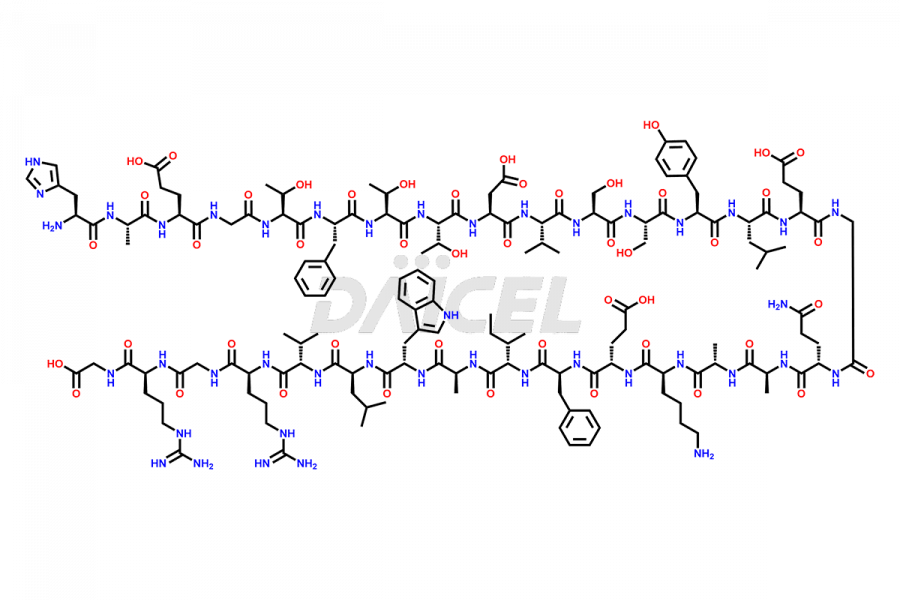
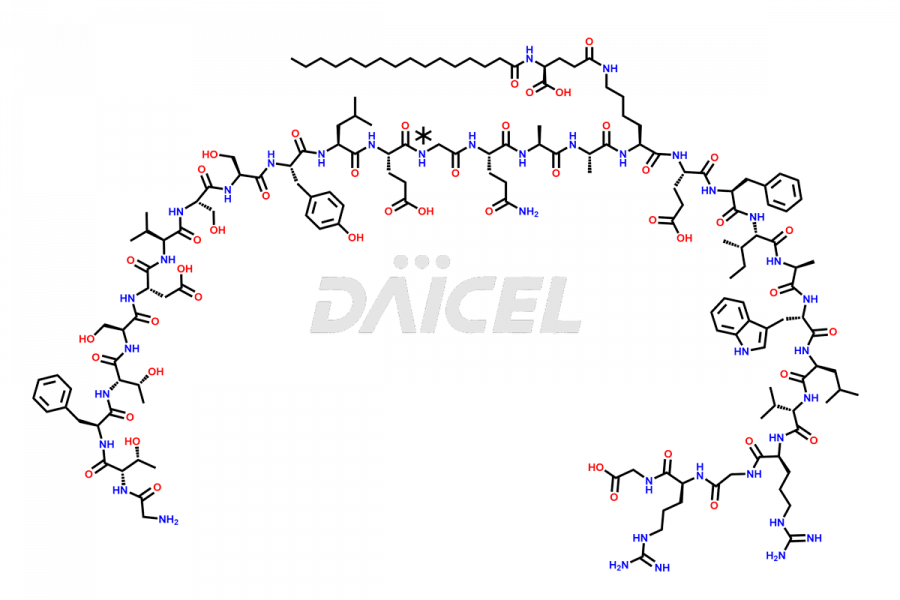
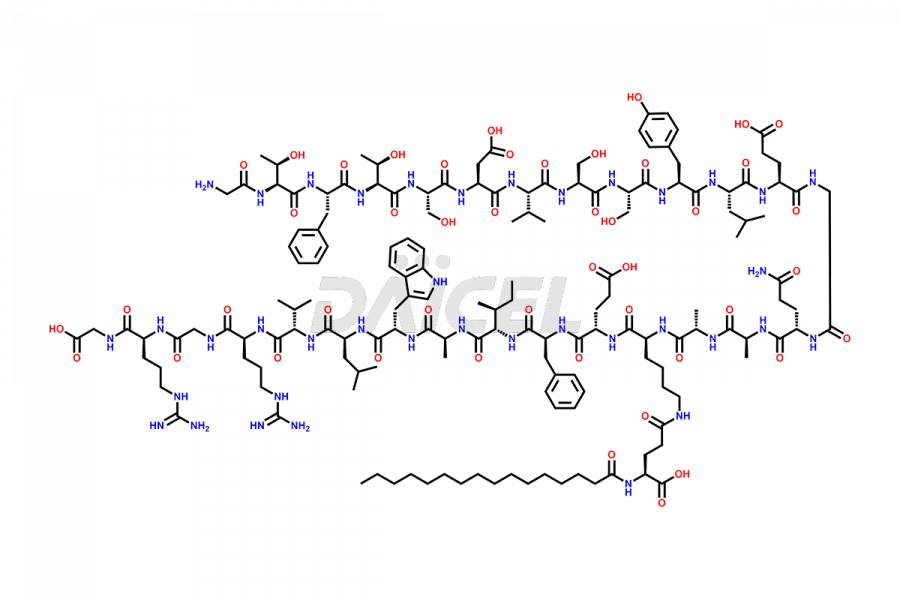
[5-31]-Liraglutide
- CAT Number DCTI-D-000191
- CAS NUMBER NA
- MOLECULAR FORMULA C156H243N37O45
- MOLECULAR WEIGHT 3356.87
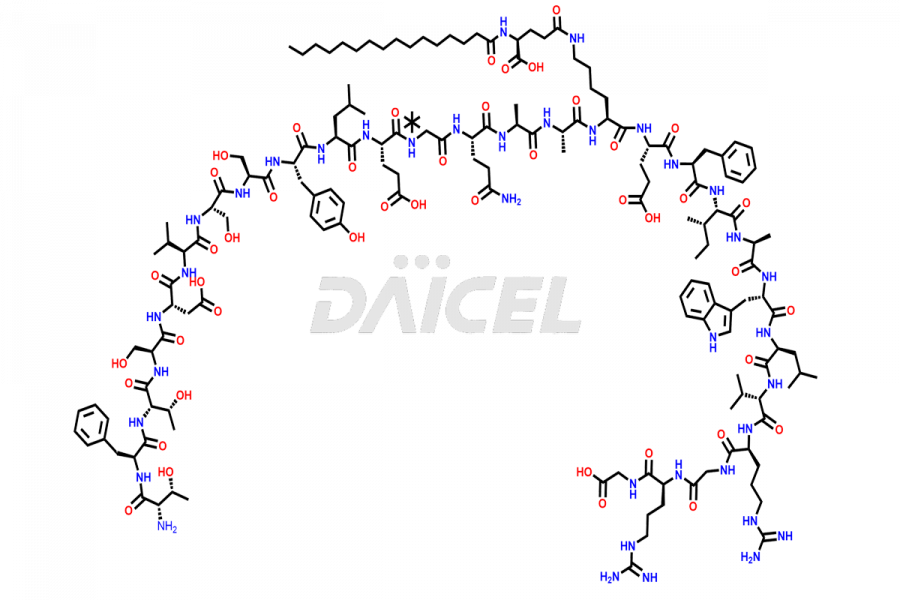
[5-31]-Liraglutide Phosphate Salt
- CAT Number DCTI-D-000581
- CAS Number NA
- Molecular Formula C156H243N37O45
- Molecular Weight 3356.87
[6-31]-Liraglutide-Phosphate Salt
- CAT Number DCTI-D-000534
- CAS Number NA
- Molecular Formula C152H236N36O43
- Molecular Weight 3255.77
[7-31]-Liraglutide
- CAT Number DCTI-D-000390
- CAS NUMBER NA
- MOLECULAR FORMULA C143H227N35O42
- MOLECULAR WEIGHT 3108.59
[7-31]-Liraglutide phosphate salt
- CAT Number DCTI-D-000536
- CAS Number NA
- Molecular Formula C143H227N35O42
- Molecular Weight 3108.59
[8-31]-Liraglutide
- CAT Number DCTI-D-000435
- CAS NUMBER NA
- MOLECULAR FORMULA C139H220N34O40
- MOLECULAR WEIGHT 3007.49
[8-31]-Liraglutide-Phosphate salt
- CAT Number DCTI-D-000547
- CAS Number NA
- Molecular Formula C139H220N34O40
- Molecular Weight 3007.49
[D-allo-Thr]13-Liraglutide-Phosphate salt
- CAT Number DCTI-D-000543
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
[Des-Thr5]-Liraglutide
- CAT Number DCTI-D-000391
- CAS NUMBER NA
- MOLECULAR FORMULA C168H258N42O49
- MOLECULAR WEIGHT 3650.16
Asn(11)-Linear Liraglutide
- CAT Number DCTI-D-000304
- CAS NUMBER NA
- MOLECULAR FORMULA C152H229N43O47
- MOLECULAR WEIGHT 3410.76
Asn(12)-Linear Liraglutide
- CAT Number DCTI-D-000303
- CAS NUMBER NA
- MOLECULAR FORMULA C152H229N43O47
- MOLECULAR WEIGHT 3410.76
Asn(8)-Linear Liraglutide
- CAT Number DCTI-D-000305
- CAS NUMBER NA
- MOLECULAR FORMULA C152H229N43O47
- MOLECULAR WEIGHT 3410.76
Beta-Asp Linear Liraglutide-1
- CAT Number DCTI-D-000376
- CAS NUMBER NA
- MOLECULAR FORMULA C151H228N42O47
- MOLECULAR WEIGHT 3383.73
Cyclic-His-Liraglutide
- CAT Number DCTI-D-000378
- CAS NUMBER NA
- MOLECULAR FORMULA C173H265N43O51
- MOLECULAR WEIGHT 3763.27
D-Ala (24)-Liraglutide
- CAT Number DCTI-D-000750
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Ala-(19)-Liraglutide. Phosphate salt
- CAT Number DCTI-D-000737
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Ala(18)-Liraglutide
- CAT Number DCTI-D-000749
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Ala(2)-Liraglutide
- CAT Number DCTI-D-000722
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Allo-Thr(11)-Liraglutide
- CAT Number DCTI-D-000206
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.56
D-Allo-Thr(13)-Liraglutide
- CAT Number DCTI-D-000199
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.56
D-Arg (30)-Liraglutide
- CAT Number DCTI-D-000773
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Arg(28)-Liraglutide
- CAT Number DCTI-D-000151
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Gln(17)-Liraglutide
- CAT Number DCTI-D-000401
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Glu (3)-Liraglutide (phosphate salt)
- CAT Number DCTI-D-000772
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Glu-(17)-Liraglutide
- CAT Number DCTI-D-000426
- CAS NUMBER NA
- MOLECULAR FORMULA C172H264N42O52
- MOLECULAR WEIGHT 3752.25
D-Glu-(17)-Liraglutide Phosphate salt
- CAT Number DCTI-D-000556
- CAS Number NA
- Molecular Formula C172H264N42O52
- Molecular Weight 3752.25
D-Glu(3)-Liraglutide
- CAT Number DCTI-D-000152
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Glu(3)-Liraglutide.Phosphate salt
- CAT Number DCTI-D-000735
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Glu(side chain)-Lira
- CAT Number DCTI-D-000257
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-His(1)-Liraglutide
- CAT Number DCTI-D-000153
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-His(1)-Liraglutide (phosphate salt)
- CAT Number DCTI-D-000507
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Ile(23)-Liraglutide phosphate salt
- CAT Number DCTI-D-000558
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Ile(29)-Liraglutide
- CAT Number DCTI-D-000433
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Leu(14)-Liraglutide
- CAT Number DCTI-D-000753
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Leu(26)-Liraglutide
- CAT Number DCTI-D-000751
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Phe(6)-Liraglutide
- CAT Number DCTI-D-000473
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Phe(6)-Liraglutide-Phosphate salt
- CAT Number DCTI-D-000548
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Ser (12)-Liraglutide (Phosphate salt)
- CAT Number DCTI-D-000780
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Ser(12)-Liraglutide. Phosphate Salt
- CAT Number DCTI-D-000738
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Ser(17)-Liraglutide
- CAT Number DCTI-D-000192
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.56
D-Ser(18)-Liraglutide
- CAT Number DCTI-D-000198
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.56
D-Ser(8)-Liraglutide
- CAT Number DCTI-D-000154
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Thr(5)-Liraglutide
- CAT Number DCTI-D-000726
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.56
D-Thr(7)-Liraglutide
- CAT Number DCTI-D-000476
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.26
D-Thr(7)-Liraglutide-Phosphate salt
- CAT Number DCTI-D-000544
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Trp(25)-Liraglutide
- CAT Number DCTI-D-000752
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.26
D-Tyr(13)-Liraglutide
- CAT Number DCTI-D-000744
- CAS Number NA
- Molecular Formula C172H265N43O51
- Molecular Weight 3751.56
D-Val(16)-Liraglutide
- CAT Number DCTI-D-000211
- CAS NUMBER NA
- MOLECULAR FORMULA C172H265N43O51
- MOLECULAR WEIGHT 3751.56
Des-Ala(2)-Liraglutide
- CAT Number DCTI-D-000200
- CAS NUMBER NA
- MOLECULAR FORMULA C169H260N42O50
- MOLECULAR WEIGHT 3680.18
Des-Gly (29)-Liraglutide
- CAT Number DCTI-D-000140
- CAS NUMBER NA
- MOLECULAR FORMULA C170H262N42O50
- MOLECULAR WEIGHT 3694.21
Des-Gly (4)-Liraglutide
- CAT Number DCTI-D-000138
- CAS NUMBER NA
- MOLECULAR FORMULA C170H262N42O50
- MOLECULAR WEIGHT 3694.21
Des-Gly(31)-Liraglutide
- CAT Number DCTI-D-000155
- CAS NUMBER NA
- MOLECULAR FORMULA C170H262N42O50
- MOLECULAR WEIGHT 3694.21
Des-His(1)-Liraglutide
- CAT Number DCTI-D-000227
- CAS NUMBER NA
- MOLECULAR FORMULA C166H258N40O50
- MOLECULAR WEIGHT 3614.12
Des-His(1)-Liraglutide. Phosphate salt
- CAT Number DCTI-D-000545
- CAS Number NA
- Molecular Formula C166H258N40O50
- Molecular Weight 3614.12
Des-Ser (8)-Liraglutide
- CAT Number DCTI-D-000141
- CAS NUMBER NA
- MOLECULAR FORMULA C169H260N42O49
- MOLECULAR WEIGHT 3664.18
Des-Ser(8)-Liraglutide Phosphate Salt
- CAT Number DCTI-D-000563
- CAS NUMBER NA
- MOLECULAR FORMULA C169H260N42O49
- MOLECULAR WEIGHT 3664.18
Des-Thr(5)-Liraglutide
- CAT Number DCTI-D-000193
- CAS NUMBER NA
- MOLECULAR FORMULA C168H258N42O49
- MOLECULAR WEIGHT 3650.16
Des-Thr(7)-Liraglutide
- CAT Number DCTI-D-000379
- CAS NUMBER NA
- MOLECULAR FORMULA C168H258N42O49
- MOLECULAR WEIGHT 3650.16
Des-Thr(7)-Liraglutide phosphate salt
- CAT Number DCTI-D-000539
- CAS Number NA
- Molecular Formula C168H258N42O49
- Molecular Weight 3650.16
Endo Thr(7)-Liraglutide. Phosphate salt
- CAT Number DCTI-D-000549
- CAS Number NA
- Molecular Formula C176H272N44O53
- Molecular Weight 3852.37
Endo-Ala(18)-Liraglutide
- CAT Number DCTI-D-000641
- CAS Number NA
- Molecular Formula C175H270N44O52
- Molecular Weight 3822.34
Endo-Ala(18)-Liraglutide Phosphate Salt
- CAT Number DCTI-D-000791
- CAS Number NA
- Molecular Formula C175H270N44O52
- Molecular Weight 3822.34
Endo-Ala(18)-Liraglutide. Phosphate Salt
- CAT Number DCTI-D-000736
- CAS Number NA
- Molecular Formula C175H270N44O52
- Molecular Weight 3822.34
Endo-Gly(31)-Liragltuide
- CAT Number DCTI-D-000481
- CAS Number NA
- Molecular Formula C174H268N44O52
- Molecular Weight 3808.31
Endo-Gly(31)-Liraglutide phosphate salt
- CAT Number DCTI-D-000529
- CAS Number NA
- Molecular Formula C174H268N44O52
- Molecular Weight 3808.31
Endo-Thr(7)-Liraglutide
- CAT Number DCTI-D-000384
- CAS NUMBER NA
- MOLECULAR FORMULA C176H272N44O53
- MOLECULAR WEIGHT 3852.37
Formaldehyde Adduct-Liraglutide
- CAT Number DCTI-D-000230
- CAS NUMBER NA
- MOLECULAR FORMULA C173H265N43O51
- MOLECULAR WEIGHT 3763.27 Da
Formaldehyde-Schiff base-Liraglutide Phosphate Salt
- CAT Number DCTI-D-000559
- CAS Number NA
- Molecular Formula C173H265N43O51
- Molecular Weight 3763.27
Fragment 21-28 Leu Liraglutide
- CAT Number DCTI-D-000506
- CAS NUMBER NA
- MOLECULAR FORMULA C52H78N12O11
- MOLECULAR WEIGHT 1047.27
Glu (17)-Liraglutide
- CAT Number DCTI-D-000137
- CAS NUMBER NA
- MOLECULAR FORMULA C172H264N42O52
- MOLECULAR WEIGHT 3752.25
Glu(17)-Liraglutide (phosphate salt)
- CAT Number DCTI-D-000540
- CAS Number NA
- Molecular Formula C172H264N42O52
- Molecular Weight 3752.25
Glu(9)-Linear Liraglutide
- CAT Number DCTI-D-000307
- CAS NUMBER NA
- MOLECULAR FORMULA C152H230N42O47
- MOLECULAR WEIGHT 3397.76
Kyn(25)-Liraglutide
- CAT Number DCTI-D-000150
- CAS NUMBER NA
- MOLECULAR FORMULA C171H265N43O52
- MOLECULAR WEIGHT 3755.25 Da
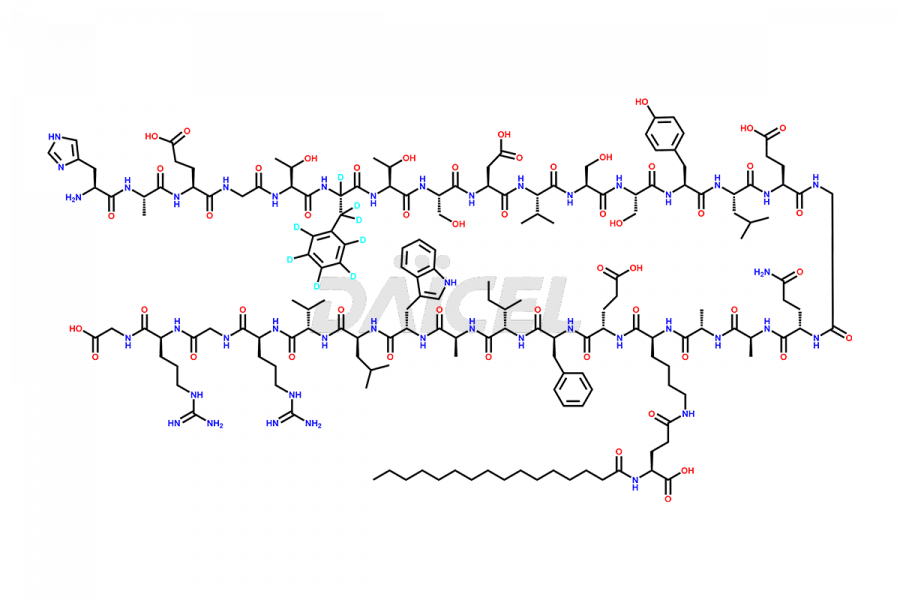
Liraglutide D8
- CAT Number DCTI-D-000316
- CAS NUMBER NA
- MOLECULAR FORMULA C172H257D8N43O51
- MOLECULAR WEIGHT 3759.31 Da
Liraglutide-Acetaldehyde adduct
- CAT Number DCTI-D-000424
- CAS NUMBER NA
- MOLECULAR FORMULA C174H267N43O51
- MOLECULAR WEIGHT 3777.3
Liraglutide-Acetaldehyde adduct Phosphate Salt
- CAT Number DCTI-D-000565
- CAS NUMBER NA
- MOLECULAR FORMULA C174H267N43O51
- MOLECULAR WEIGHT 3777.3
Liraglutide-Propionaldehyde adduct
- CAT Number DCTI-D-000440
- CAS NUMBER NA
- MOLECULAR FORMULA C175H269N43O51
- MOLECULAR WEIGHT 3791.33
Liraglutide-Propionaldehyde adduct Phosphate Salt
- CAT Number DCTI-D-000576
- CAS Number NA
- Molecular Formula C175H269N43O51
- Molecular Weight 3791.33
N-Acetyl-Liraglutide
- CAT Number DCTI-D-000423
- CAS NUMBER NA
- MOLECULAR FORMULA C174H267N43O52
- MOLECULAR WEIGHT 3793.3
N-Acetyl-Liraglutide Phosphate Salt
- CAT Number DCTI-D-000560
- CAS NUMBER NA
- MOLECULAR FORMULA C174H267N43O52
- MOLECULAR WEIGHT 3793.3
Thr(11)-Linear Liraglutide
- CAT Number DCTI-D-000309
- CAS NUMBER NA
- MOLECULAR FORMULA NA
- MOLECULAR WEIGHT 3397.76
Thr(12)-Linear Liraglutide
- CAT Number DCTI-D-000308
- CAS NUMBER NA
- MOLECULAR FORMULA C152H230N42O47
- MOLECULAR WEIGHT 3397.76
Thr(8)-Linear Liraglutide
- CAT Number DCTI-D-000306
- CAS NUMBER NA
- MOLECULAR FORMULA C152H230N42O47
- MOLECULAR WEIGHT 3397.76
Trp(4-hydroxy benzyl)25-Liraglutide
- CAT Number DCTI-D-000371
- CAS NUMBER NA
- MOLECULAR FORMULA C179H271N43O52
- MOLECULAR WEIGHT 3857.39
Trp(4-hydroxy benzyl)25-Liraglutide (Phosphate salt)
- CAT Number DCTI-D-000541
- CAS Number NA
- Molecular Formula C179H271N43O52
- Molecular Weight 3857.39
LOAD MORE
You're viewed 100 of 107 products

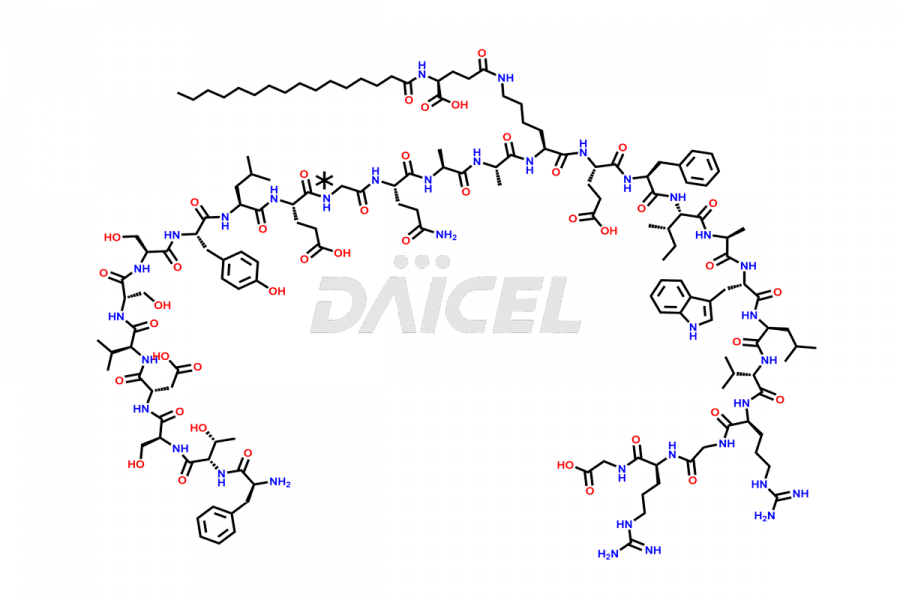
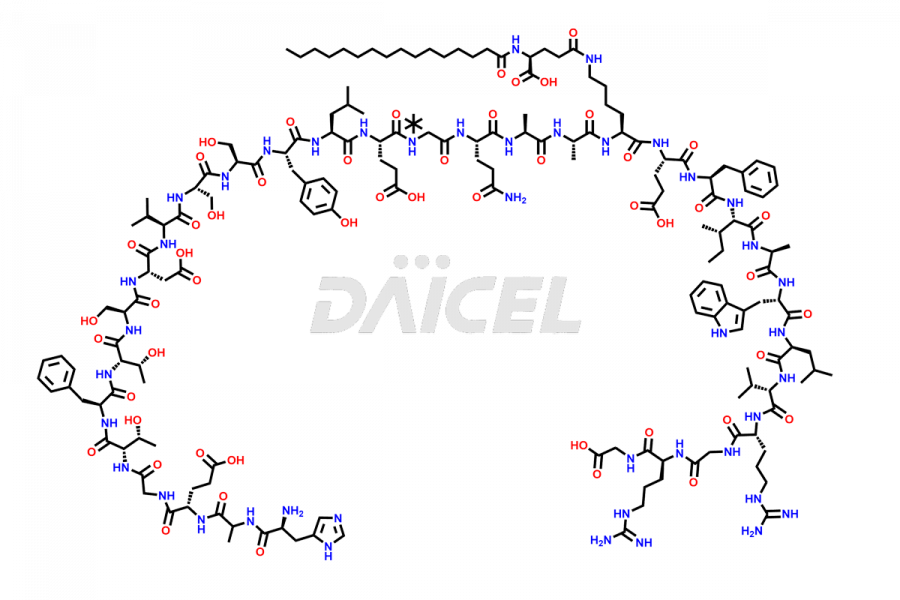
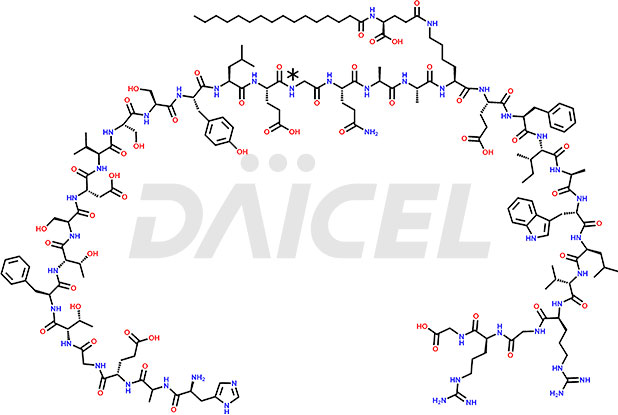
![[10-31]-Liraglutide phosphate salt](https://www.daicelpharmastandards.com/wp-content/uploads/2025/04/dcti-d-537-900x600.png)
![[11-31]-Liraglutide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/10/dcti-d-420-900x600.png)

![[3-31]-Liraglutide](https://www.daicelpharmastandards.com/wp-content/uploads/2023/05/DCTI-D-225-900x600.png)
![[5-31]-Liraglutide Phosphate Salt](https://www.daicelpharmastandards.com/wp-content/uploads/2025/04/dcti-d-581-900x600.png)