Tirzepatide Impurities
Daicel offers a variety of impurities for Tirzepatide, a peptide used in managing type 2 diabetes. Daicel supplies complex tirzepatide impurity standards addressing sequence-related, truncation, and process-related impurities essential for robust GLP-1/GIP dual agonist control.
References
FAQ's
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.

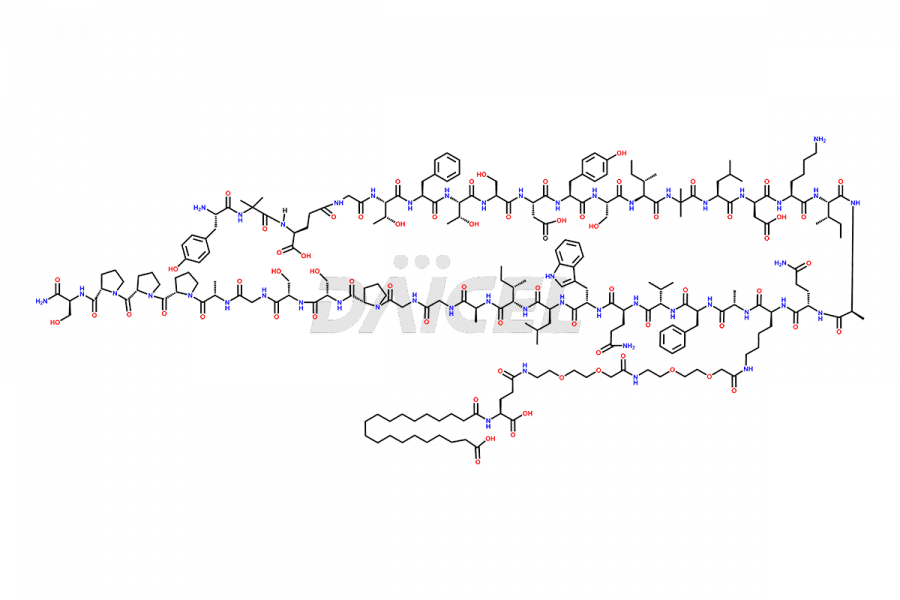
![[D-Asp15]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-312-900x359.png)
![[D-Asp9]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-314-900x359.png)
![[D-Gln19]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-320-900x358.png)
![[D-Gln24]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-318-900x358.png)
![[D-Glu-19]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2025/03/dcti-d-508-900x600.png)
![[D-Glu-3]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-319-900x358.png)
![[D-Leu14]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-311-1-900x358.png)
![[D-Phe22]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-317-900x358.png)
![[D-Phe6]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-332-900x358.png)
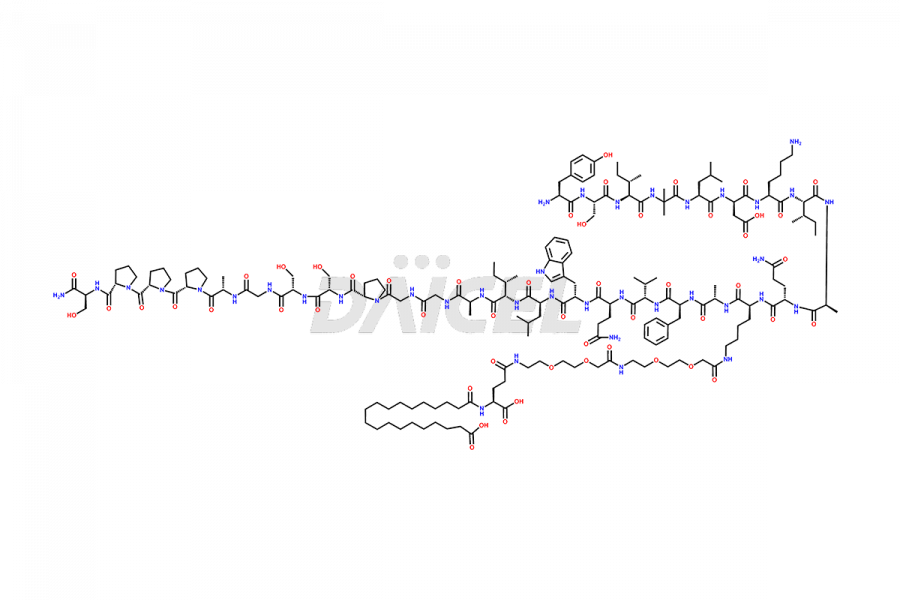
![[D-Ser11]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/05/dcti-d-361-900x600.png)
![[D-Ser8]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/05/dcti-d-358-900x600.png)
![[D-?-Glu-Side chain]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-322-900x358.png)
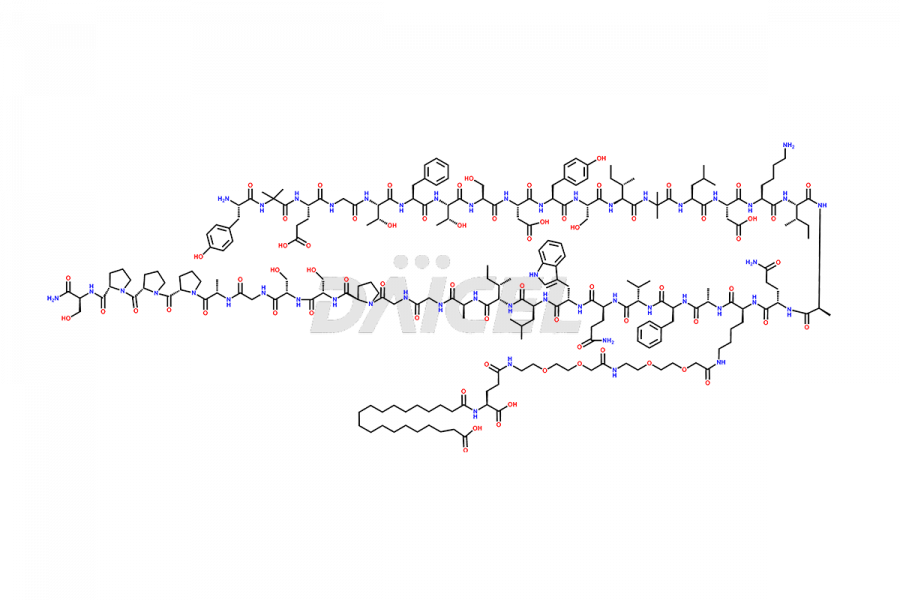
![[Des-Aib2]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/05/dcti-d-364-900x600.png)
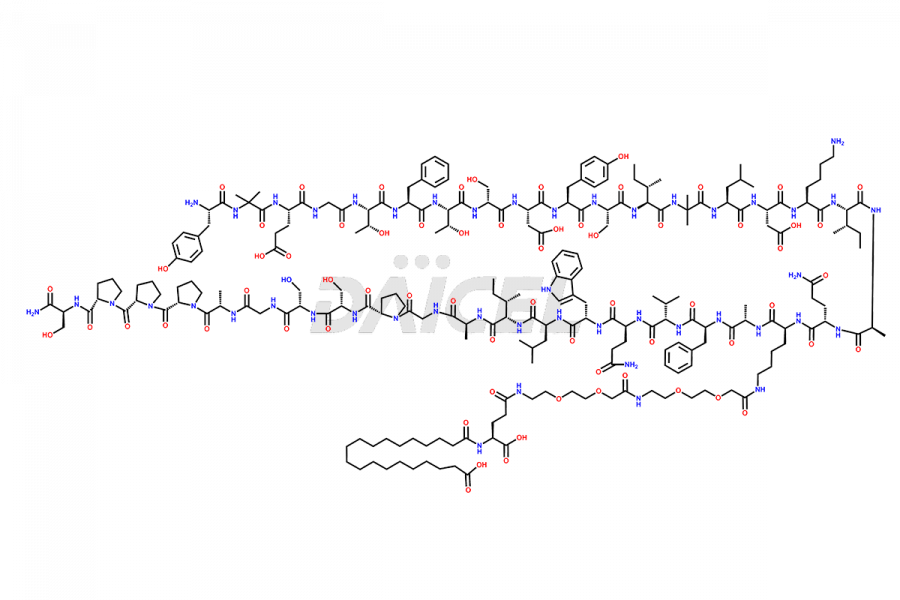
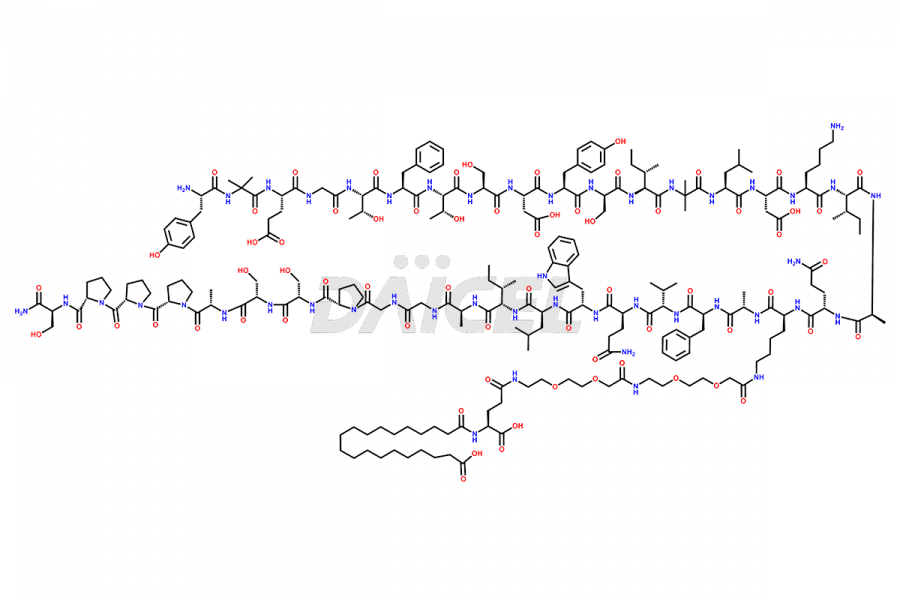
![[Des-Gly4]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/11/dcti-d-436-900x600.png)

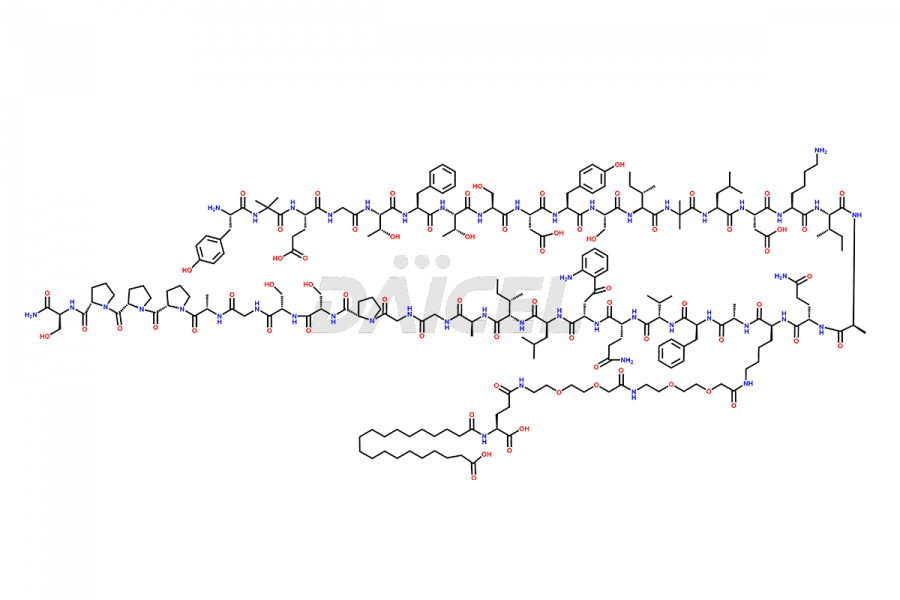
![[?-Ala-35]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-341-900x358.png)
![[?-Ala18]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-321-900x356.png)
![[?-Ala21]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-323-900x358.png)
![[?-Ala28]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-324-900x358.png)
![[?-Asp15]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-344-900x356.png)
![[?-Asp9]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/02/dcti-d-333-900x358.png)
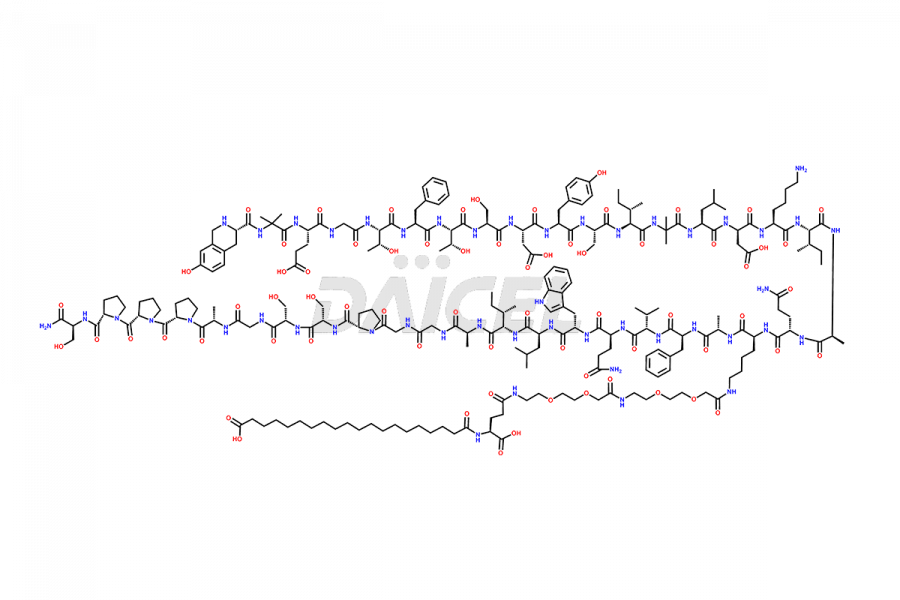
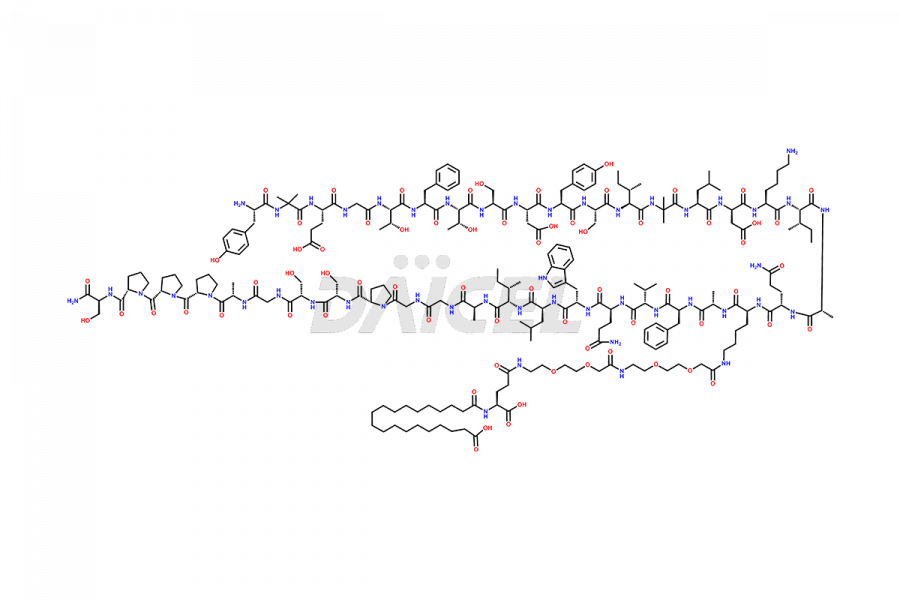

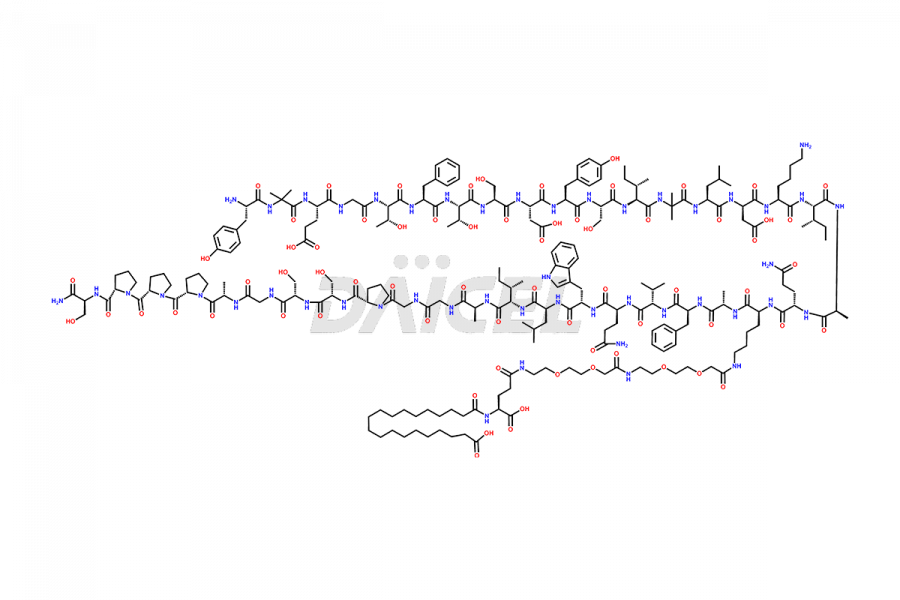
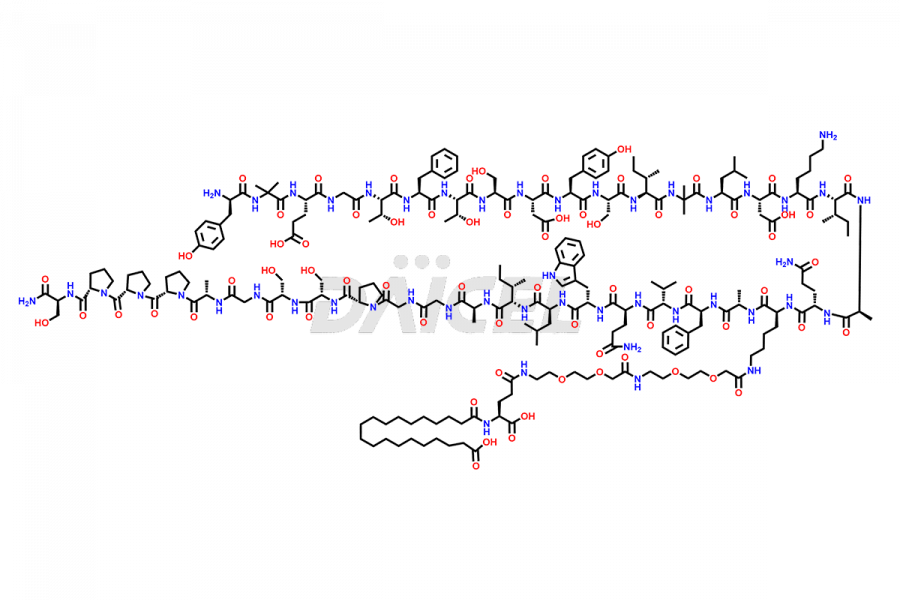
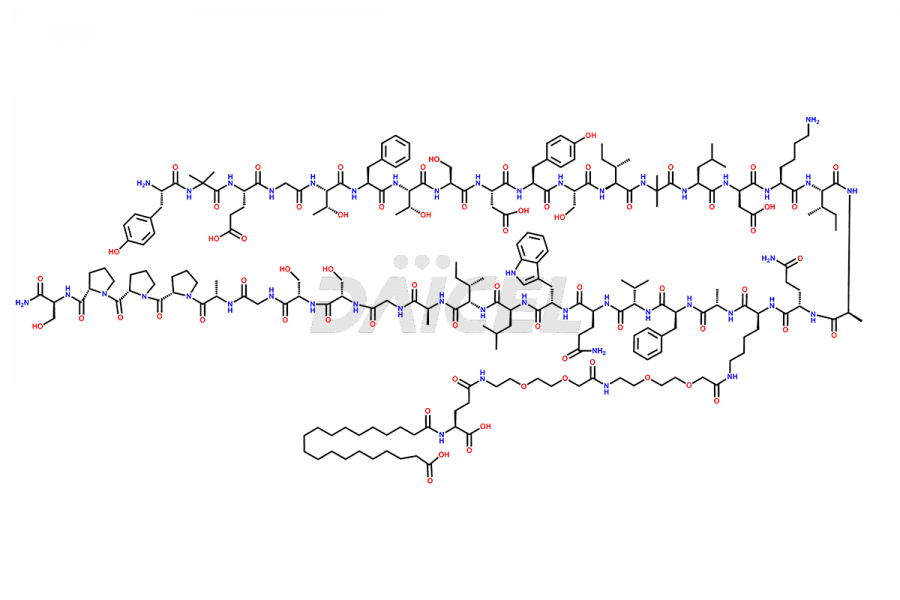
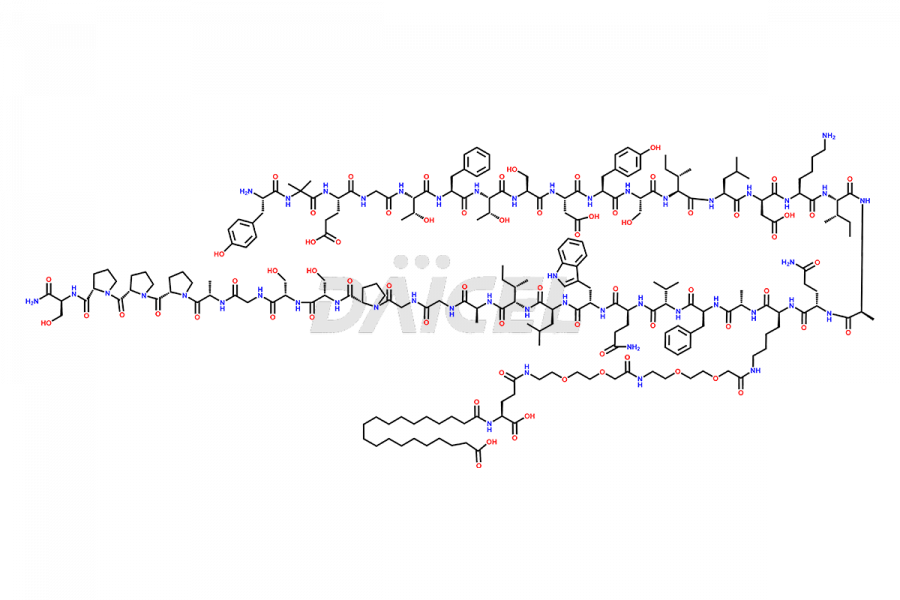

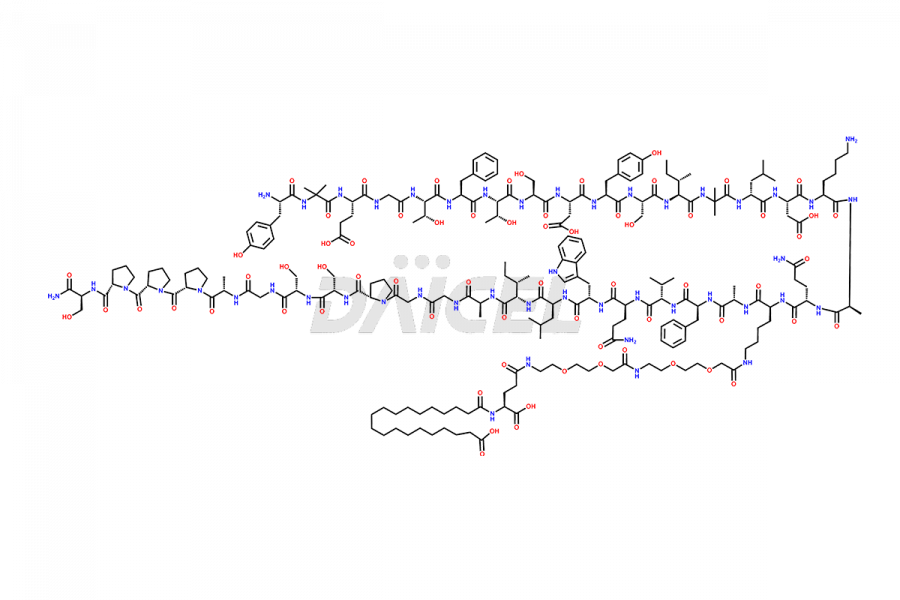
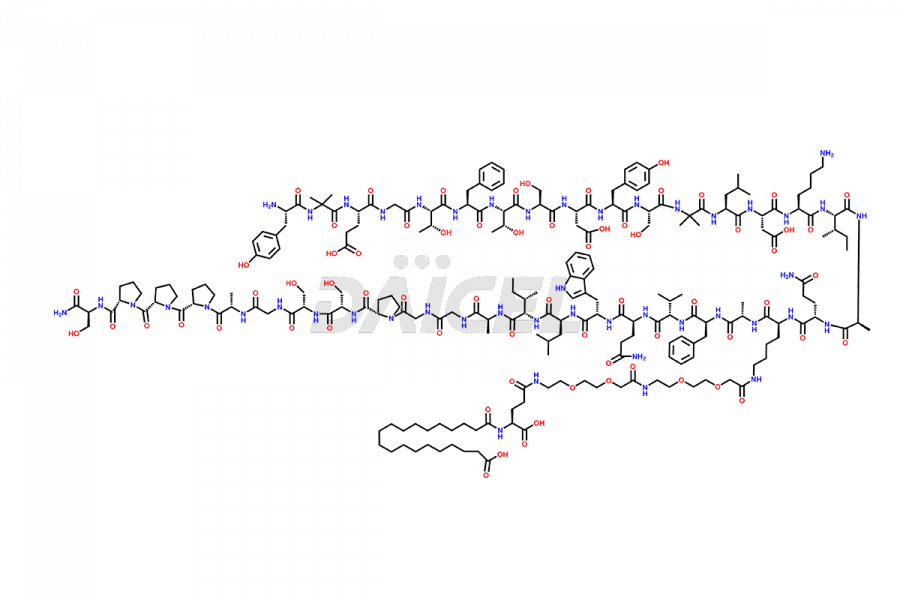
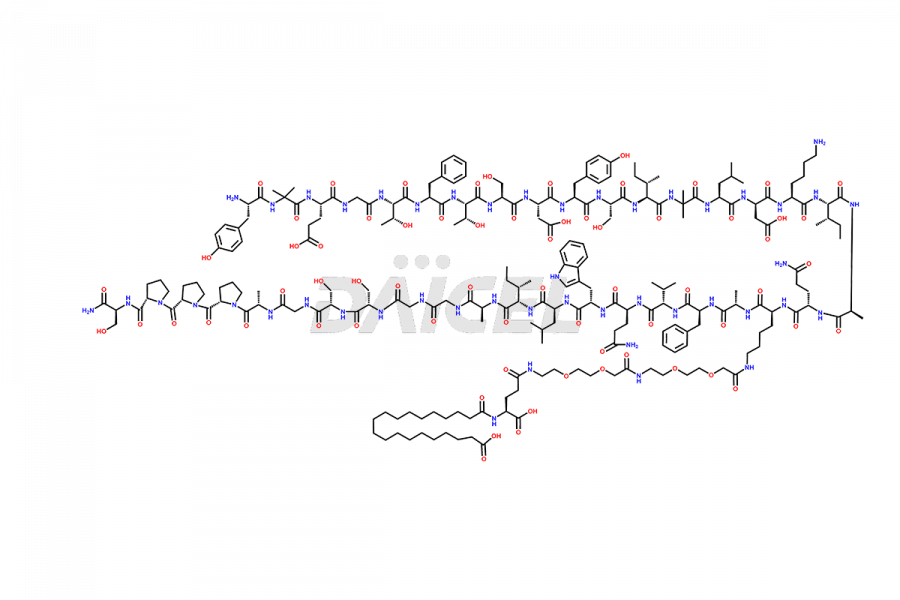
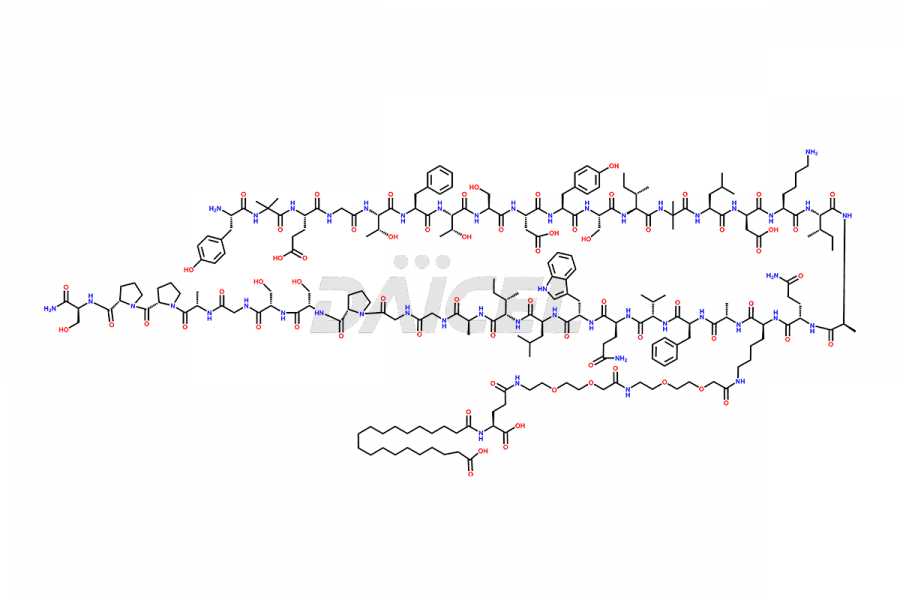
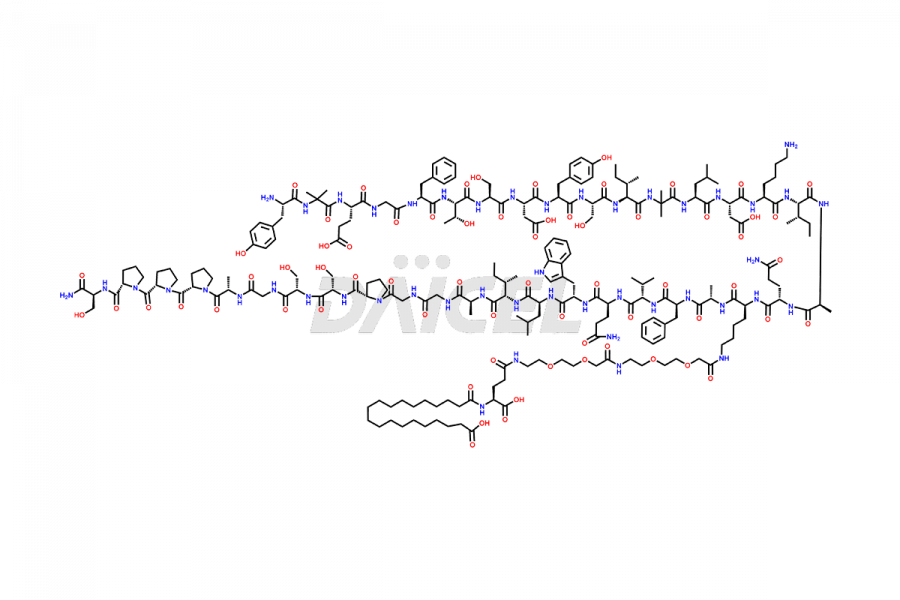
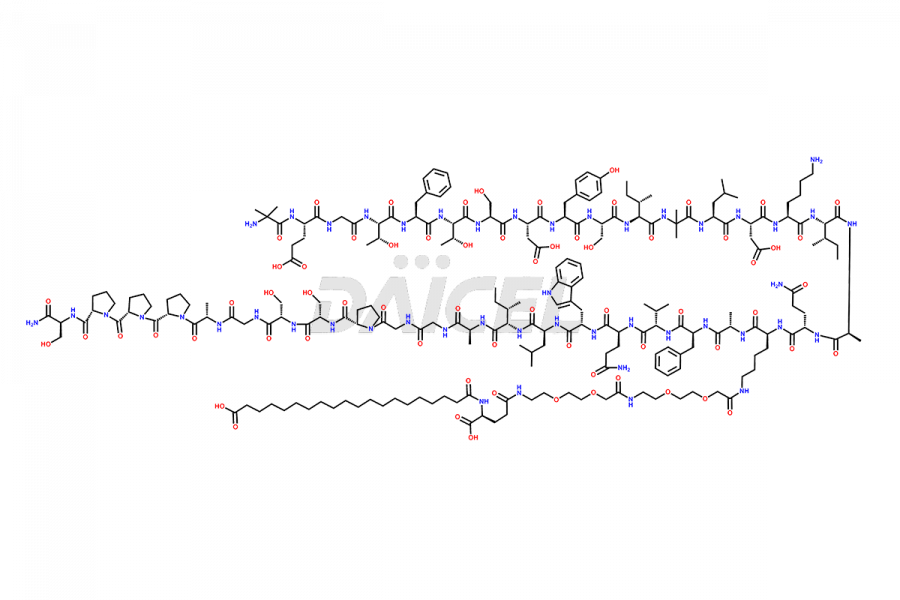

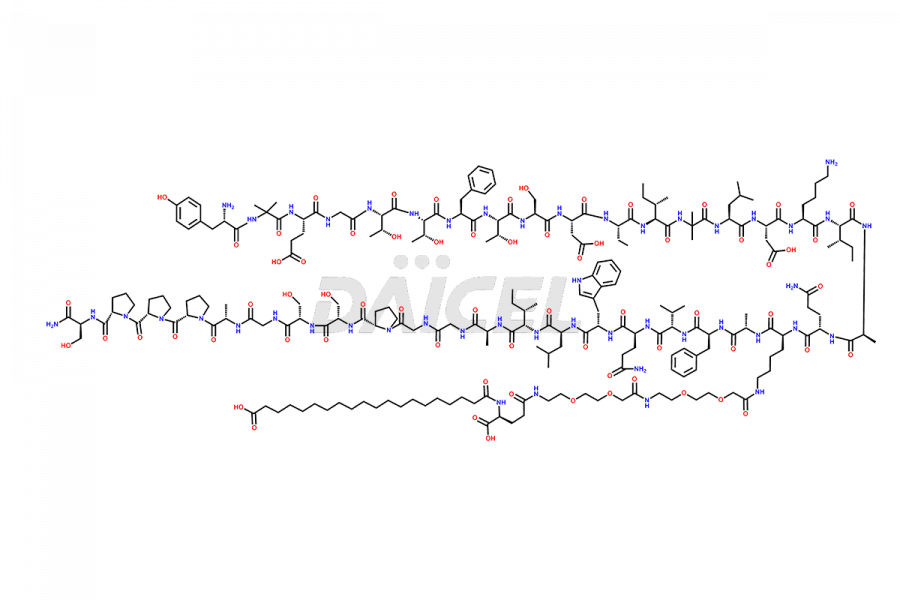
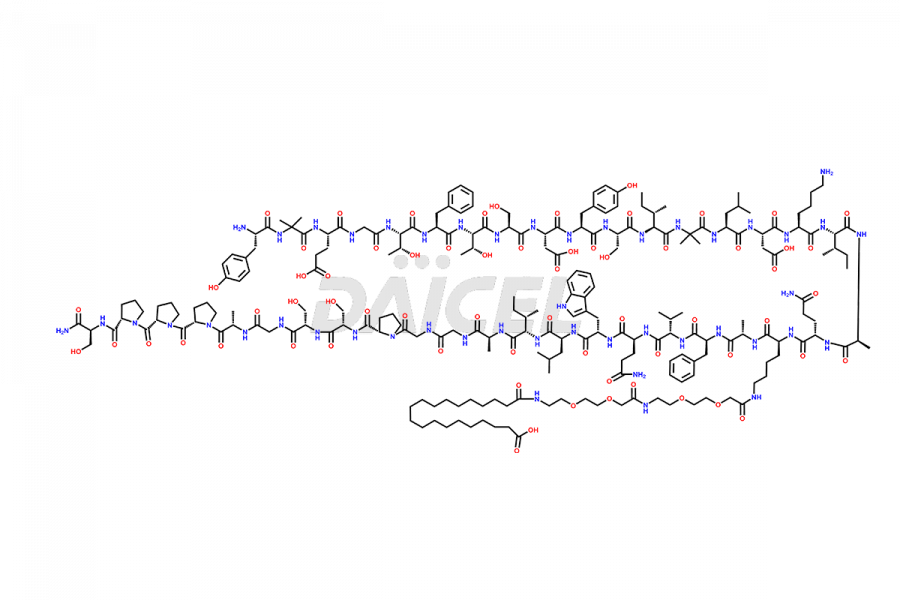
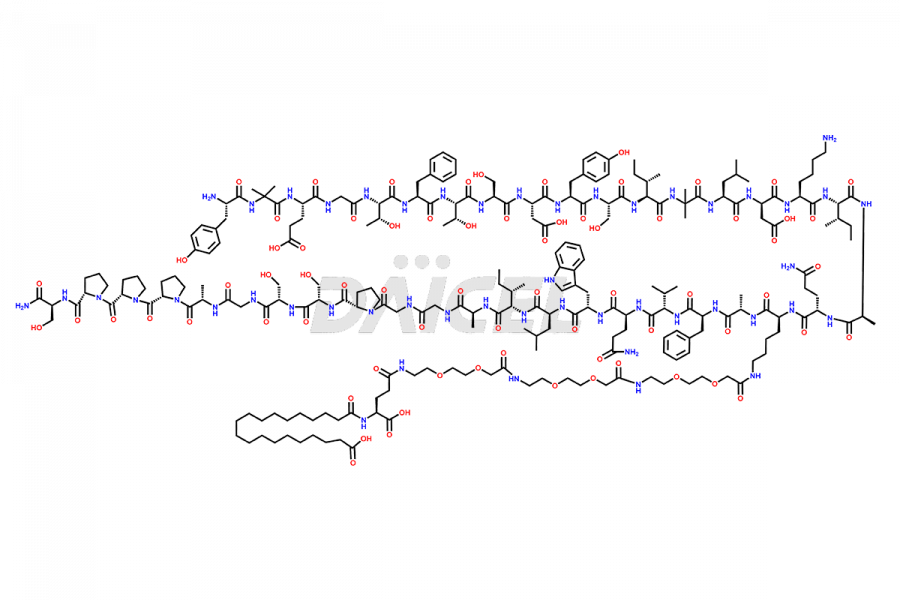
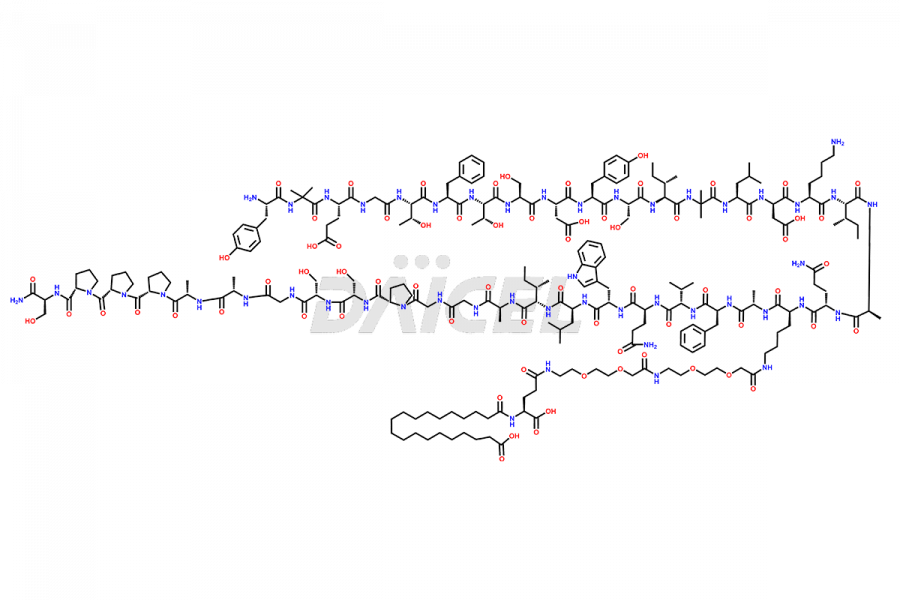
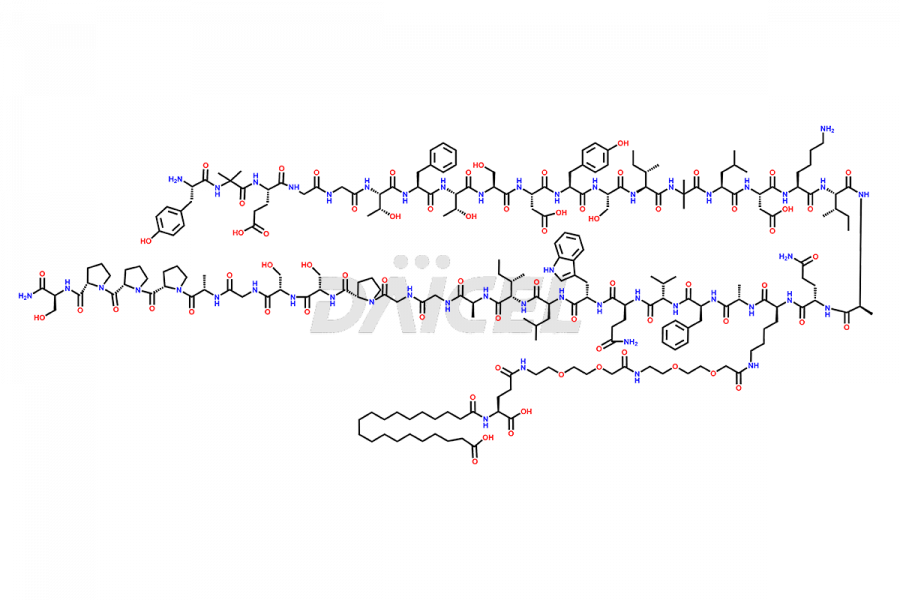

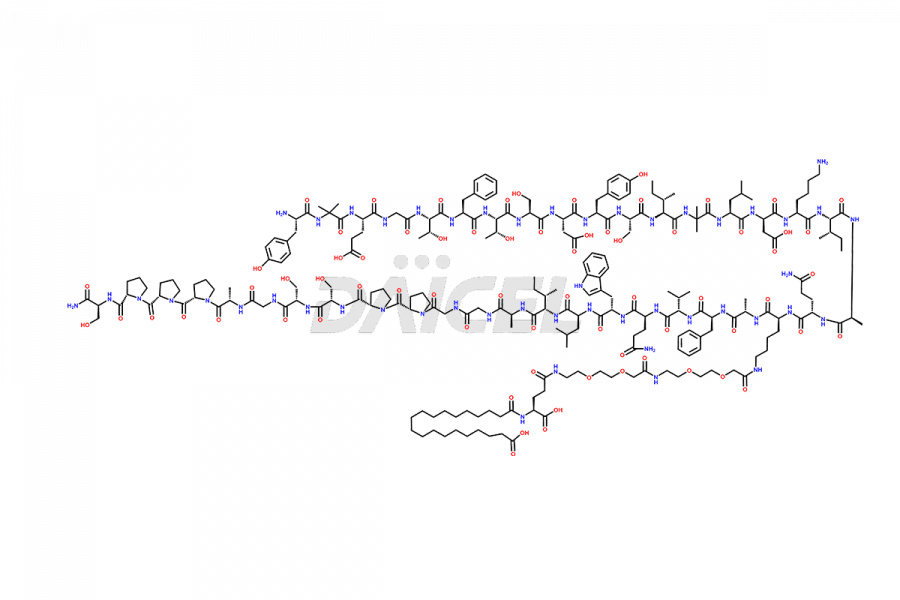
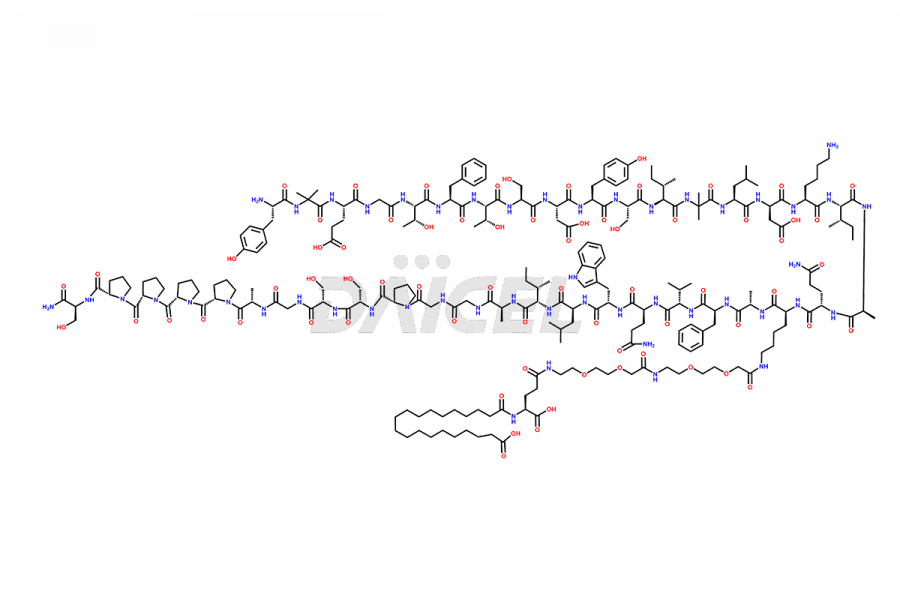
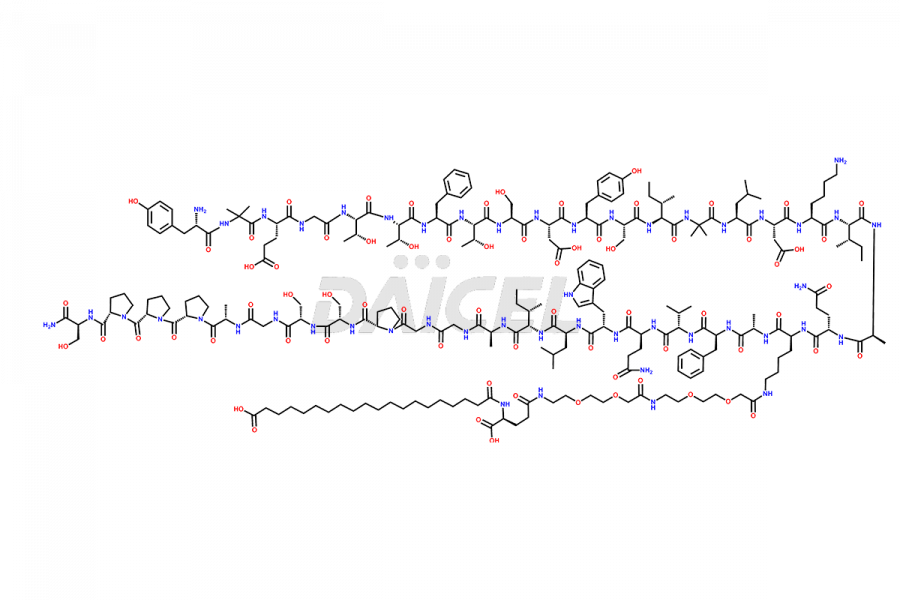
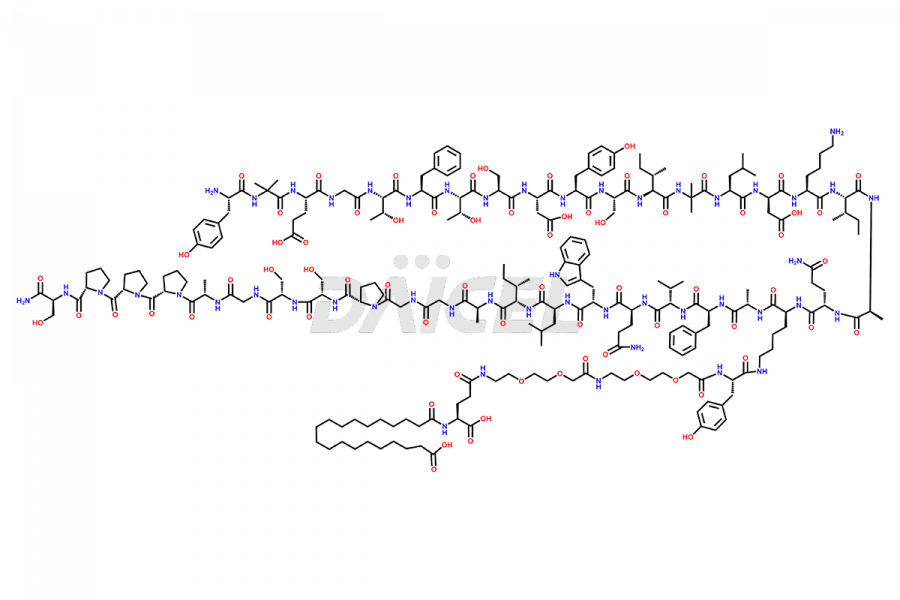
![Fragment [1-24]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2025/04/dcti-d-577-900x600.png)
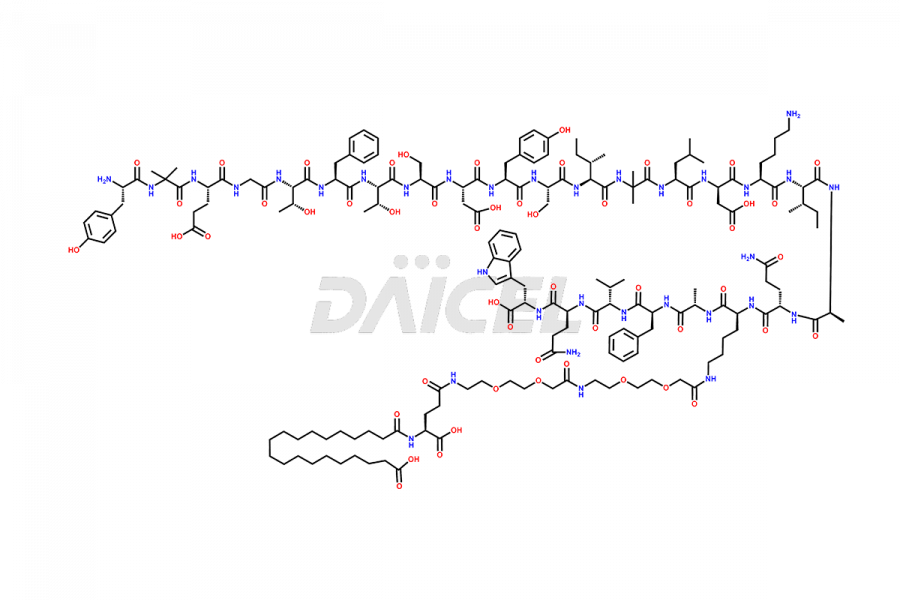
![Fragment [1-26]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2025/04/dcti-d-573-900x600.png)
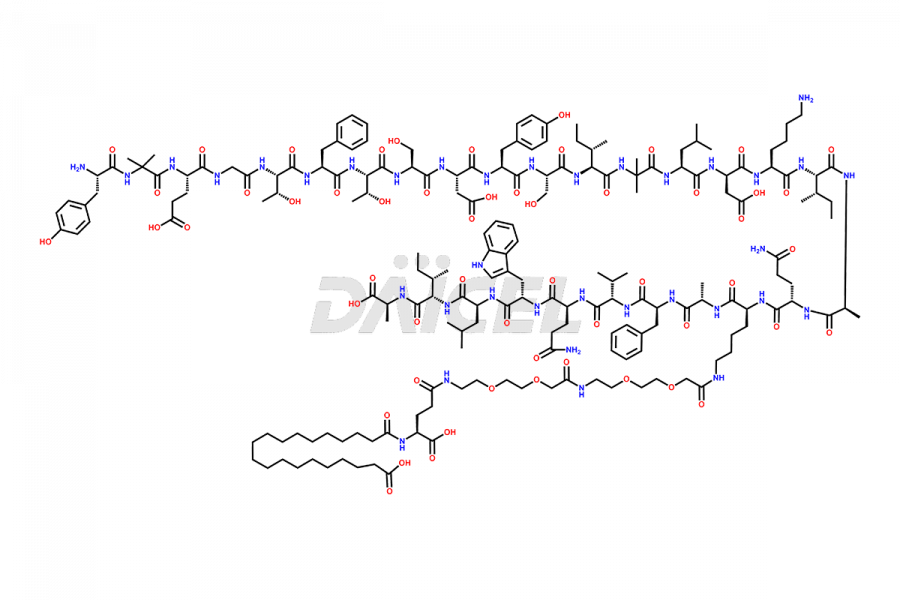
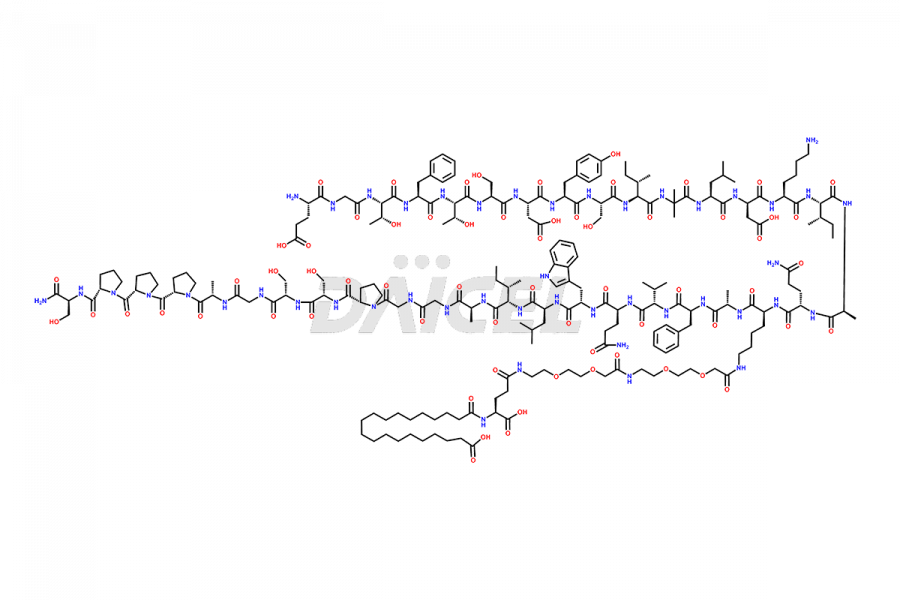
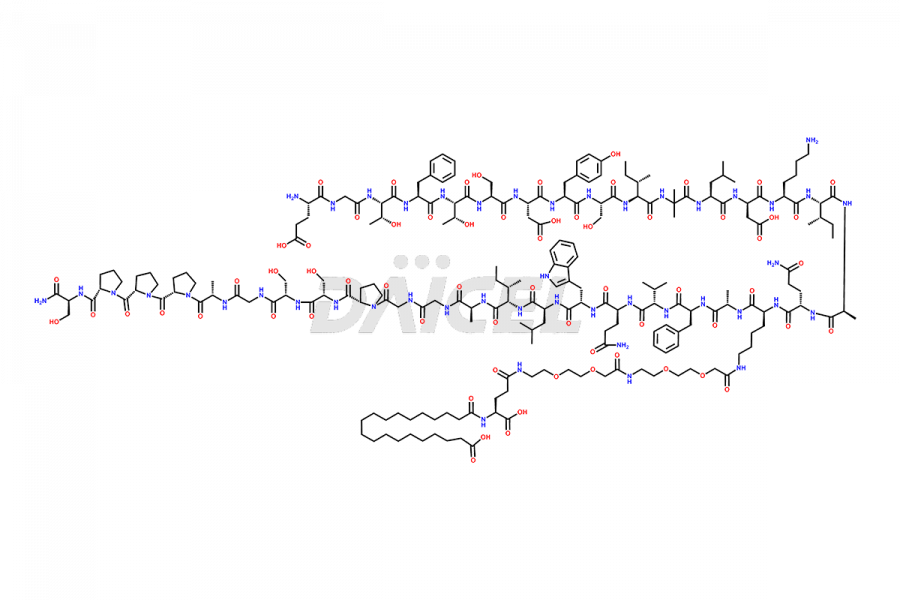
![Fragment [5-39]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2025/04/dcti-d-562-900x600.png)
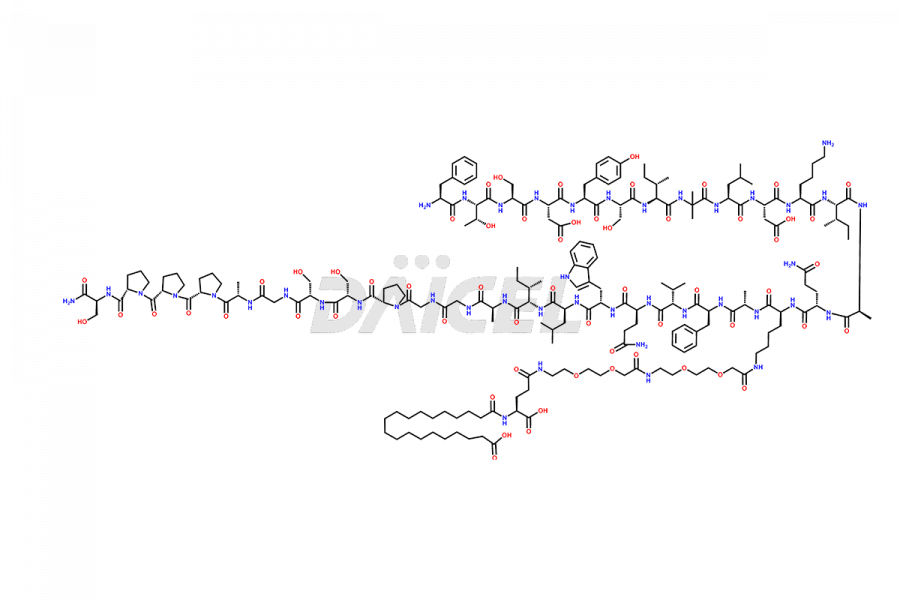
![Fragment [7-39]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2025/04/dcti-d-574-900x600.png)
![Fragment [8-39]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2025/04/dcti-d-575-900x600.png)
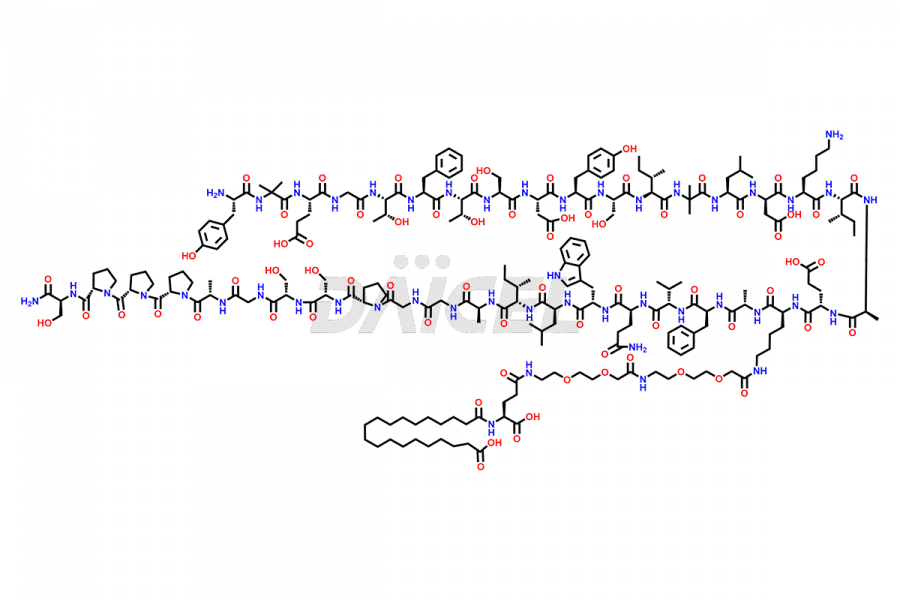
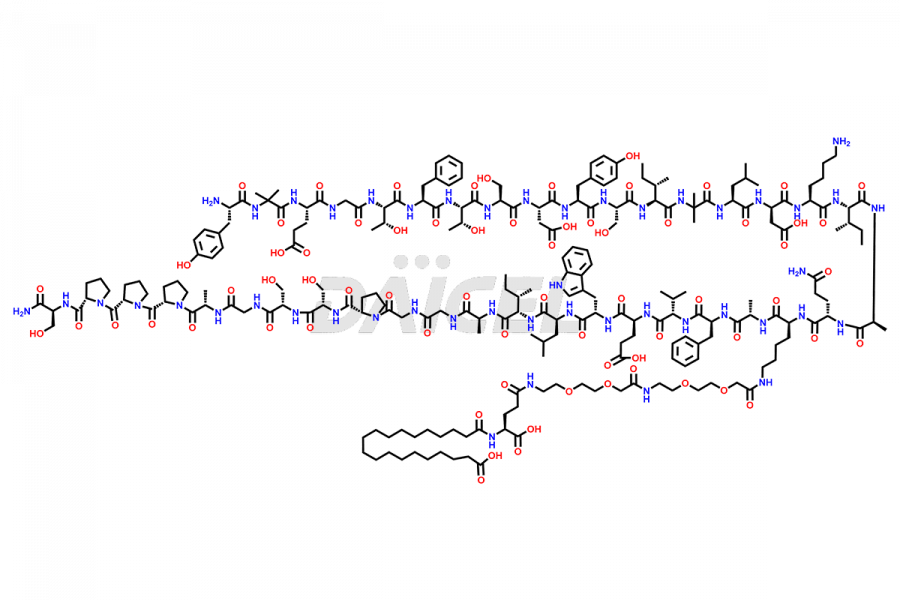
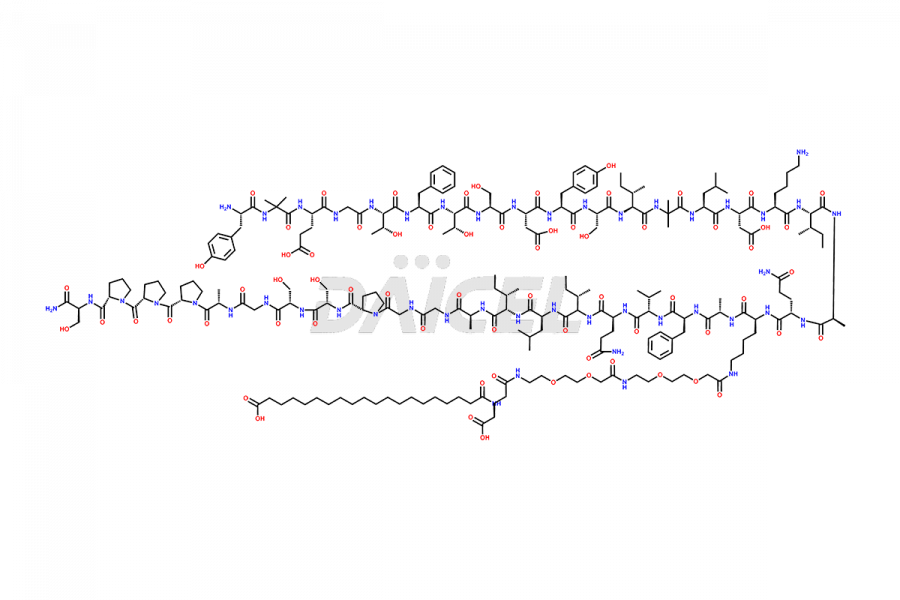
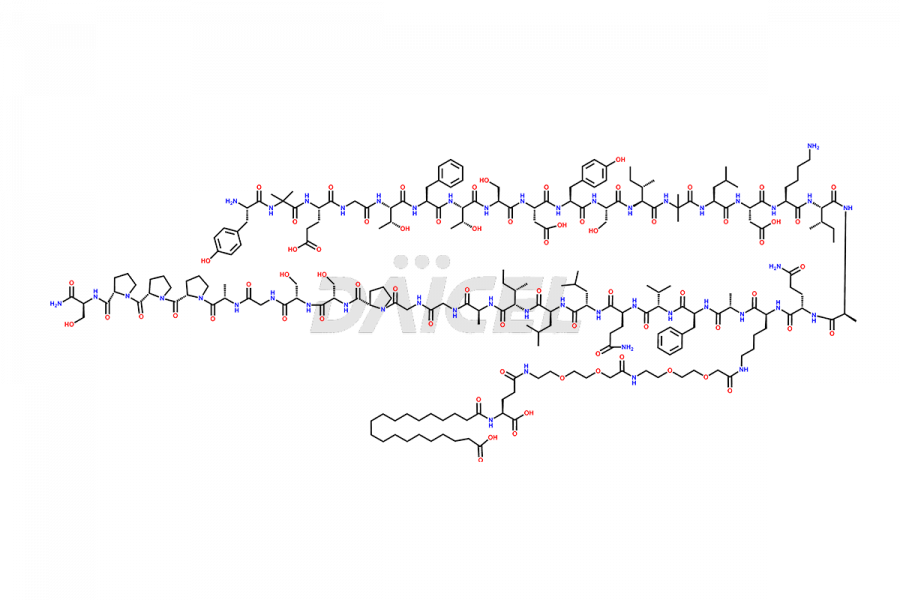
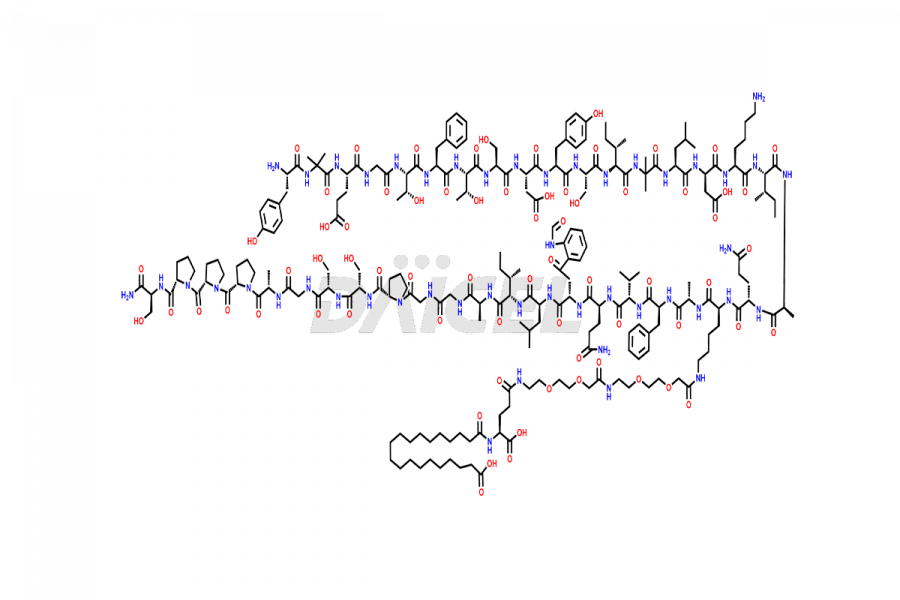
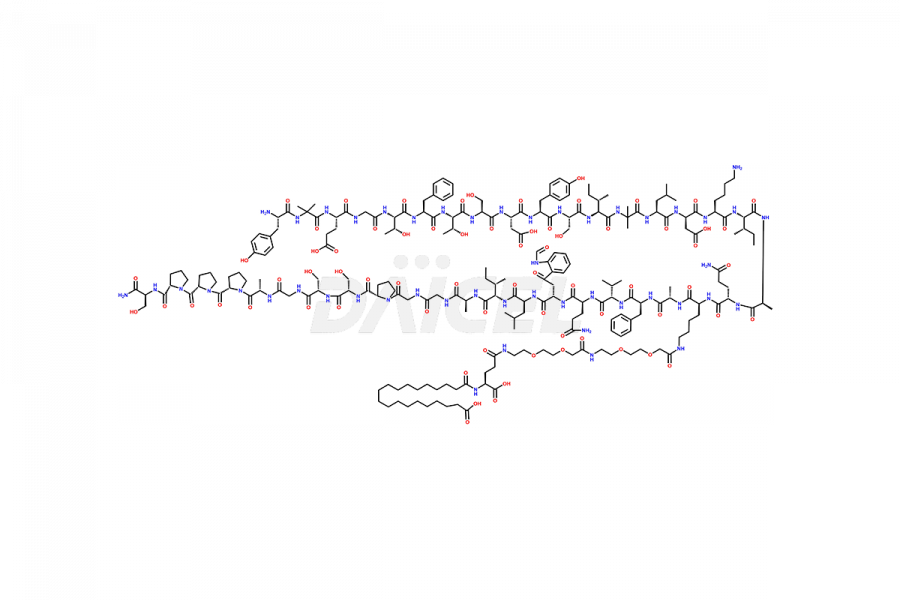
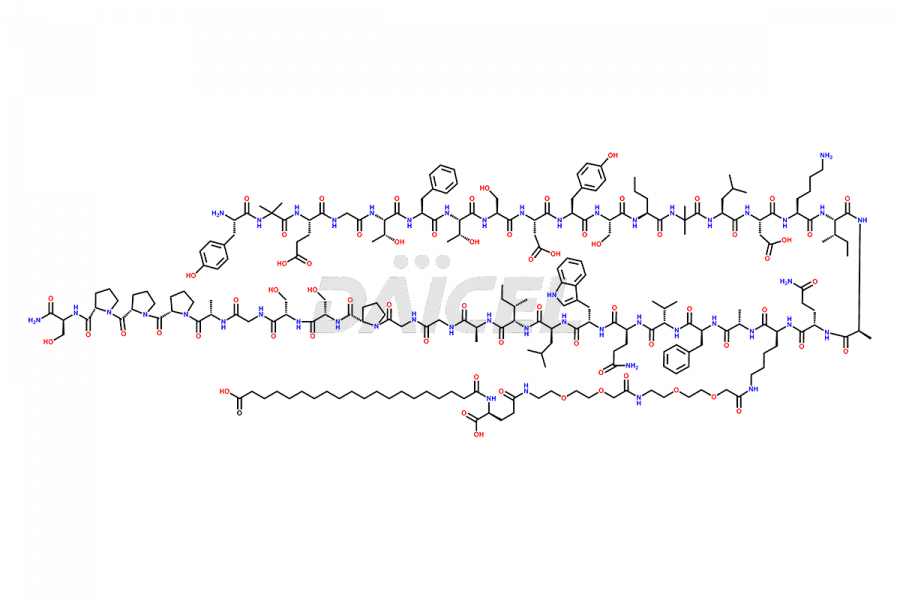
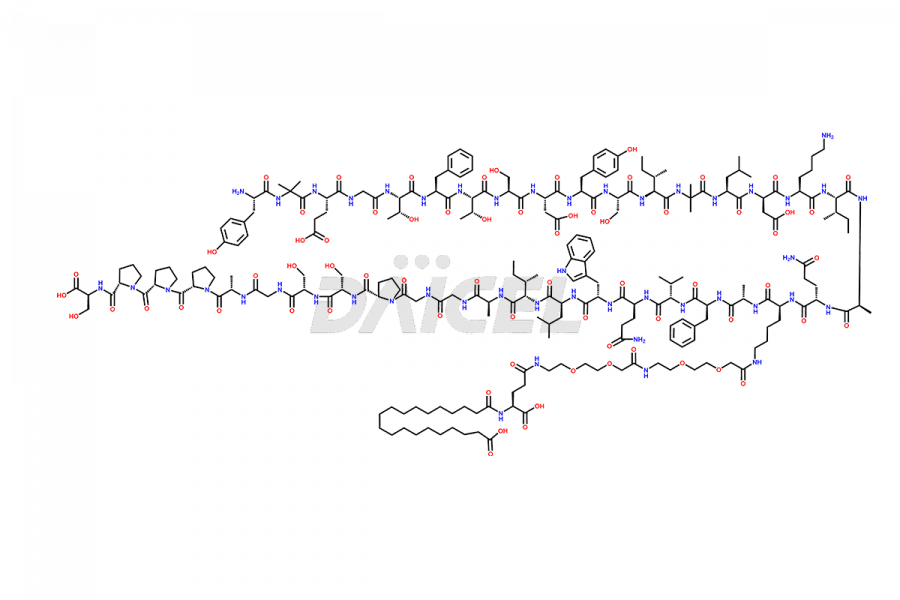
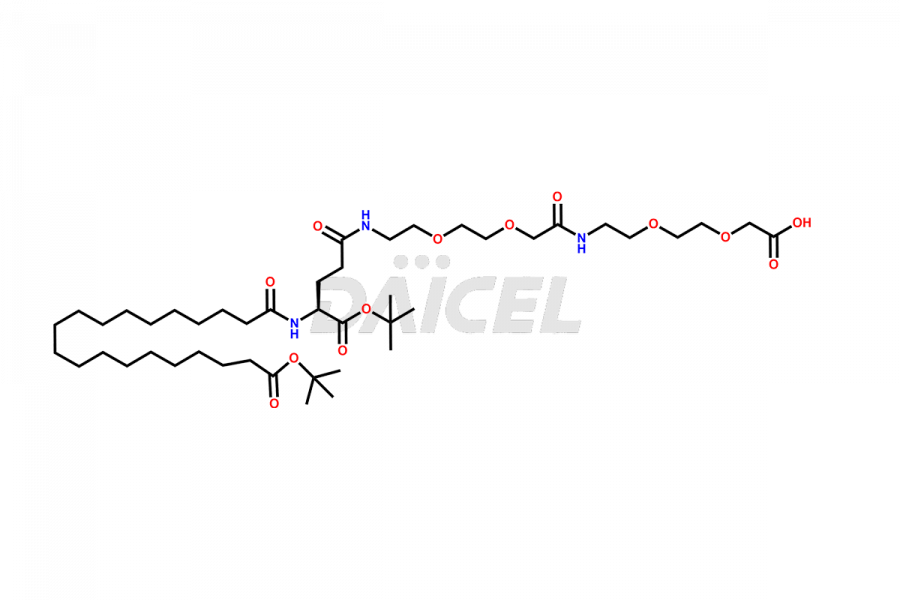
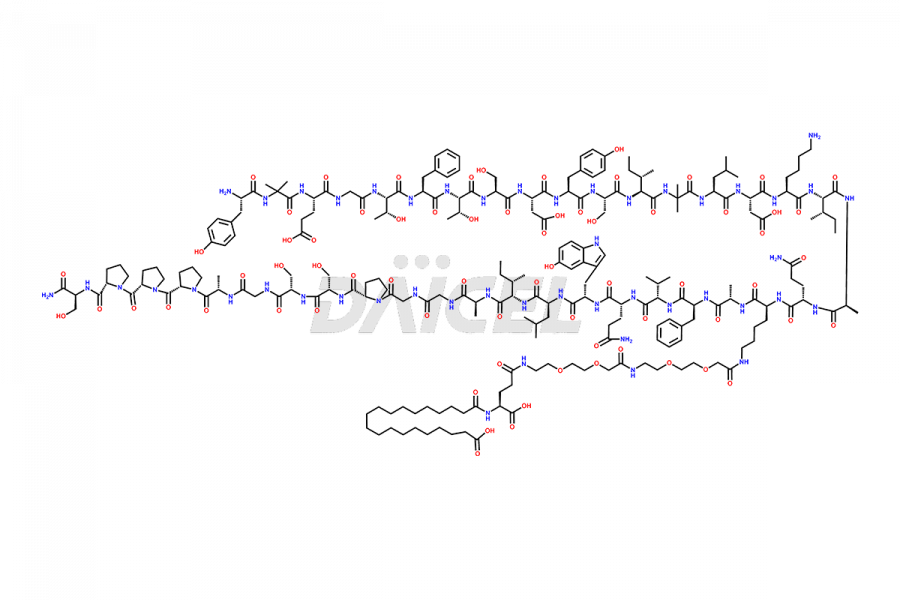
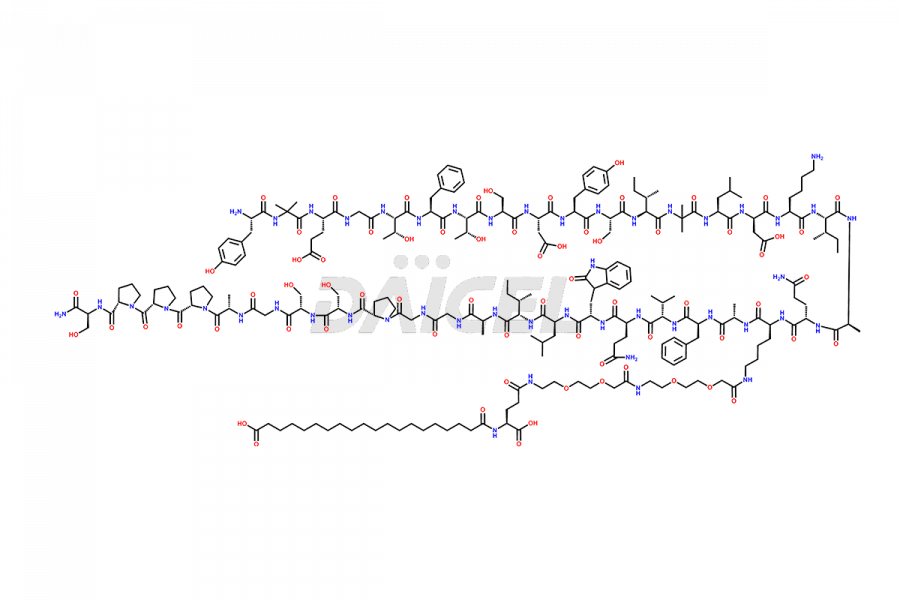
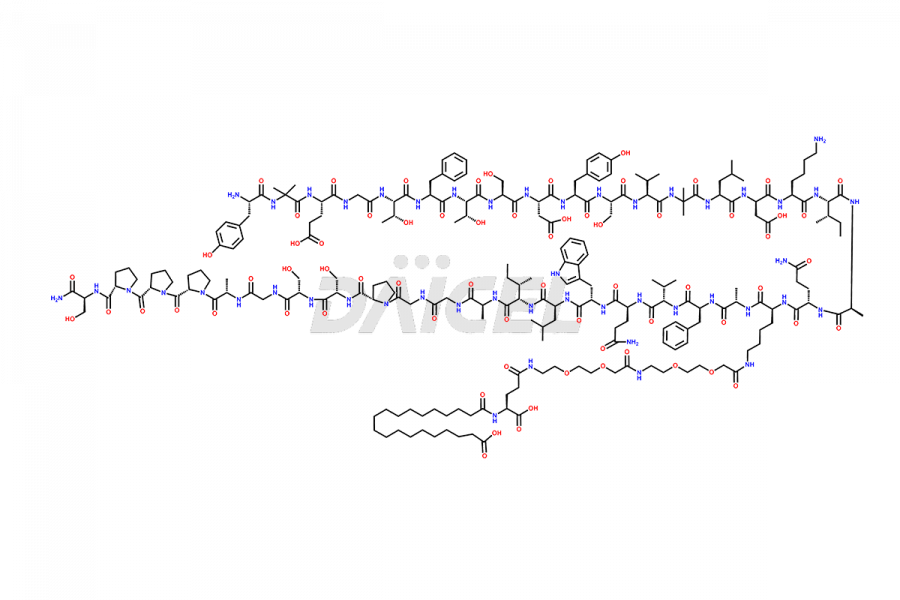
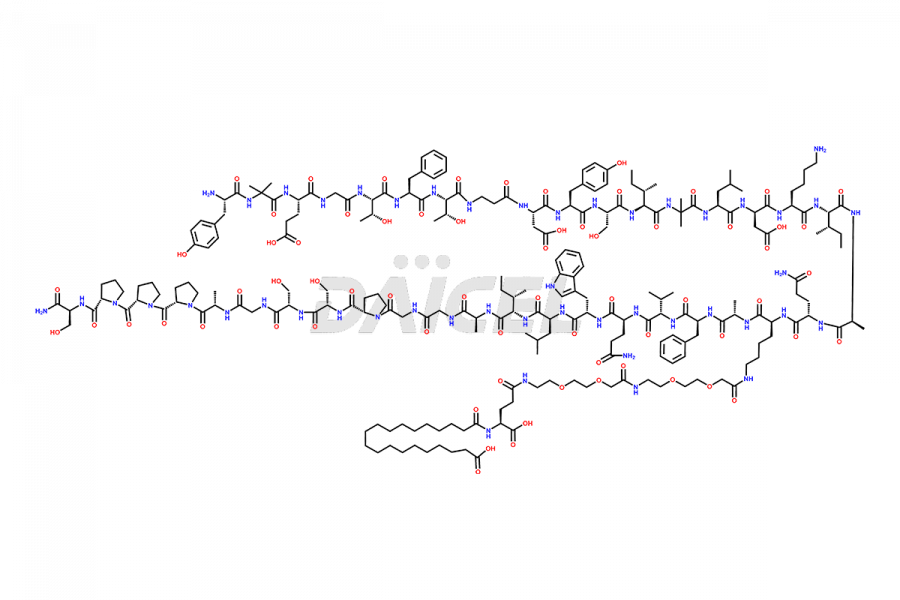
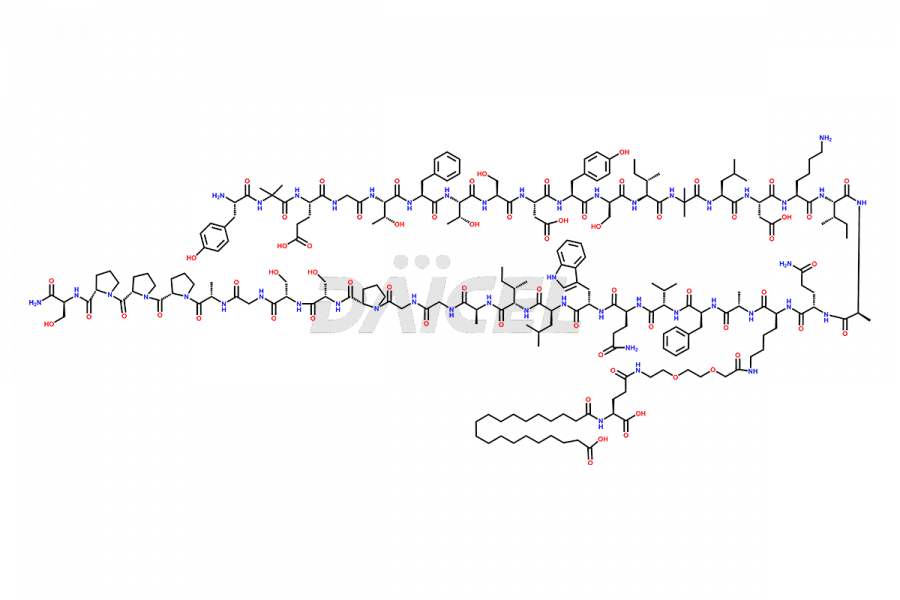
![?-Asp-9]-Tirzepatide](https://www.daicelpharmastandards.com/wp-content/uploads/2024/03/dcti-d-333-900x600.png)