From Chemistry to Consequence: What the Industry Must Now Solve
The oligonucleotide drug market is no longer a niche. Over 20 products are approved globally as of early 2026, with a pipeline that spans hundreds of candidates across phases. The first generic oligonucleotide applications have been filed. Regulatory agencies have issued new — and more specific — guidance on impurity characterisation and control. Contract manufacturing organisations, analytical service providers, and reference standard suppliers are all being asked to develop capabilities that did not exist as commercial offerings five years ago.
This blog examines what those industry demands look like in practice: which analytical methods are now standard or emerging, what the 2026 regulatory environment expects from manufacturers, and where Daicel — through both its Analytical Services division and its Pharma Standards business — fits into this increasingly complex picture.
The Industry Pressure Points: Where Quality Science Gets Tested
Three structural forces are shaping how the oligonucleotide industry approaches impurity analysis right now, and each creates a distinct kind of demand on analytical infrastructure.
Scale-Up and Manufacturing Consistency
The majority of approved oligonucleotide drugs were developed and initially manufactured at relatively small scale, with analytical characterisation carried out by specialised academic or early-phase contract labs. As products mature commercially and demand grows, manufacturing must scale — and scale introduces new process variables. Step efficiency, column loading on preparative chromatography, reagent lot-to-lot variability, and lyophilisation parameters can all shift the impurity profile in ways that require systematic re-characterisation. Regulatory agencies expect manufacturers to demonstrate that the impurity profile is consistent across manufacturing scales and that any changes are evaluated for their impact on drug substance quality.
Generic Oligonucleotides: The Sameness Problem
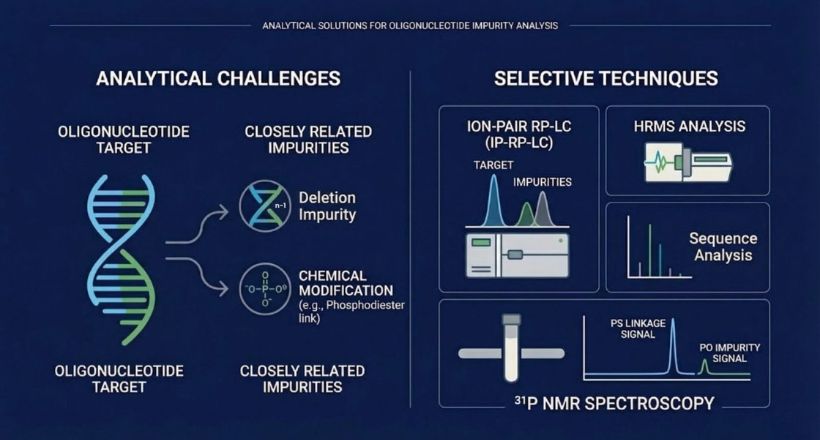
The loss of market exclusivity for nusinersen (Spinraza) has opened the first serious chapter of oligonucleotide generics development. Demonstrating that a generic oligonucleotide is the same as its reference-listed drug is not a straightforward bioequivalence exercise. For a fully phosphorothioated 20-mer ASO with 19 PS linkages, the theoretical diastereomeric space contains more than 500,000 stereoisomeric species. Demonstrating that a generic product has the same diastereomeric composition as the reference product — across the entire distribution, not just at a summary level — requires analytical capabilities that push current technology to its practical limits.
The FDA’s Office of Generic Drugs has been explicit about this expectation. Its FY2025 generic drug science initiative flagged diastereomeric characterisation as a key unresolved challenge and endorsed a multi-method approach — multiple LC strategies, MS/MS fragmentation sequencing, and ³¹P NMR — as the recommended framework for sameness assessment. This is now the expected standard for generic oligonucleotide submissions, and it is driving rapid investment in the relevant analytical capabilities across the industry.
Stereopure Oligonucleotides: A New Quality Paradigm
Simultaneously, the frontiers of innovator oligonucleotide development are moving toward stereopure synthesis — where each PS linkage is synthesised with a defined, single Rp or Sp configuration, using chiral auxiliary or stereoselective coupling chemistry. Ionis Pharmaceuticals pioneered this approach, and clinical data have demonstrated that specific diastereomeric configurations confer superior potency or tolerability depending on sequence context and target tissue.
For stereopure drugs, the analytical requirement is fundamentally different from characterising a diastereomeric mixture. The question is no longer ‘what is the distribution of stereoisomers?’ but ‘is the stereochemistry at each defined position correct?’ This is a higher-resolution chiral analysis challenge that requires methods capable of confirming positional stereochemical fidelity — and it represents a growing area of demand for specialised analytical services.
The Regulatory Landscape in 2026
EMA Draft Guideline on Oligonucleotide Development and Manufacture
The EMA issued its first guideline dedicated specifically to oligonucleotide drug development and manufacture in July 2024. The consultation period closed in January 2025; finalisation is expected during 2026. The guideline establishes that impurities above 1.0% must be identified and those above 1.5% must be qualified through appropriate safety data. It requires multiple orthogonal purity methods — sole reliance on a single technique is explicitly insufficient. For PS-containing oligonucleotides, diastereomeric characterisation is mandatory. The guideline also clarifies that oligonucleotides fall outside the scope of ICH Q3A, meaning manufacturers cannot apply small-molecule impurity frameworks and must develop oligonucleotide-specific qualification strategies.
FDA Nonclinical Safety Guidance and Generic Drug Expectations
FDA CDER’s 2024 draft guidance classifies oligonucleotide impurities as sequence-related species (for which manufacturing process control is the primary management tool) and process-related impurities (governed by ICH Q3C, Q3D, and applicable environmental standards). The FDA’s Office of Generic Drugs has simultaneously elevated its expectations for generic oligonucleotide applications — particularly around diastereomeric sameness — requiring a multi-method analytical package that reflects the current state of the art.
| Guideline / Framework | Key Threshold / Scope | Impurity-Specific Requirement |
|---|---|---|
| EMA Draft Guideline (2024 → final 2026) | Identify ≥1.0%; Qualify ≥1.5% | Orthogonal methods required; PS diastereomers must be characterised |
| FDA Nonclinical Safety Guidance (Draft 2024) | Sequence-related vs. PRI classified separately | Process control primary; ICH Q3C/Q3D govern PRIs |
| FDA OGD — Generic Oligonucleotides (FY2025) | Diastereomeric sameness required for PS-ONs | Multi-method LC + MS/MS + NMR strategy mandated |
| ICH Q3D — Elemental Impurities | PDE limits for injectables | ICP-MS required for metal catalysts |
| ICH Q3C — Residual Solvents | Class 1/2/3 limits | Applies to ACN, DCM, pyridine from SPPS |
Where Daicel Fits: Analytical Services and Pharma Standards
Daicel Chiral Technologies India expertise in the oligonucleotide space maps onto two concrete commercial capabilities: Daicel Analytical Services, which addresses the method development and characterisation needs of drug developers, and Daicel Pharma Standards, which addresses the equally critical — and chronically underserved — demand for well-characterised and certified oligonucleotide impurity standards.
The Road Ahead
Several near-term developments will shape the oligonucleotide impurity landscape through 2026 and beyond. Finalisation of the EMA guideline will bring greater regulatory certainty but also higher analytical expectations — particularly on orthogonal method validation and stability-indicating method qualification. The first generic oligonucleotide approvals will establish precedents for diastereomeric sameness assessment that the entire industry will need to follow. And the continued expansion of stereopure oligonucleotide clinical programmes will pull demand for high-resolution chiral characterisation out of academic and early-phase settings and into commercial QC.
Platform-based impurity qualification strategies — where the structural similarity of oligonucleotide sequence variants to the parent molecule justifies streamlined nonclinical qualification — are gaining regulatory acceptance, reducing the burden of individualised safety studies while maintaining scientific rigour. The modality has earned its place in the therapeutic mainstream. The analytical science needed to support it is catching up fast — and the organisations that build genuine depth in oligonucleotide impurity characterisation, particularly in its stereochemical dimensions, are well-positioned for the decade ahead.

Write a comment
Your email address will not be published. All fields are required