Valacyclovir Impurities
General Information
Valacyclovir Impurities and Valacyclovir
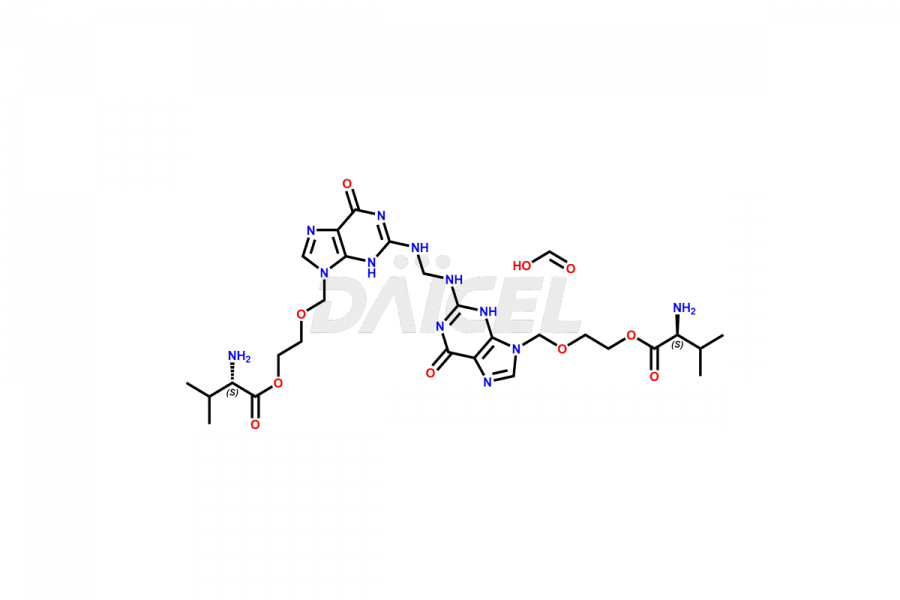
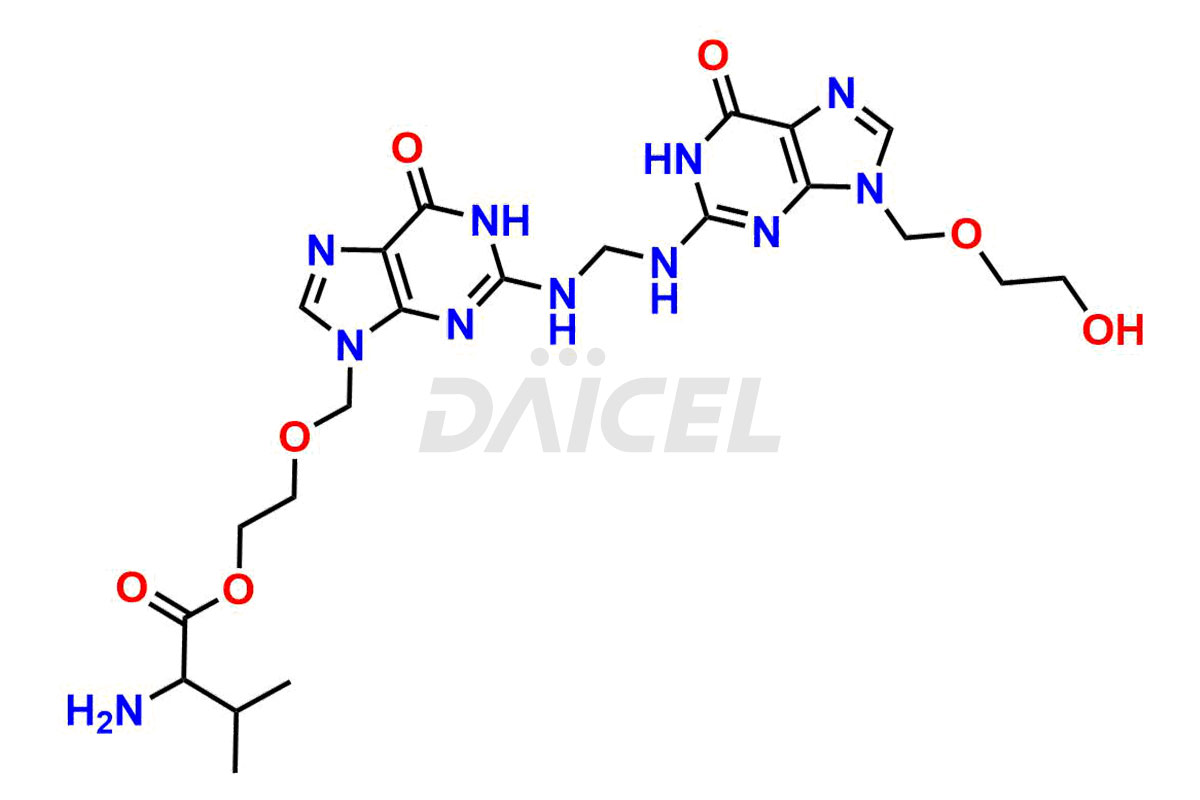
Daicel Pharma is a trusted provider of quality Valacyclovir impurity standards, including Bis Valacyclovir and Bis Valacyclovir mono valine. These impurities are crucial in meticulously evaluating the quality, stability, and safety of the active pharmaceutical ingredient, Valacyclovir. Furthermore, Daicel Pharma specializes in the custom synthesis of Valacyclovir impurities, guaranteeing that individual client specifications are met with precision. With global shipping capabilities, their delivery to customers worldwide is possible.
Valacyclovir [CAS: 124832-26-4] is an antiviral medicine, a nucleoside analog DNA polymerase inhibitor. It acts as a prodrug of acyclovir and treats infections caused by the herpes simplex and varicella-zoster viruses.
Valacyclovir: Use and Commercial Availability
Valacyclovir is an ester prodrug, an L-valine ester of acyclovir, that treats herpes, varicella zoster, and cytomegaloviruses. It is also effective in suppressing recurrent episodes of genital herpes. Valacyclovir is available under the brand name Valtrex.
Valacyclovir Structure and Mechanism of Action
The chemical name of Valacyclovir is 2-[(2-Amino-6-oxo-3H-purin-9-yl)methoxy]ethyl (2S)-2-amino-3-methylbutanoate. Its chemical formula is C13H20N6O4, and its molecular weight is approximately 324.34 g/mol.
Valacyclovir converts to acyclovir, which inhibits viral DNA polymerase and incorporates and terminates the viral DNA chain. It inactivates viral DNA polymerase.
Valacyclovir Impurities and Synthesis
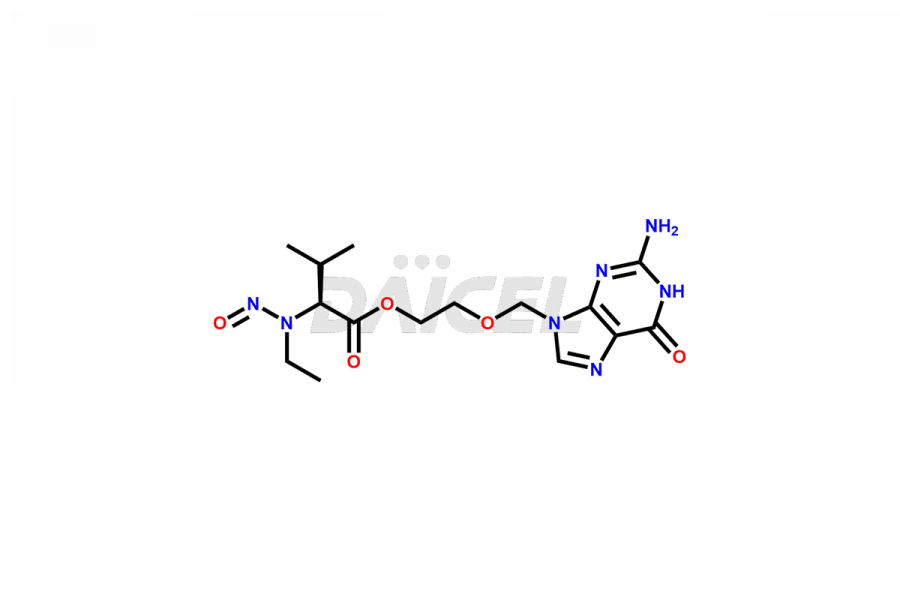
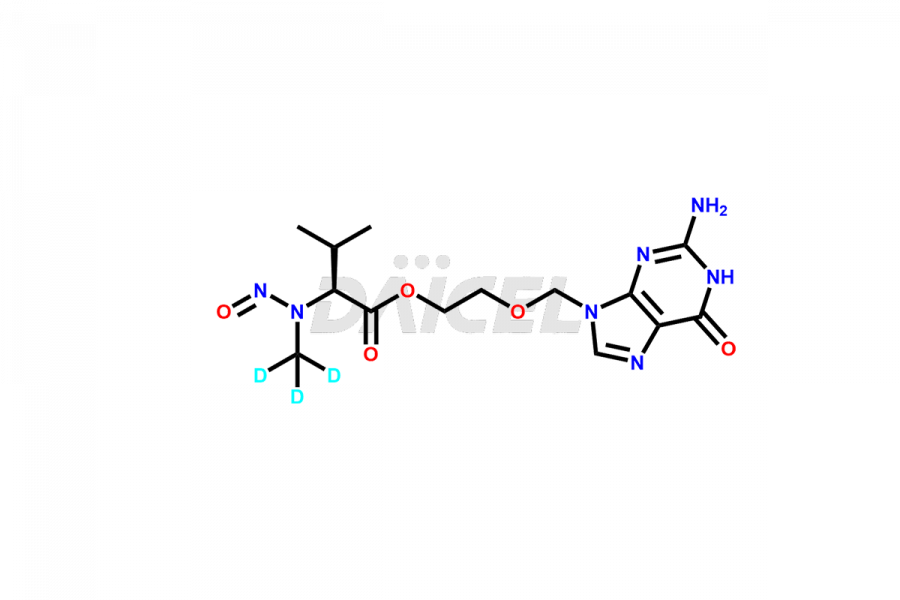
Valacyclovir impurities can arise during synthesis1 due to storage or using specific raw materials and intermediates in manufacturing. They encompass related compounds, degradation products, and process impurities. Stringent quality control measures and analytical methods are crucial to ensure the purity and safety of Valacyclovir for patient use.
Daicel provides a comprehensive Certificate of Analysis (CoA) for Valacyclovir impurity standards, such as Bis Valacyclovir and Bis Valacyclovir mono valine. The CoA includes detailed characterization data such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity2. Additionally, upon delivery, we give 13C-DEPT. Daicel possesses the technology and expertise to synthesize any unknown Valacyclovir impurity or degradation product.
References
FAQ's
References
- Krenitsky, Thomas Anthony; Beauchamp, Lilia Marie, Therapeutic nucleosides, Wellcome Foundation Ltd., United Kingdom, EP308065B1, January 4, 1995
- Pham-Huy, C.; Stathoulopoulou, F.; Sandouk, P.; Scherrmann, J.-M.; Palombo, S.; Girre, C., Rapid determination of valaciclovir and acyclovir in human biological fluids by high-performance liquid chromatography using isocratic elution, Journal of Chromatography B: Biomedical Sciences and Applications, Volume: 732, Issue: 1, Pages: 47-53, 1999
Frequently Asked Questions
Why is it vital to analyze Valacyclovir impurities?
Analysis of Valacyclovir impurities helps assess the purity profile and identify potentially harmful substances.
Which solvent helps in analyzing Valacyclovir impurities?
Dimethyl sulfoxide (DMSO) is a common solvent for analyzing many Valacyclovir impurities.
What analytical methods are used for Valacyclovir impurities?
High-performance liquid chromatography (HPLC) is employed to separate and analyze the impurities of Valacyclovir.
How should Valacyclovir impurities be stored in terms of temperature?
The recommendation is to store Valacyclovir impurities at room temperature, within 2-8 °C.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.