(1R,2R)-2-(3,4-difluorophenyl)-N-hydroxycyclopropane-1-carboxamide
- CAT Number DCTI-C-004182
- CAS NUMBER 1353964-60-9
- MOLECULAR FORMULA C10H9F2NO2
- MOLECULAR WEIGHT 213.18
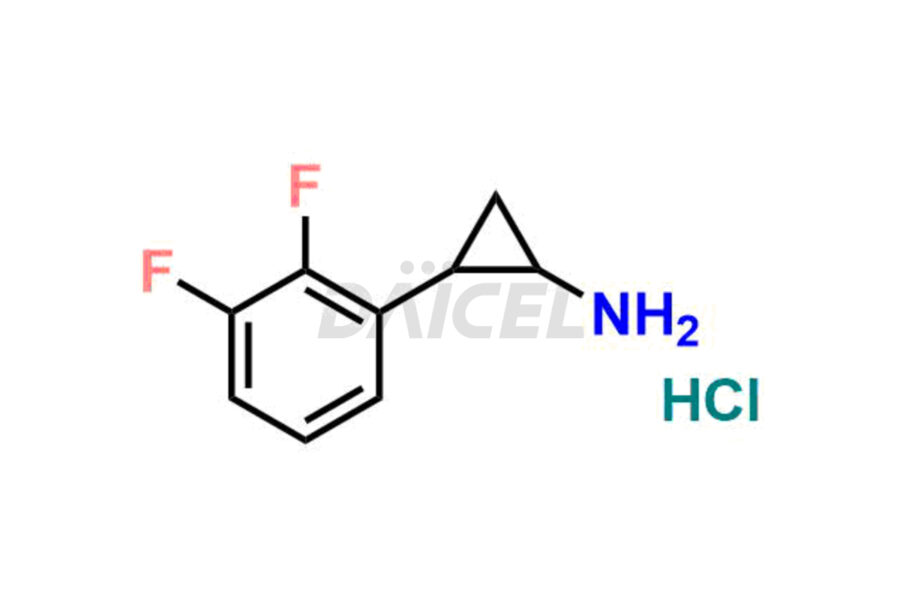
(1R,2R)-2-(3,4-difluorophenyl)cyclopropane-1-amine Hydrochloride
- CAT Number DCTI-C-003395
- CAS NUMBER 1414348-35-8 (Free base)
- MOLECULAR FORMULA C9H9F2N (Free base) C9H10ClF2N (HCl Salt)
- MOLECULAR WEIGHT 169.17 (Free base) 205.63 (HCl Salt)
(1R,2R)-2-(3,4-difluorophenyl)cyclopropane-1-carboxamide
- CAT Number DCTI-C-004178
- CAS NUMBER 1006376-62-0
- MOLECULAR FORMULA C10H9F2NO
- MOLECULAR WEIGHT 197.18
(1R,2S)-2-(2,3-difluorophenyl)cyclopropan-1-amine hydrochloride-Rel
- CAT Number DCTI-C-004148
- CAS NUMBER 1807939-83-8
- MOLECULAR FORMULA C9H9F2N (Free base); C9H10ClF2N (HCl Salt)
- MOLECULAR WEIGHT 169.17 (Free base); 205.63 (HCl salt)
(1R,2S)-2-(3-fluoro-4-methoxyphenyl)cyclopropan-1-amine hydrochloride
- CAT Number DCTI-C-003892
- CAS NUMBER NA
- MOLECULAR FORMULA C10H12FNO (Free base); C10H13ClFNO (HCl Salt)
- MOLECULAR WEIGHT 181.21 (Free base); 217.67 (HCl Salt)
(1R,2S)-2-(3,4-difluorophenyl)cyclopropan-1-amine ...
- CAT Number DCTI-C-004326
- CAS Number 1402222-66-5
- Molecular Formula C9H9F2N (Free base) C9H10ClF2N (HCl Salt)
- Molecular Weight 169.17(Free base) 205.63 (HCl Salt)
(1R,2S)-2-(4-Fluorophenyl)cyclopropane-1-amine hydrochloride
- CAT Number DCTI-C-004169
- CAS NUMBER 1314324-00-9
- MOLECULAR FORMULA C9H10FN (Free base) C9H11ClFN (HCl Salt)
- MOLECULAR WEIGHT 151.18(Free base) 187.64 (HCl Salt)
(1S,2R)-2-(3,4-difluorophenyl)cyclopropan-1-amine ...
- CAT Number DCTI-C-004327
- CAS Number NA
- Molecular Formula C9H9F2N (Free base) C9H10ClF2N (HCl Salt)
- Molecular Weight 169.17(Free base) 205.63 (HCl Salt)
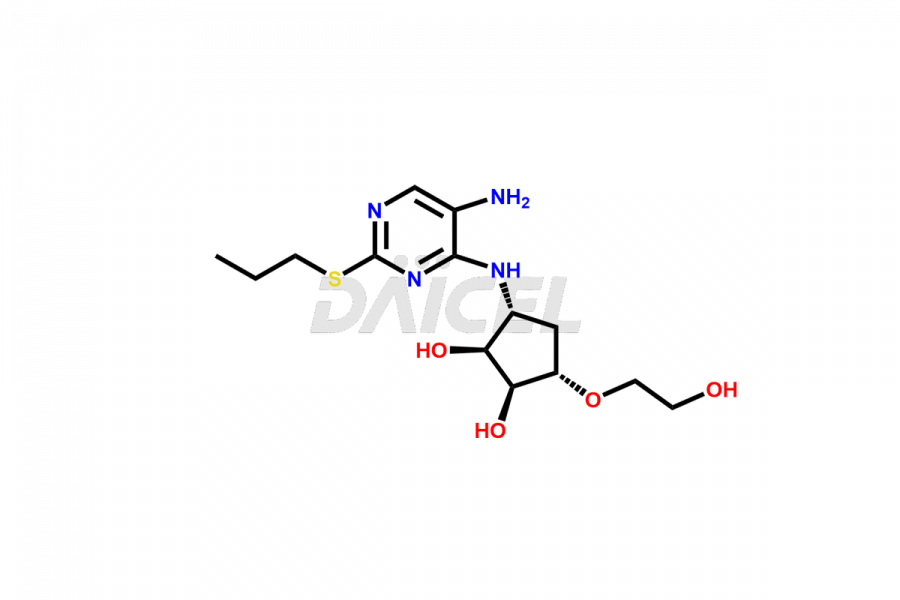
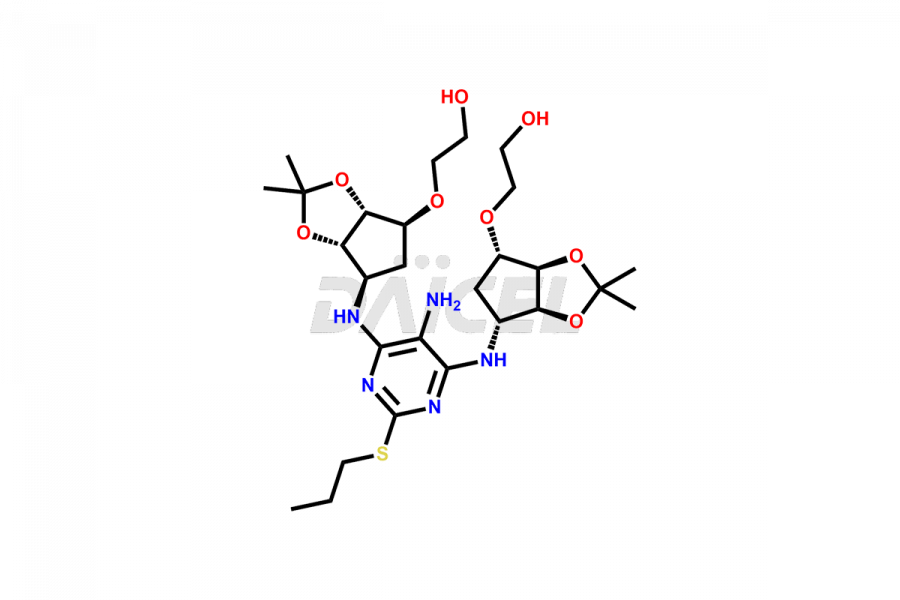
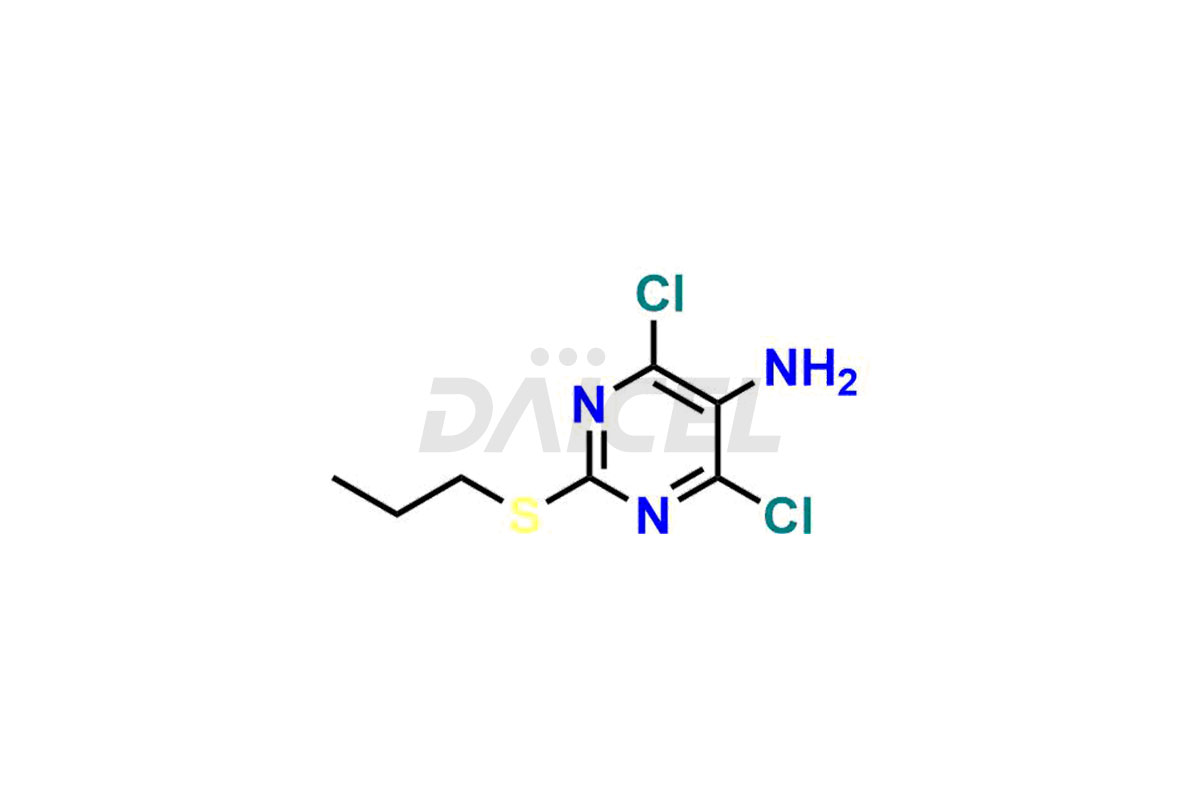
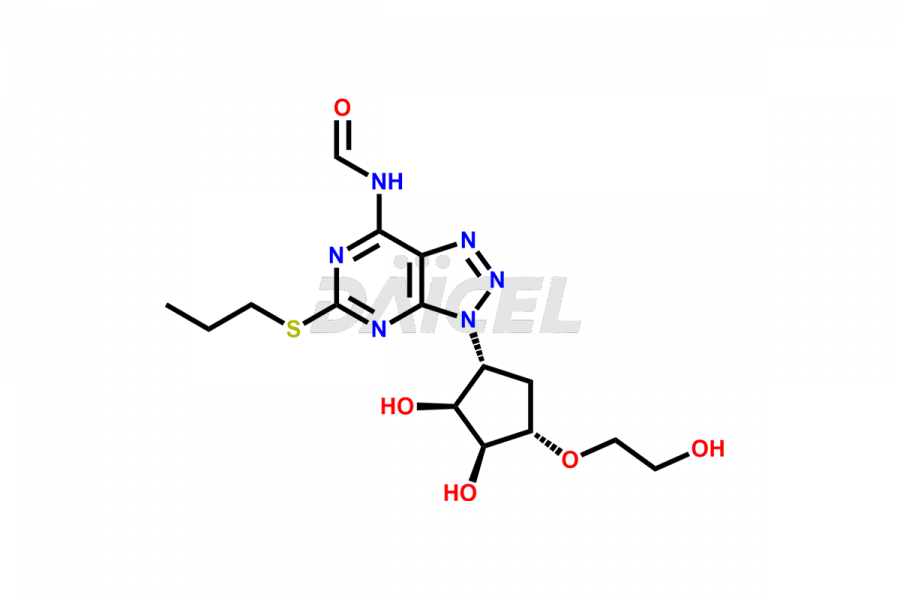
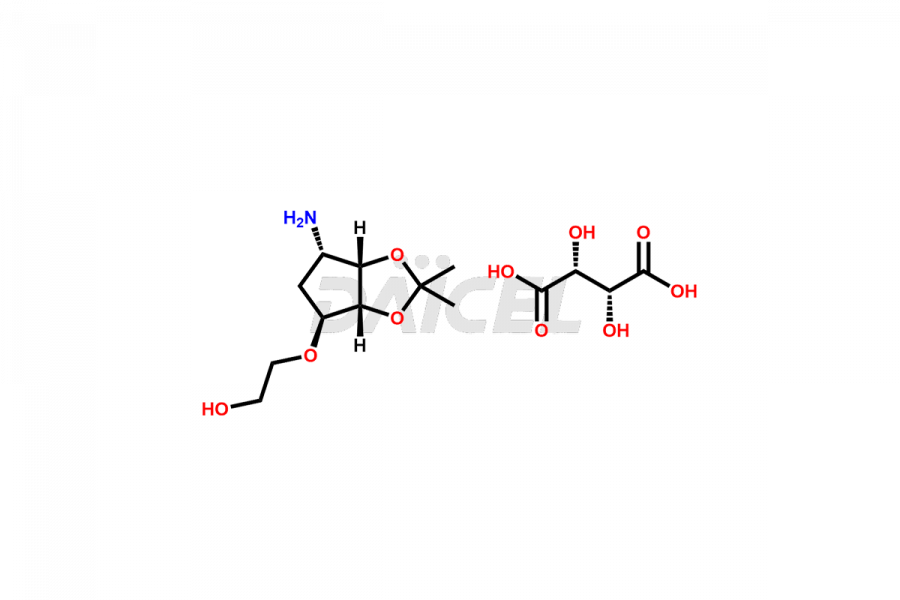
(1S,2S,3R,5S)-3-((5-amino-2-(propylthio)pyrimidin-...
- CAT Number DCTI-C-002662
- CAS NUMBER NA
- MOLECULAR FORMULA C14H24N4O4S
- MOLECULAR WEIGHT 344.43
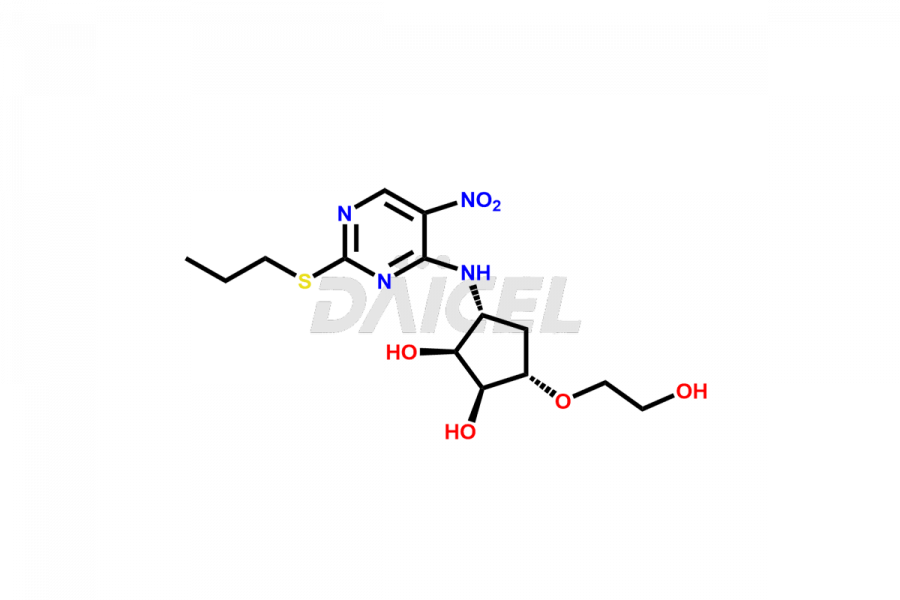
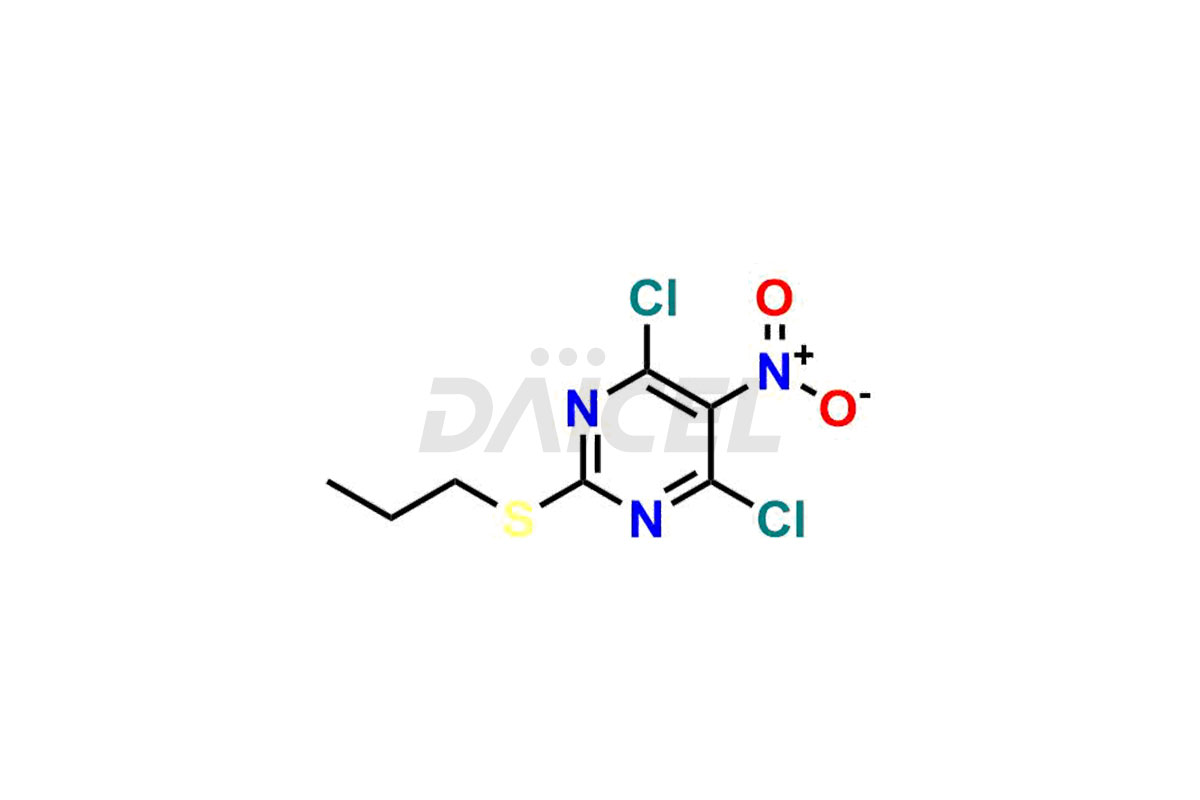
(1S,2S,3S,5R)-3-(2-hydroxyethoxy)-5-((5-nitro-2-(p...
- CAT Number DCTI-C-002663
- CAS NUMBER NA
- MOLECULAR FORMULA C14H22N4O6S
- MOLECULAR WEIGHT 374.41
(1S,2S)-2-(3,4-difluorophenyl)cyclopropan-1-amine Hydrochloride
- CAT Number DCTI-C-003394
- CAS NUMBER 1459719-81-3 (Free base)
- MOLECULAR FORMULA C9H9F2N (Free base) C9H10ClF2N (HCl Salt)
- MOLECULAR WEIGHT 169.17 (Free base) 205.63 (HCl Salt)
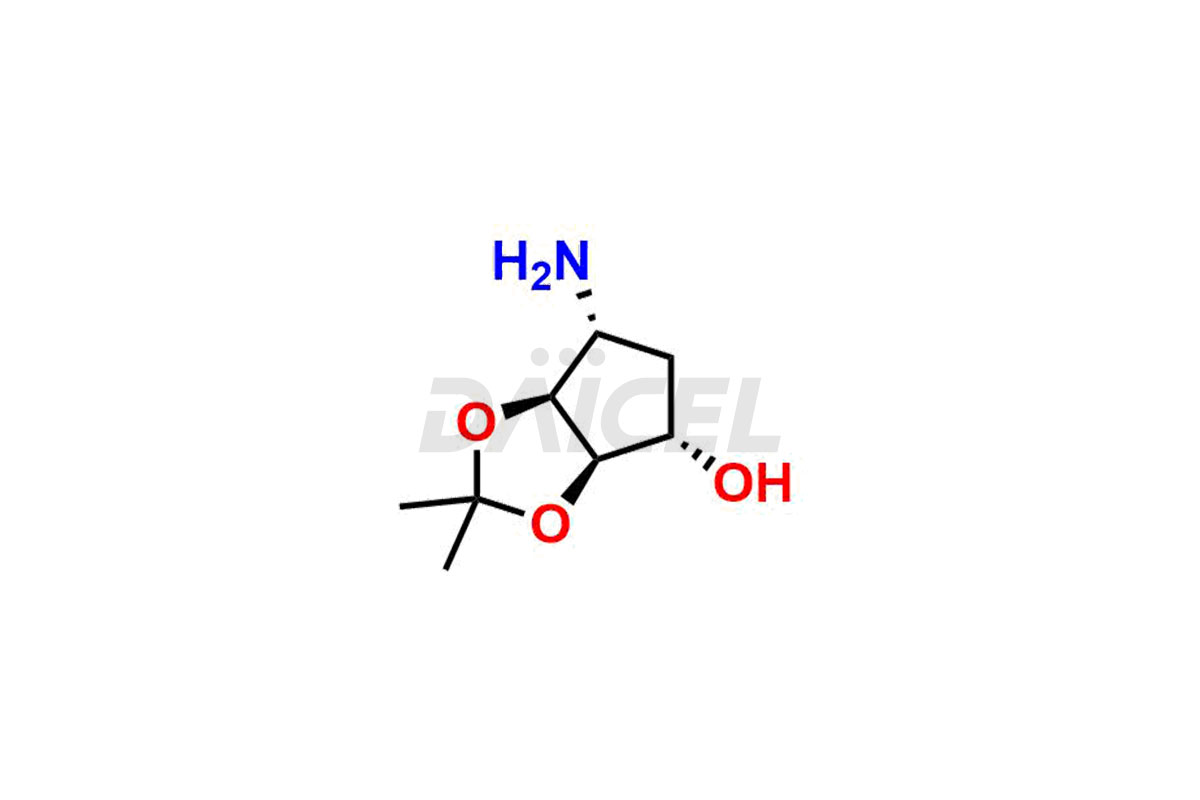
(3aR,4R,6R,6aS)-6-amino-2,2-dimethyltetrahydro-4H-...
- CAT Number DCTI-C-003437
- CAS NUMBER 151527-83-2
- MOLECULAR FORMULA C8H15NO3
- MOLECULAR WEIGHT 173.21
(3aR,4R,6S,6aS)-6-amino-2,2-dimethyltetrahydro-4H-...
- CAT Number DCTI-C-003474
- CAS NUMBER 2715109-35-4
- MOLECULAR FORMULA C8H15NO3
- MOLECULAR WEIGHT 173.21
(3aR,4S,6R,6aS)-6-amino-2,2-dimethyltetrahydro-3aH...
- CAT Number DCTI-C-004209
- CAS NUMBER 155899-66-4
- MOLECULAR FORMULA C8H15NO3
- MOLECULAR WEIGHT 173.21
(3aR,4S,6S,6aS)-6-amino-2,2-dimethyltetrahydro-4H-...
- CAT Number DCTI-C-003460
- CAS NUMBER 2376278-80-5
- MOLECULAR FORMULA C8H15NO3
- MOLECULAR WEIGHT 173.21
(Z)-N-benzyl-1-((4S,5R)-2,2-dimethyl-5-vinyl-1,3-d...
- CAT Number DCTI-C-003504
- CAS NUMBER 345898-95-5
- MOLECULAR FORMULA C15H19NO3
- MOLECULAR WEIGHT 261.32
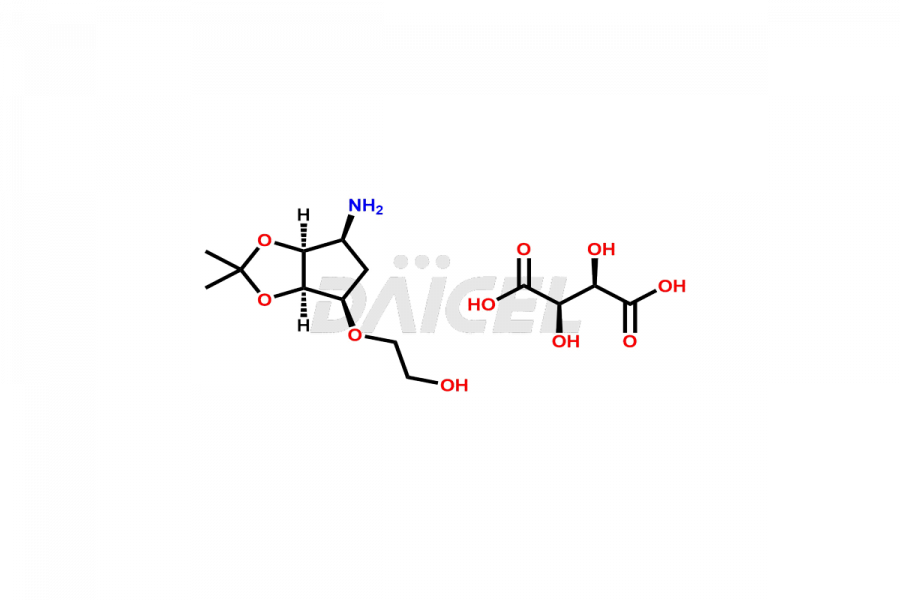
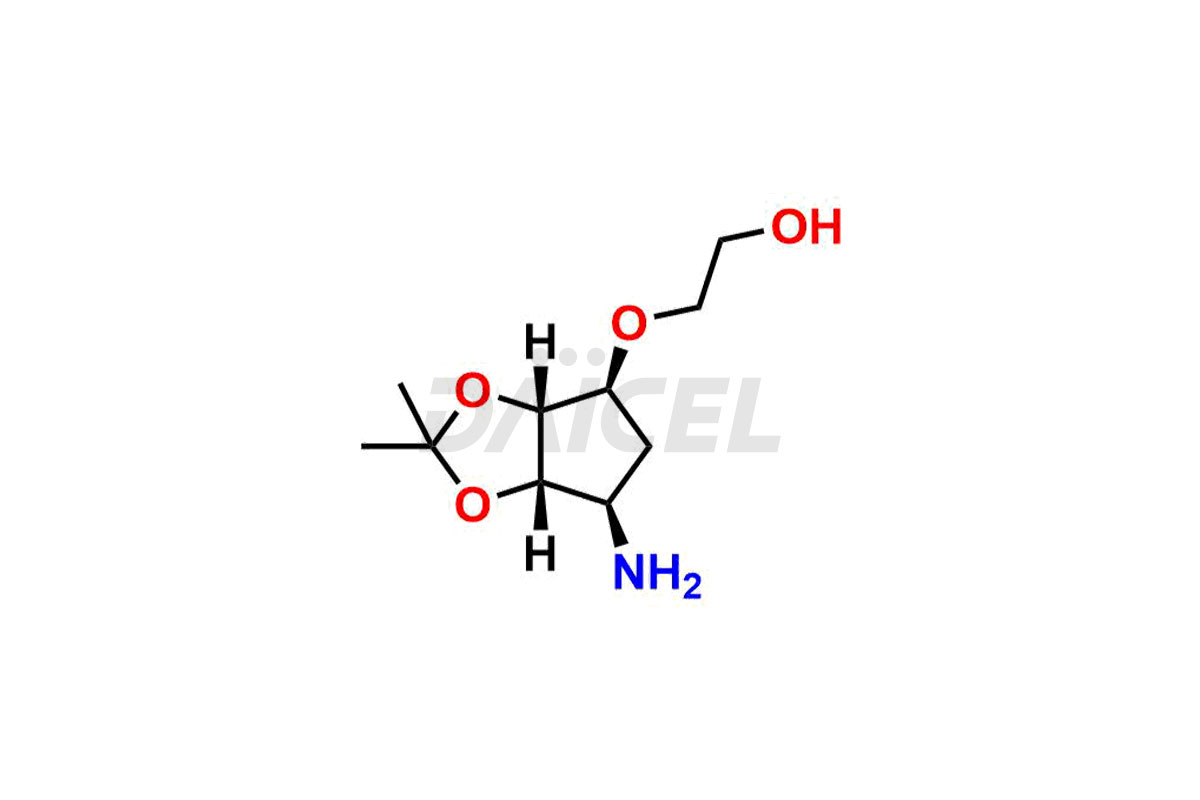
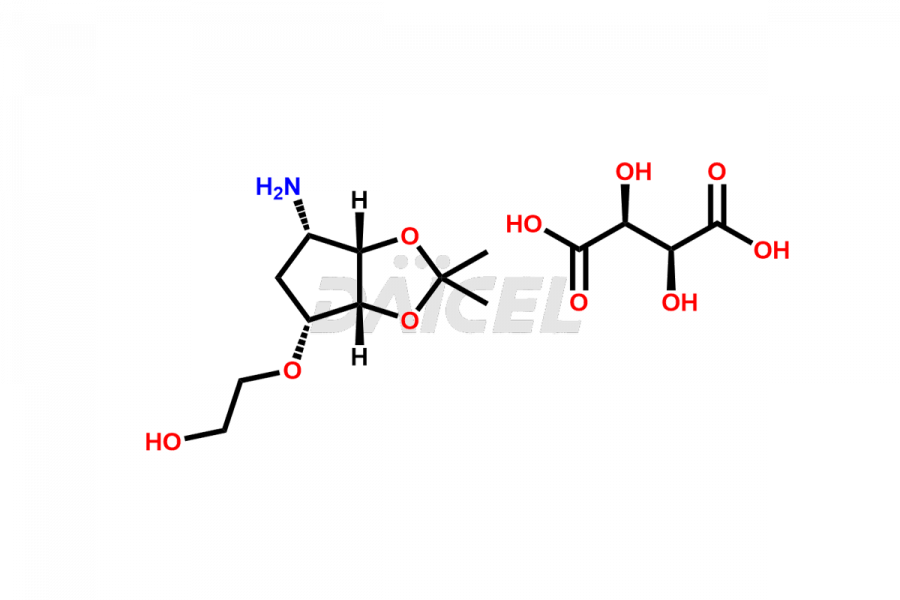
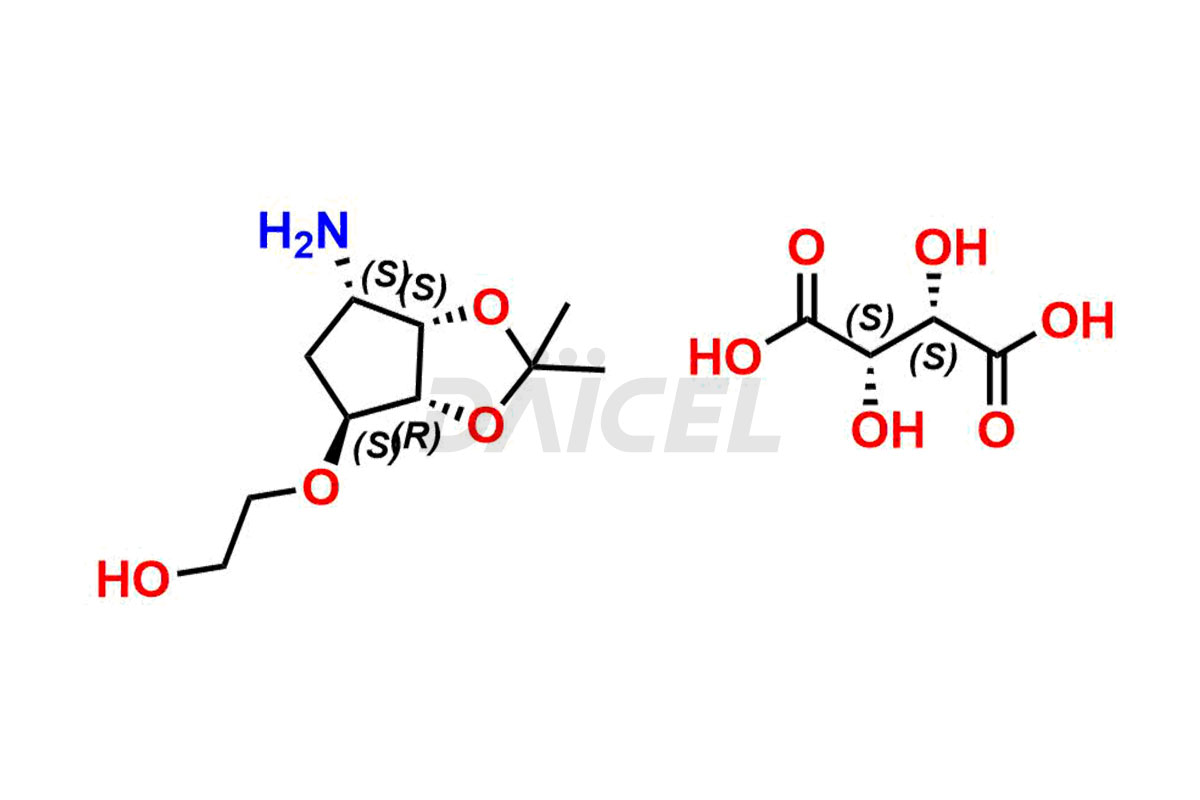
2-(((3aR,4R,6S,6aS)-6-amino-2,2-dimethyltetrahydro...
- CAT Number DCTI-C-005103
- CAS Number 2376278-71-4
- Molecular Formula C14H25NO10 (Tartrate salt); C10H19NO4 (Free Base)
- Molecular Weight 367.35 (Tartrate salt); 217.27 (Free Base)
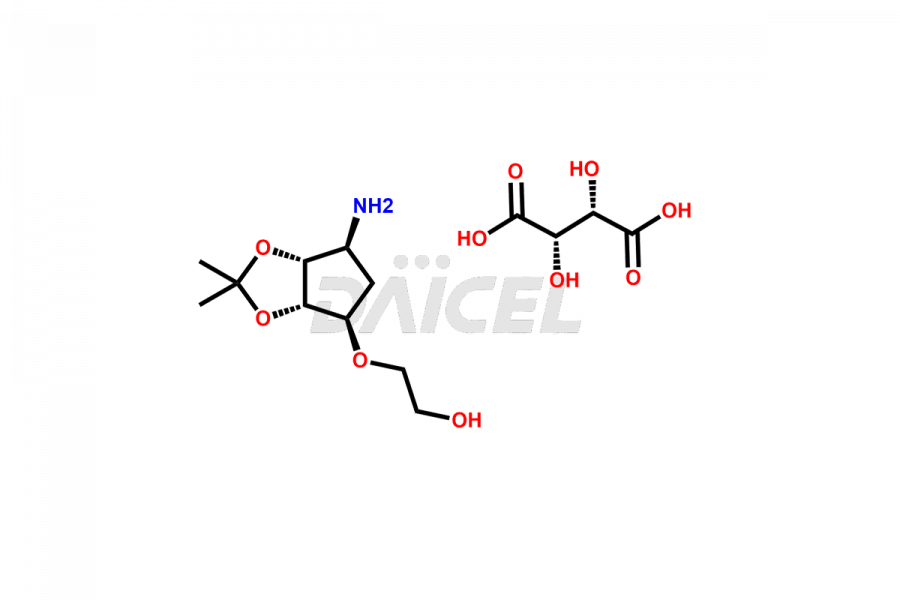
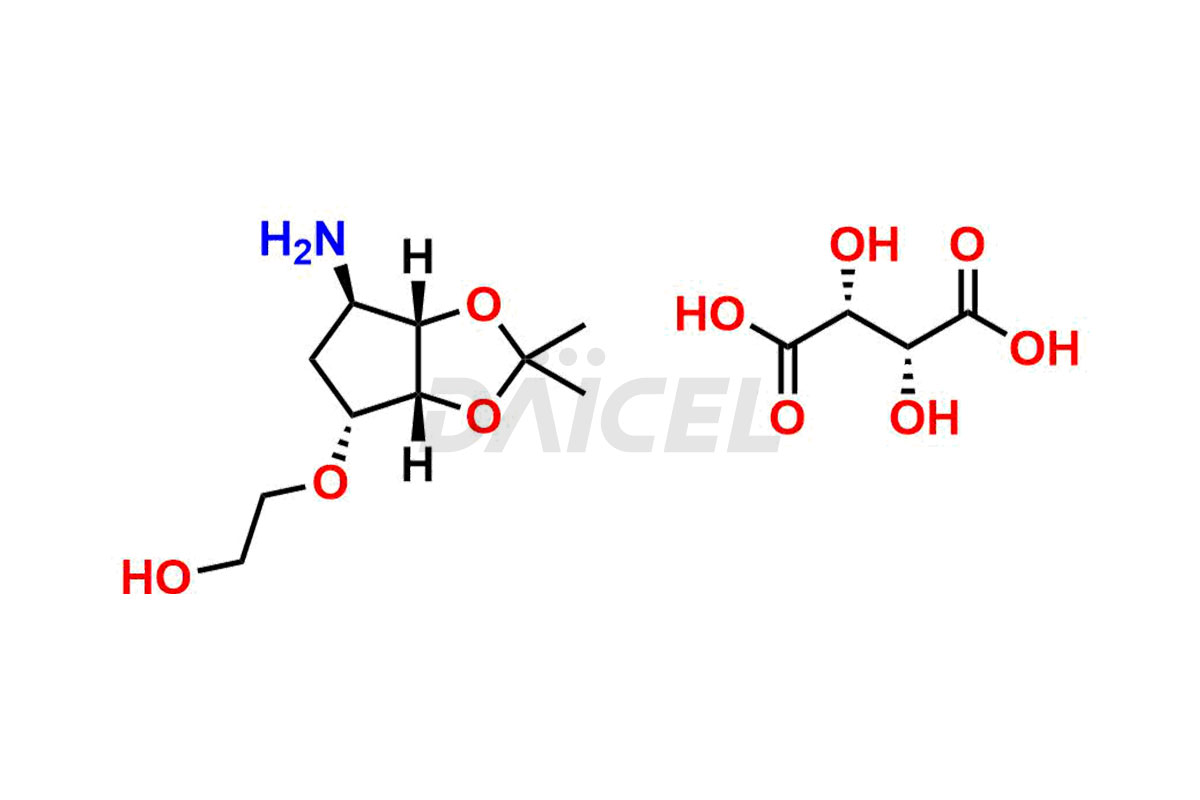
2-(((3aS,4R,6S,6aR)-6-amino-2,2-dimethyltetrahydro...
- CAT Number DCTI-C-004339
- CAS Number 2376278-72-5
- Molecular Formula C14H25NO10 (Free base) C10H19NO4 (tartrate salt)
- Molecular Weight 217.27 (Free base) 367.35 (tartrate salt)
2-(2,3-difluorophenyl)cyclopropanamine hydrochloride
- CAT Number DCTI-C-001585
- CAS Number 1354951-34-0
- Molecular Formula C9H9F2N (free base) C9H10ClF2N (HCl salt)
- Molecular Weight 169.17 (free base) 205.63 (HCl salt)
2-(3-chloro-4-fluorophenyl)cyclopropan-1-amine hydrochloride
- CAT Number DCTI-C-004161
- CAS NUMBER NA
- MOLECULAR FORMULA C9H9ClFN (Free base) C9H10Cl2FN (HCl Salt)
- MOLECULAR WEIGHT 185.63 (Free base) 222.09 (HCl salt)
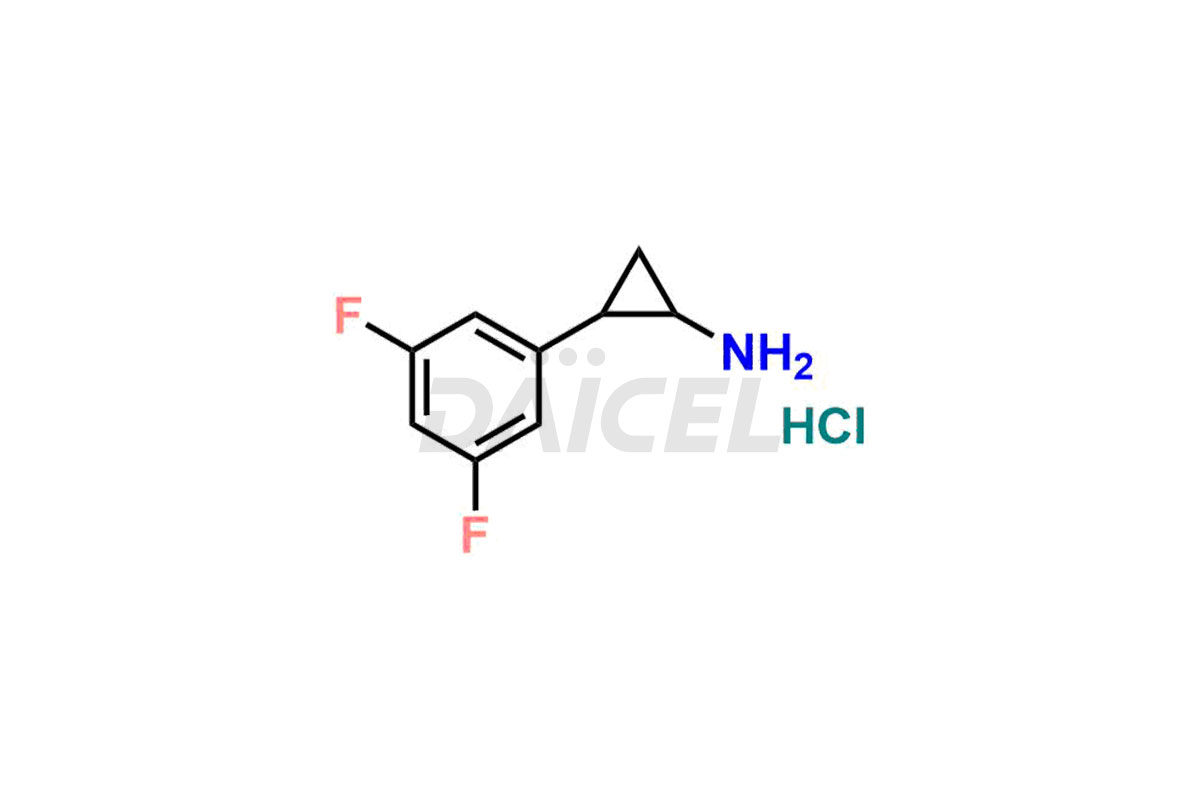
2-(3,5-difluorophenyl)cyclopropanamine hydrochloride
- CAT Number DCTI-C-001586
- CAS Number 1511636-19-3 (free acid)
- Molecular Formula C9H9F2N (free base) C9H10ClF2N (HCl salt)
- Molecular Weight 169.17 (free base) 205.63 (HCl salt)
2-(propylthio)pyrimidine-4,6-Diol
- CAT Number DCTI-C-004149
- CAS NUMBER 145783-12-6
- MOLECULAR FORMULA C7H10N2O2S
- MOLECULAR WEIGHT 186.23
2-thioxodihydropyrimidine-4,6(1H,5H)-dione
- CAT Number DCTI-C-004208
- CAS NUMBER 504-17-6
- MOLECULAR FORMULA C4H4N2O2S
- MOLECULAR WEIGHT 144.15
2,2′-(((3aR,3a’R,4S,4’S,6R,6aS,6...
- CAT Number DCTI-C-004354
- CAS Number NA
- Molecular Formula C27H45N5O8S
- Molecular Weight 599.74
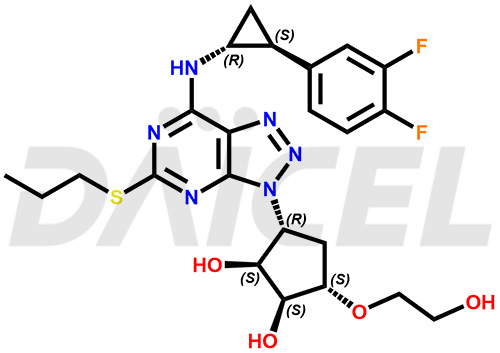
3-(3,4-difluorophenyl)-N-(3-((1R,2S,3S,4S)-2,3-dih...
- CAT Number DCTI-C-004357
- CAS NUMBER NA
- MOLECULAR FORMULA C23H28F2N6O6S
- MOLECULAR WEIGHT 554.57
3-Methoxy ticagrelor
- CAT Number DCTI-C-003851
- CAS NUMBER NA
- MOLECULAR FORMULA C24H31FN6O5S
- MOLECULAR WEIGHT 534.61
3,4-Di desfluoro ticagrelor
- CAT Number DCTI-C-005067
- CAS Number 220346-67-8
- Molecular Formula C23H30N6O4S
- Molecular Weight 486.59
4-chloro-5-nitro-2(propylthio)pyrimidine
- CAT Number DCTI-C-004325
- CAS Number 2518331-26-3
- Molecular Formula C7H8ClN3O2S
- Molecular Weight 233.67
4-Methoxy ticagrelor
- CAT Number DCTI-C-003844
- CAS NUMBER NA
- MOLECULAR FORMULA C24H31FN6O5S
- MOLECULAR WEIGHT 534.61
4-methoxy-5-nitro-2(propylthio)pyrimidine
- CAT Number DCTI-C-004216
- CAS NUMBER 2514951-26-7
- MOLECULAR FORMULA C8H11N3O3S
- MOLECULAR WEIGHT 229.25
4,6-dichloro-2-(propylthio)pyrimidin-5-amine
- CAT Number DCTI-C-001520
- CAS Number 145783-15-9
- Molecular Formula C7H9Cl2N3S (free base)
- Molecular Weight 238.13
4,6-dichloro-5-nitro-2-(propylthio)pyrimidine
- CAT Number DCTI-C-001519
- CAS Number 145783-14-8
- Molecular Formula C7H7Cl2N3O2S
- Molecular Weight 268.11
5-amino-6-chloro-2-(propylthio)pyrimidin-4-ol
- CAT Number DCTI-C-004194
- CAS NUMBER 2512210-21-6
- MOLECULAR FORMULA C7H10ClN3OS
- MOLECULAR WEIGHT 219.69
5-Desthiopropyl-5-hydroxy-ticagrelor
- CAT Number DCTI-C-004480
- CAS Number 1644461-81-3
- Molecular Formula C20H22F2N6O5
- Molecular Weight 464.43
5-nitro-2-(propylthio)pyrimidine-4,6-diol
- CAT Number DCTI-C-004206
- CAS NUMBER 145783-13-7
- MOLECULAR FORMULA C7H9N3O4S
- MOLECULAR WEIGHT 231.23
6-chloro-5-nitro-2-(propylthio)pyrimidin-4(3H)-one
- CAT Number DCTI-C-004193
- CAS NUMBER 1548397-10-9
- MOLECULAR FORMULA C7H8ClN3O3S
- MOLECULAR WEIGHT 249.67
Cyclopropyl-cyclopentyl-diol Impurity
- CAT Number DCTI-C-004308
- CAS NUMBER 2271293-30-0
- MOLECULAR FORMULA C23H26F2N6O4S
- MOLECULAR WEIGHT 520.56
Ethanol, 2-[[(3aR,4S,6R,6aS)-6-aminotetrahydro-2,2...
- CAT Number DCTI-C-001004
- CAS Number 274693-55-9
- Molecular Formula C10H19NO4
- Molecular Weight 217.27
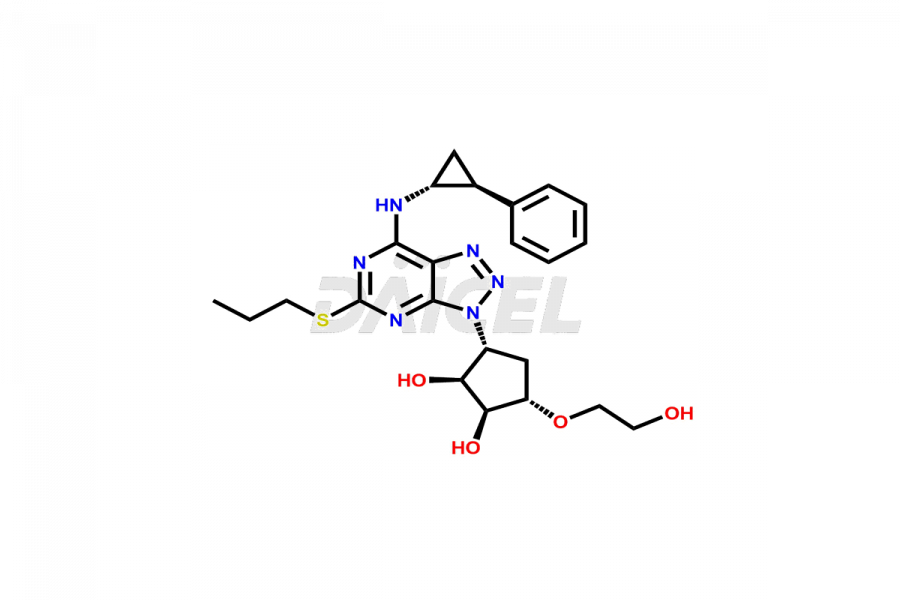
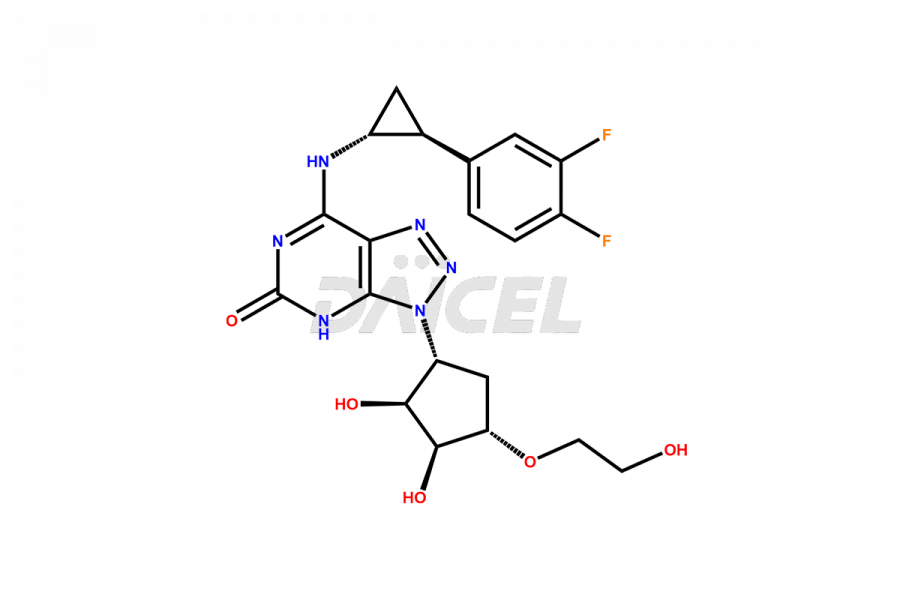
N-(3-((1R,2S,3S,4S)-2,3-dihydroxy-4-(2-hydroxyetho...
- CAT Number DCTI-C-004369
- CAS Number NA
- Molecular Formula C15H22N6O5S
- Molecular Weight 398.44
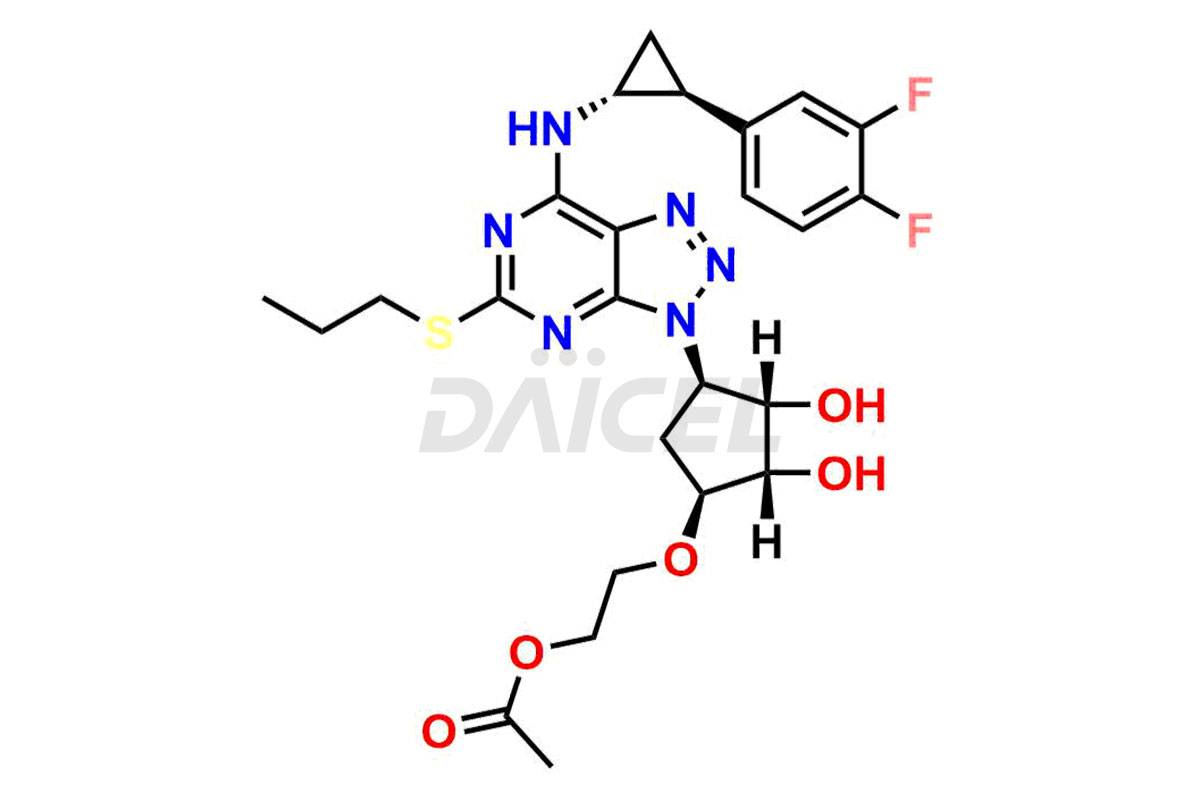
O-acetyl Ticagrelor
- CAT Number DCTI-C-001587
- CAS Number 1616703-93-5
- Molecular Formula C25H30F2N6O5S
- Molecular Weight 564.61
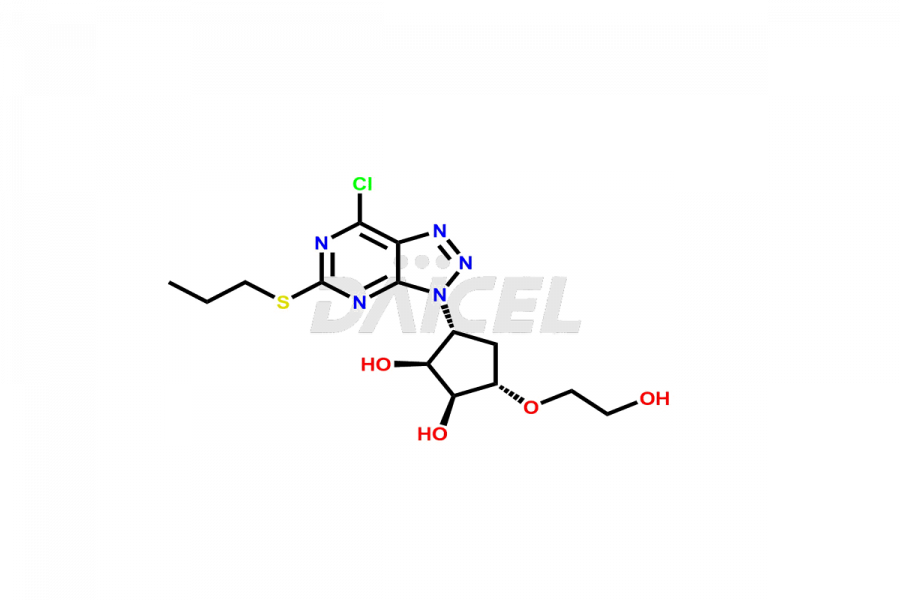
Ticagrelor 7-Chloro Impurity
- CAT Number DCTI-C-005069
- CAS Number 1354945-69-9
- Molecular Formula C14H20ClN5O4S
- Molecular Weight 389.86
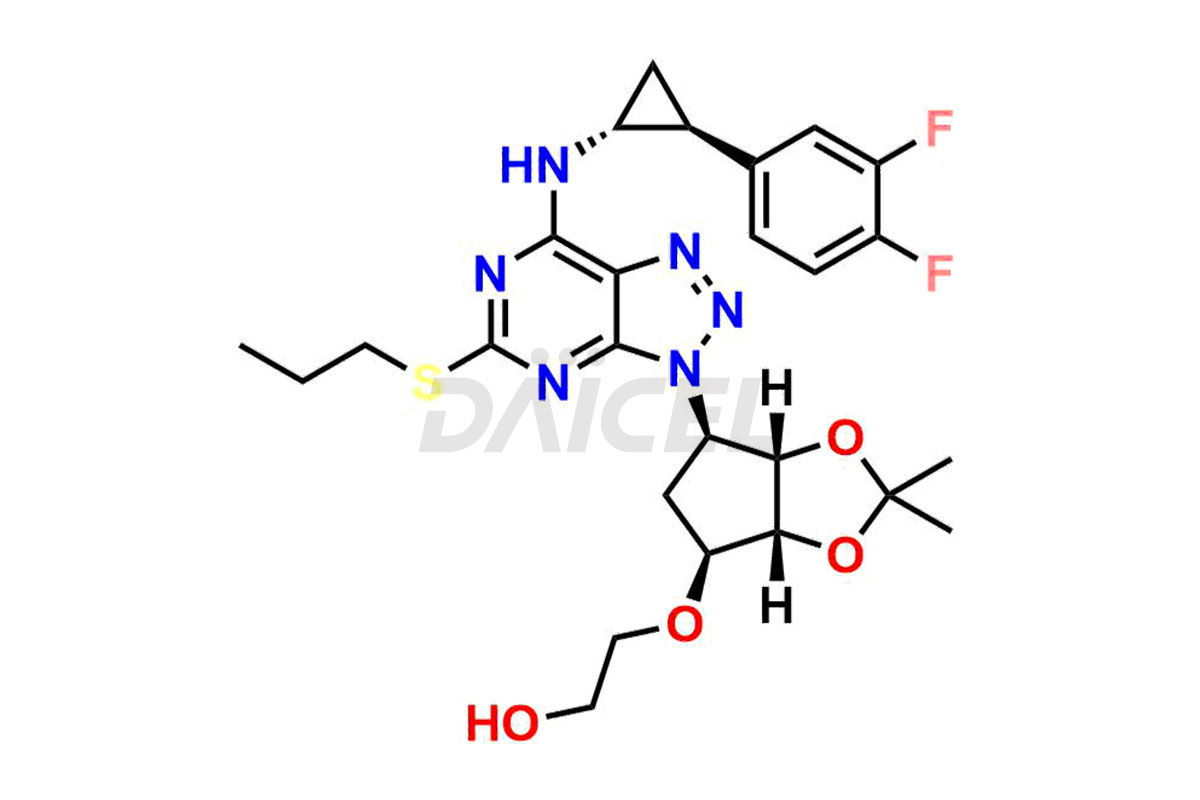
Ticagrelor acetonide
- CAT Number DCTI-C-000957
- CAS Number 274693-26-4
- Molecular Formula C26H32F2N6O4S
- Molecular Weight 562.63
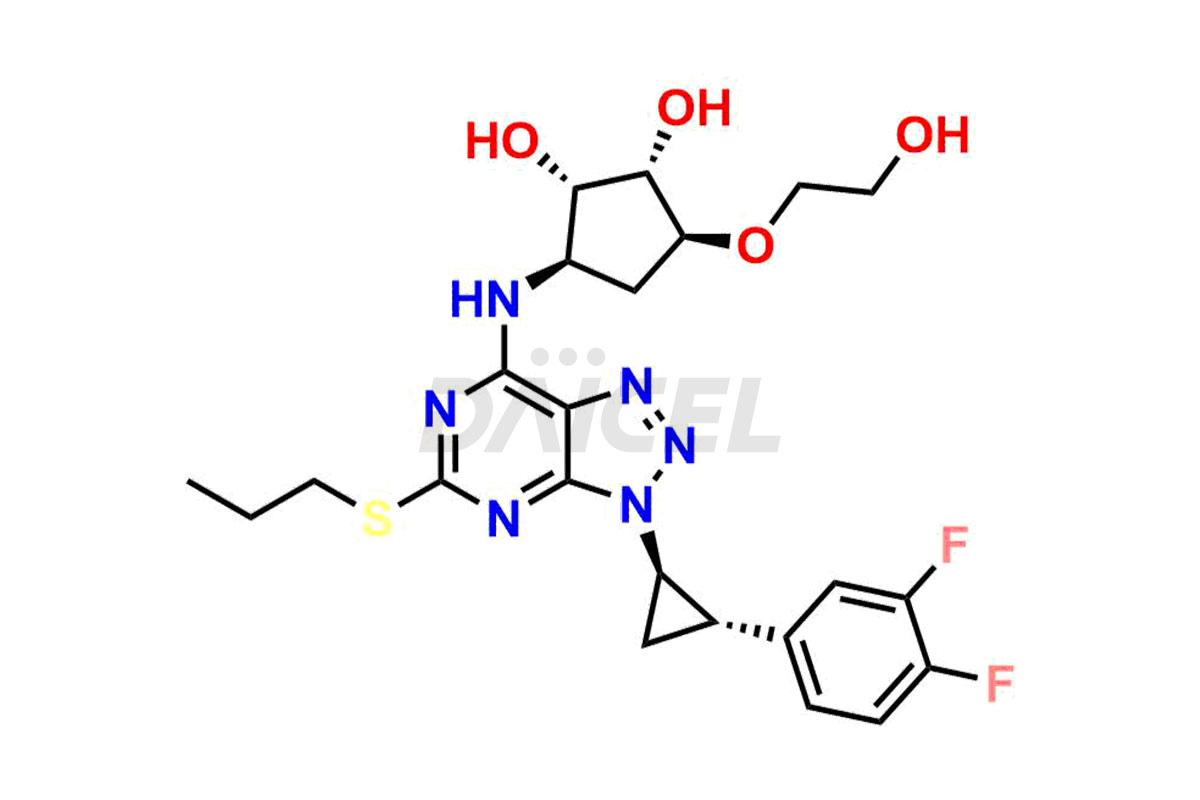
Ticagrelor cyclized impurity
- CAT Number DCTI-C-004236
- CAS NUMBER NA
- MOLECULAR FORMULA C23H27F2N7O5S
- MOLECULAR WEIGHT 551.57
Ticagrelor Diastereo isomer (C-6)
- CAT Number DCTI-C-002933
- CAS NUMBER 2376278-70-3
- MOLECULAR FORMULA C14H25NO10 (Tartrate salt); C10H19NO4 (free base)
- MOLECULAR WEIGHT 367.35 (Tartrate salt); 217.27 (Free Base)
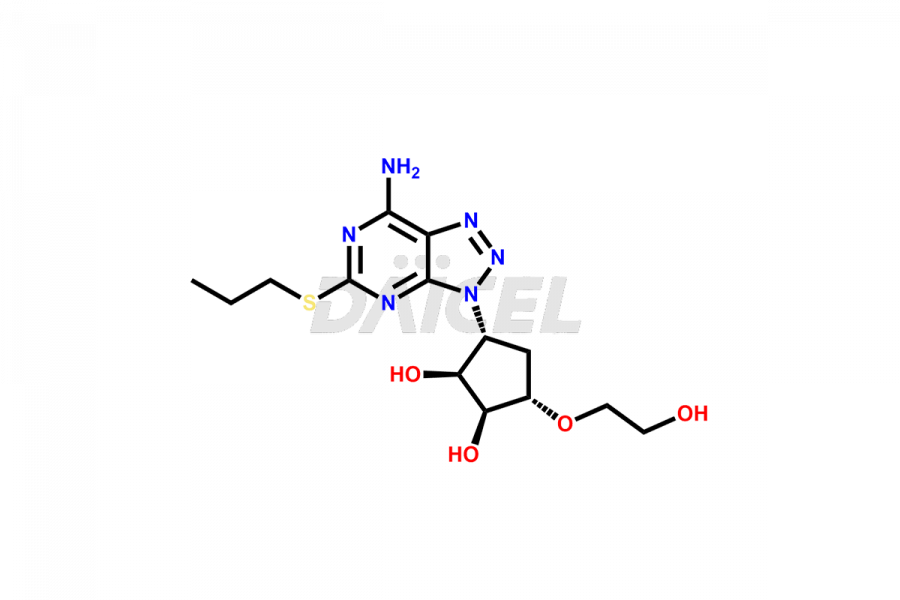
Ticagrelor EP impurity A
- CAT Number DCTI-C-000999
- CAS NUMBER NA
- MOLECULAR FORMULA C14H22N6O4S
- MOLECULAR WEIGHT 370.43
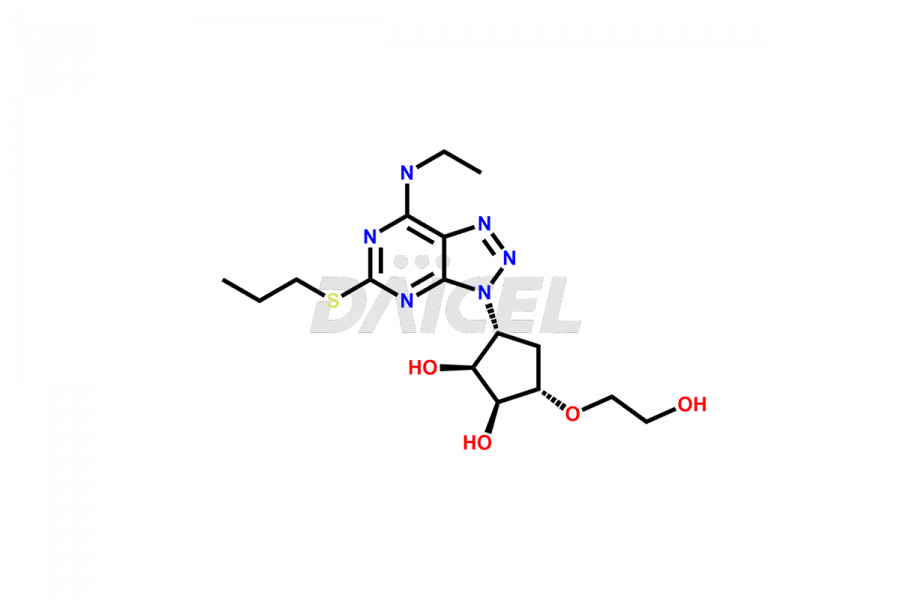
Ticagrelor Ethyl amine impurity
- CAT Number DCTI-C-003917
- CAS NUMBER NA
- MOLECULAR FORMULA C16H26N6O4S
- MOLECULAR WEIGHT 398.48
Ticagrelor impurity
- CAT Number DCTI-C-001003
- CAS Number 155899-66-4
- Molecular Formula C8H15NO3
- Molecular Weight 173.21
Ticagrelor O-acetyl impurity
- CAT Number DCTI-C-000998
- Molecular Formula C25H30F2N6O6S
- Molecular Weight 580.61
Ticagrelor O-Tert-Butyl Impurity
- CAT Number DCTI-C-004309
- CAS NUMBER NA
- MOLECULAR FORMULA C27H36F2N6O4S
- MOLECULAR WEIGHT 578.68
TICAGRELOR R,R,S,R-ISOMER
- CAT Number DCTI-C-004750
- CAS Number 2096989-55-6
- Molecular Formula C23H28F2N6O4S
- Molecular Weight 522.57
Ticagrelor R,S-cyclopropyl isomer
- CAT Number DCTI-C-004749
- CAS Number 2435586-30-2
- Molecular Formula C23H28F2N6O4S
- Molecular Weight 522.57
Ticagrelor Related Compound 27
- CAT Number DCTI-C-002406
- CAS NUMBER 1816939-47-5 (Free Base)
- MOLECULAR FORMULA C10H19NO4 (Free base); C14H25NO10(D-Tartaric acid salt)
- MOLECULAR WEIGHT 217.27 (Free base); 367.35 (D-Tartaric acid salt)
Ticagrelor Related Compound 91
- CAT Number DCTI-C-001204
- CAS Number 2376278-67-8 (acid free)
- Molecular Formula C14H25NO10 (Salt) C10H19NO4 (Free base)
- Molecular Weight 367.35 (Salt) 217.27 Free base
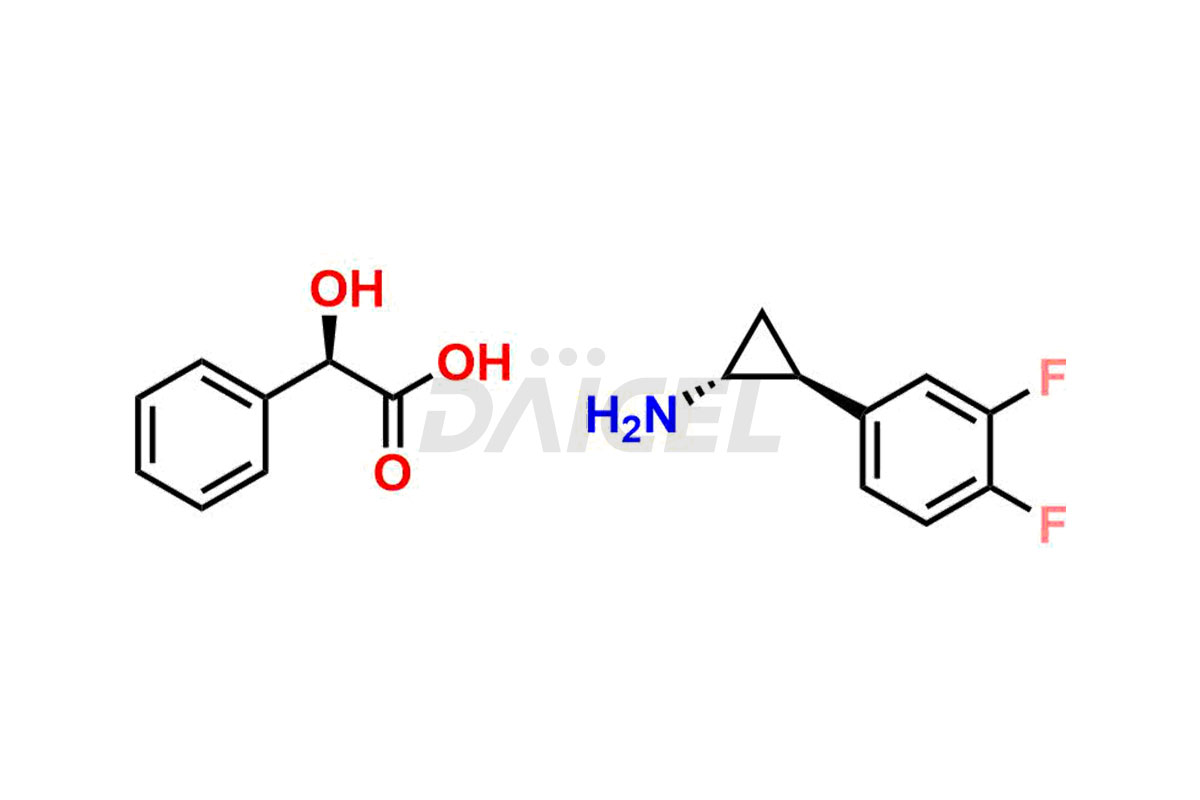
Ticagrelor Related Compound A
- CAT Number DCTI-C-001001
- CAS Number 376608-71-8
- Molecular Formula C9H9F2N (free base) C17H17F2NO3(with base)
- Molecular Weight 169.17 (free base) 321.32 (with base)
Ticagrelor Related Impurity
- CAT Number DCTI-C-001588
- CAS Number NA
- Molecular Formula C10H19NO4 (free base) C14H25NO10 (salt)
- Molecular Weight 217.27 (free base) 367.35 (salt)
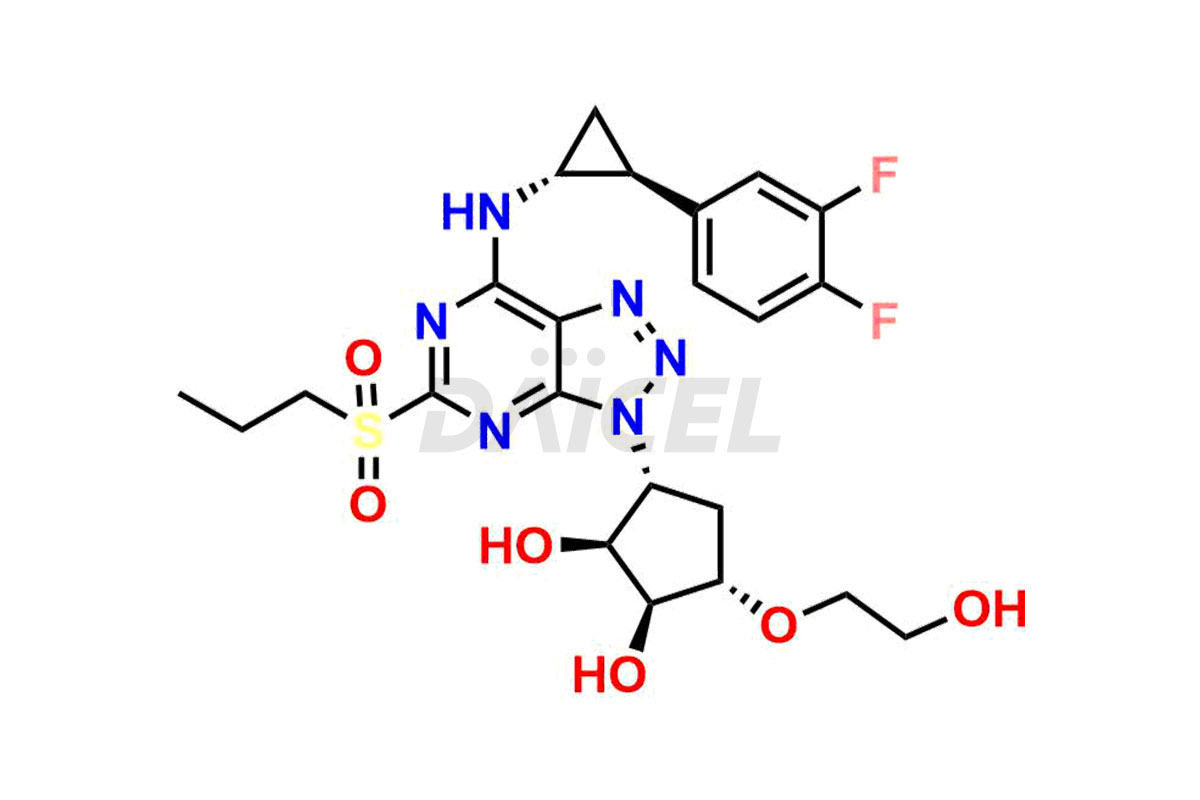
Ticagrelor Sulfone
- CAT Number DCTI-C-000955
- CAS Number 274693-39-9
- Molecular Formula C23H28F2N6O6S
- Molecular Weight 554.57
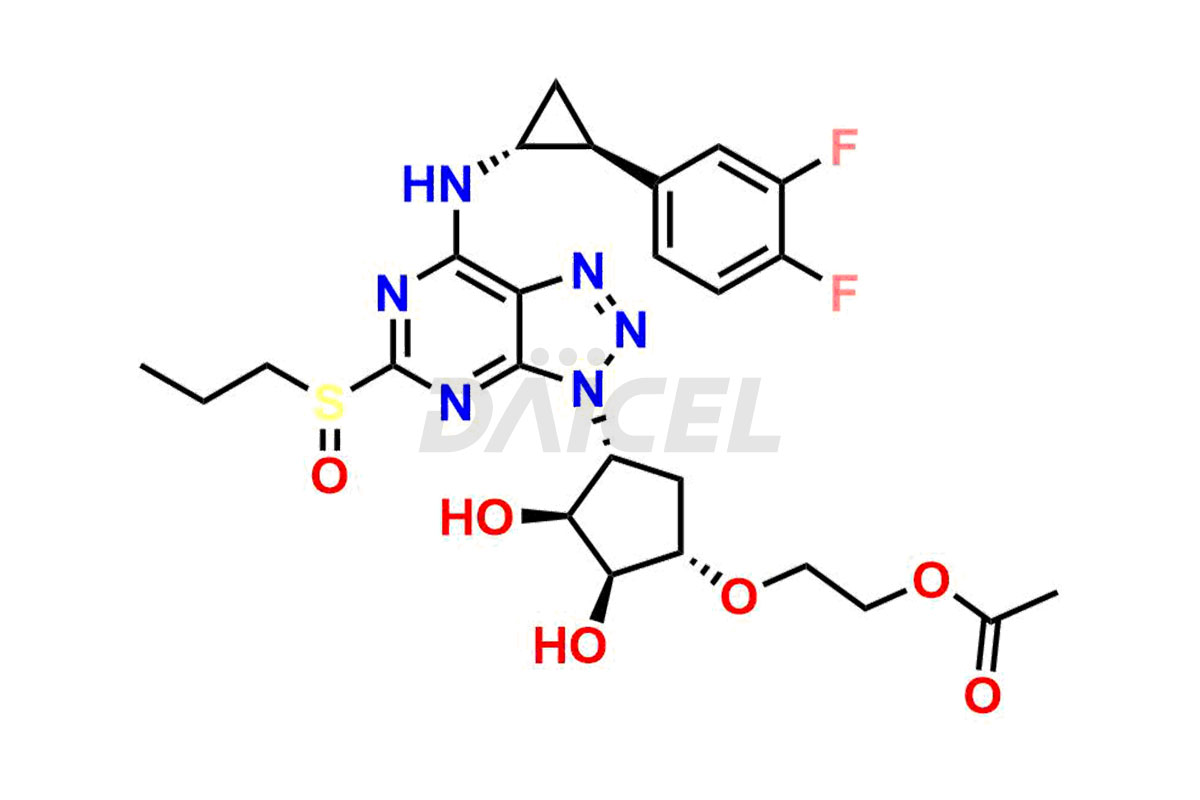
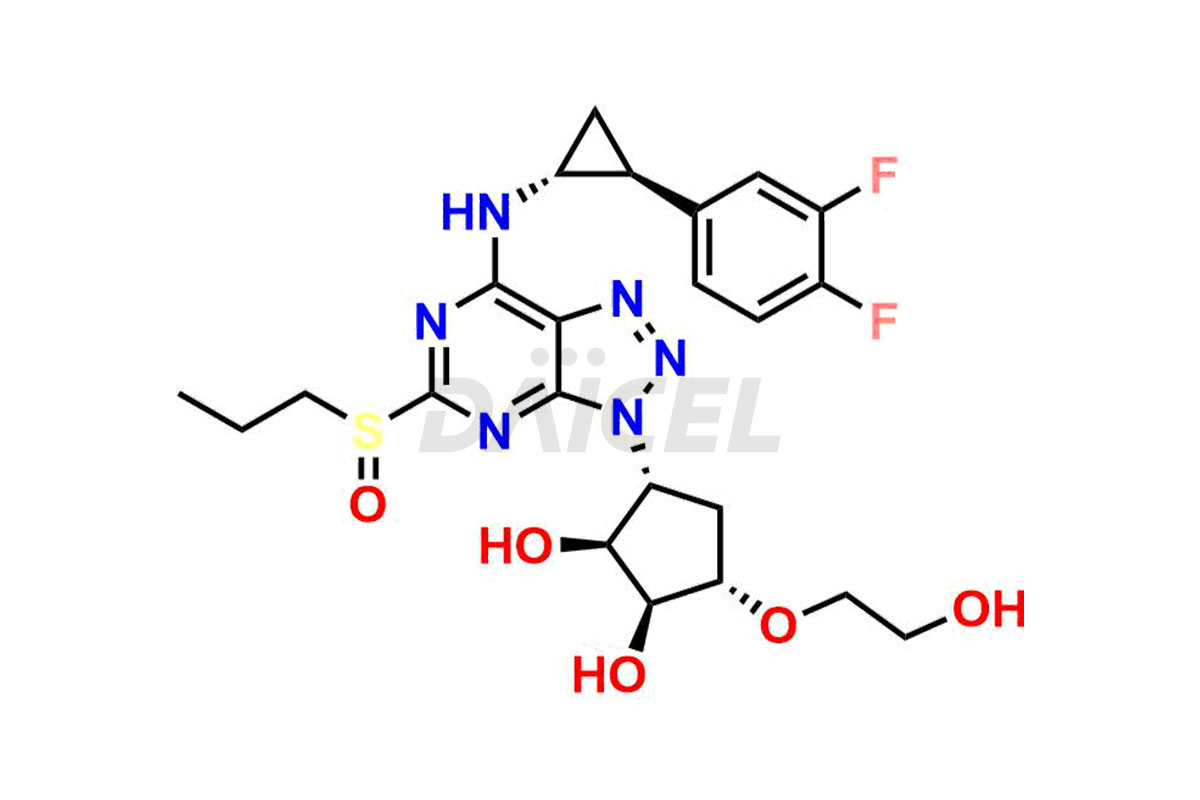
Ticagrelor Sulfoxide
- CAT Number DCTI-C-000956
- CAS Number 1644461-85-7
- Molecular Formula C23H28F2N6O5S
- Molecular Weight 538.57
Ticagrelor Triazole isomer
- CAT Number DCTI-C-001000
- CAS Number 1788033-05-5
- Molecular Formula C23H28F2N6O4S
- Molecular Weight 522.57
TKGIP-2
- CAT Number DCTI-C-004235
- CAS NUMBER 1265919-24-1
- MOLECULAR FORMULA C12H21NO5 (Free base) C12H22ClNO5 (HCl salt
- MOLECULAR WEIGHT 259.30 (Free base) 295.76 (HCl salt)