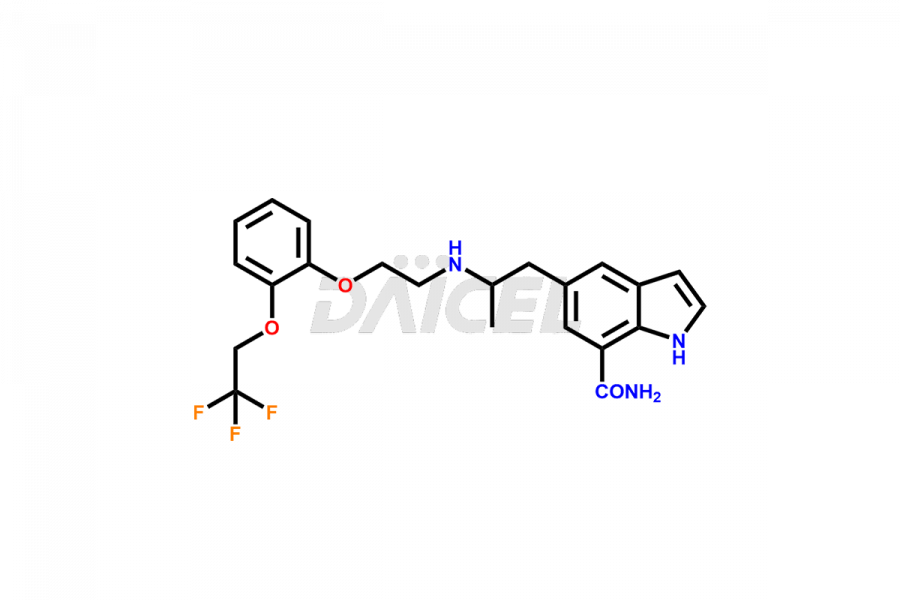
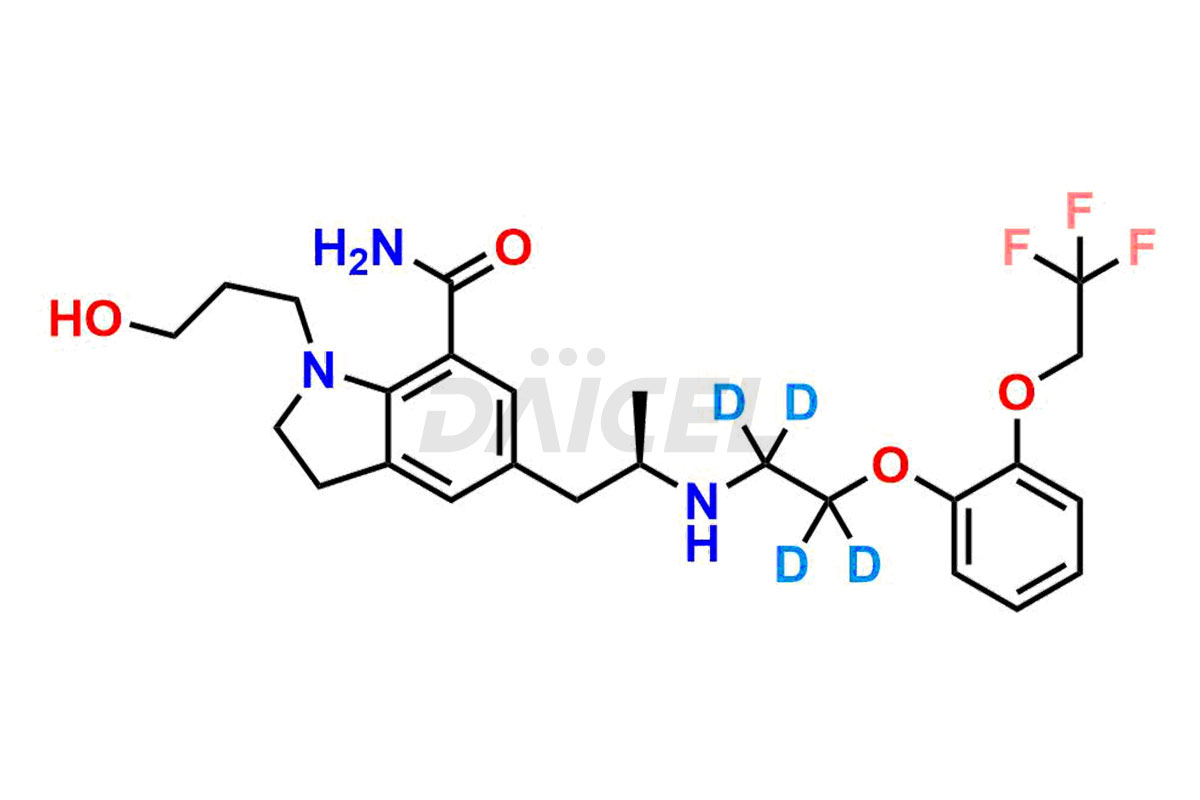
Silodosin
References
- Kitazawa, Makio; Ban, Masaaki; Okazaki, Kosuke; Ozawa, Motoyasu; Yazaki, Toshikazu; Yamagishi, Ryoichi, Indoline compounds for the treatment of dysuria, Kissei Pharmaceutical Co., Ltd., Japan, EP600675, July 8, 1998
- Zhao, Xia; Liu, Yuwang; Xu, Junyu; Zhang, Dan; Zhou, Ying; Gu, Jingkai; Cui, Yimin, Determination of silodosin in human plasma by liquid chromatography-tandem mass spectrometry, Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences, Volume: 877, Issue: 29, Pages: 3724-3728, 2009
Frequently Asked Questions
Why is the presence of impurities a concern in Silodosin?
Impurities in Silodosin can affect its quality, safety, and efficacy. Depending on the type and level of impurities, they can impact the drug's pharmacological activity and stability and pose potential risks to patient health.
How are impurities in Silodosin detected and quantified?
Impurities in Silodosin are detected and quantified using reversed-phase (RP) ultra-high-performance liquid chromatography (UHPLC). This method will allow for accurate identification and quantification of impurities.
What steps can be taken to control impurity levels in Silodosin during manufacturing?
Manufacturers can adopt several strategies to maintain control over impurity levels in Silodosin. They include using high-quality starting materials, optimizing synthesis and purification processes, implementing comprehensive quality control tests, and monitoring impurity levels at different stages of the manufacturing process.
What are the temperature conditions required to store Silodosin Impurities?
Silodosin Impurities are stored at a controlled room temperature between 2-8°C or as indicated on the Certificate of Analysis (CoA).
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.