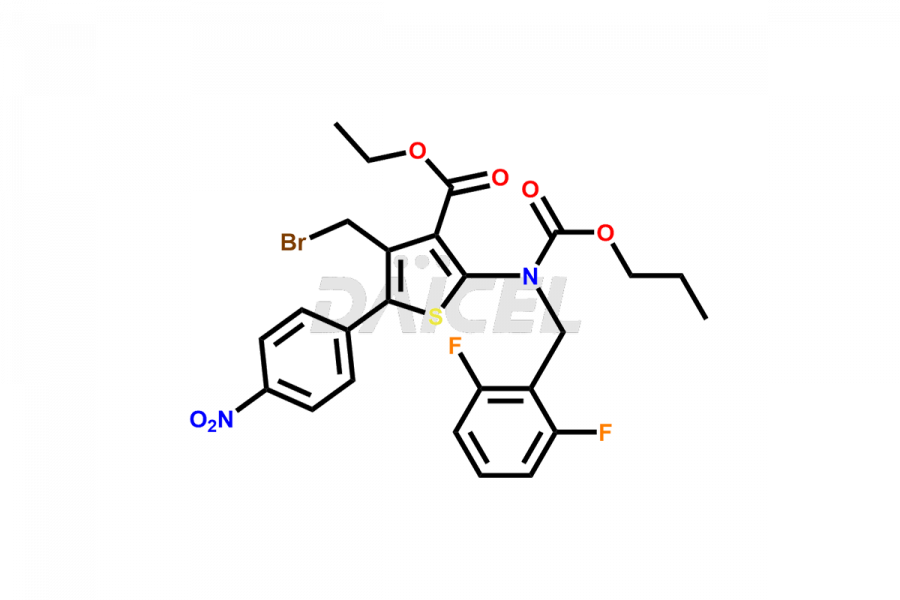
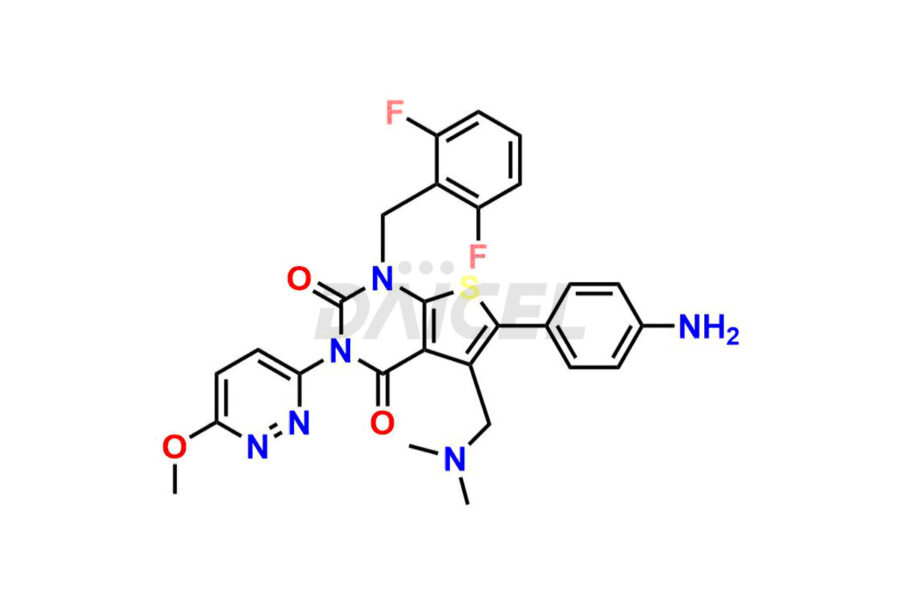
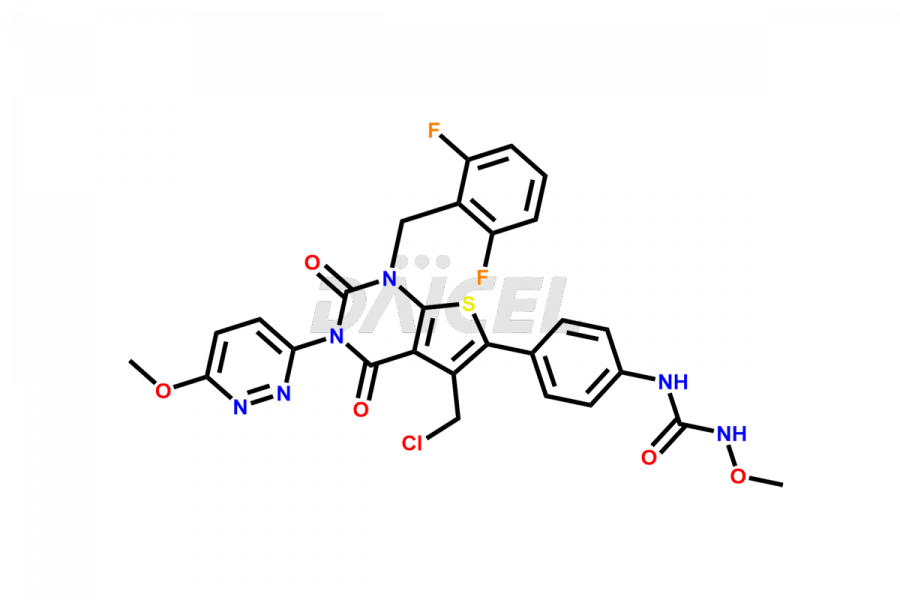
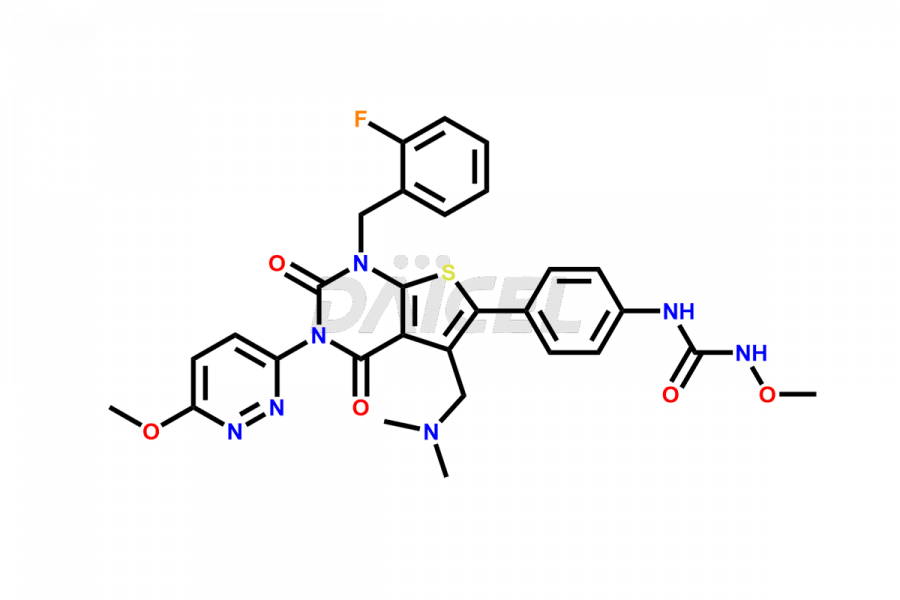
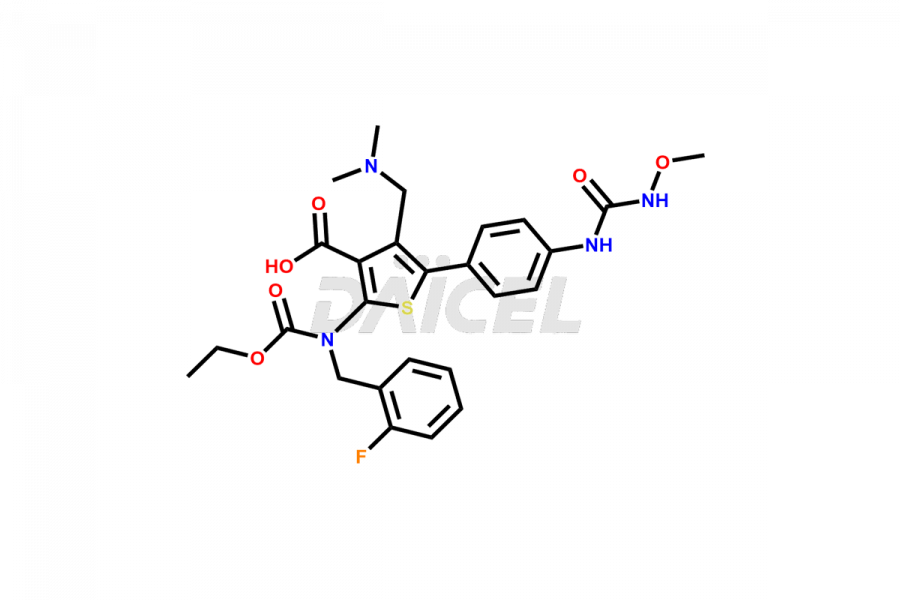
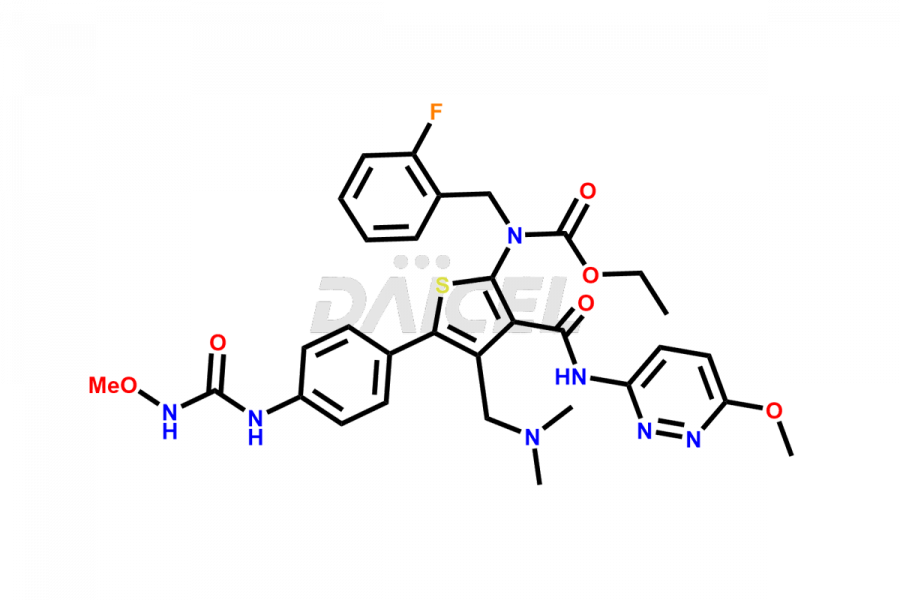
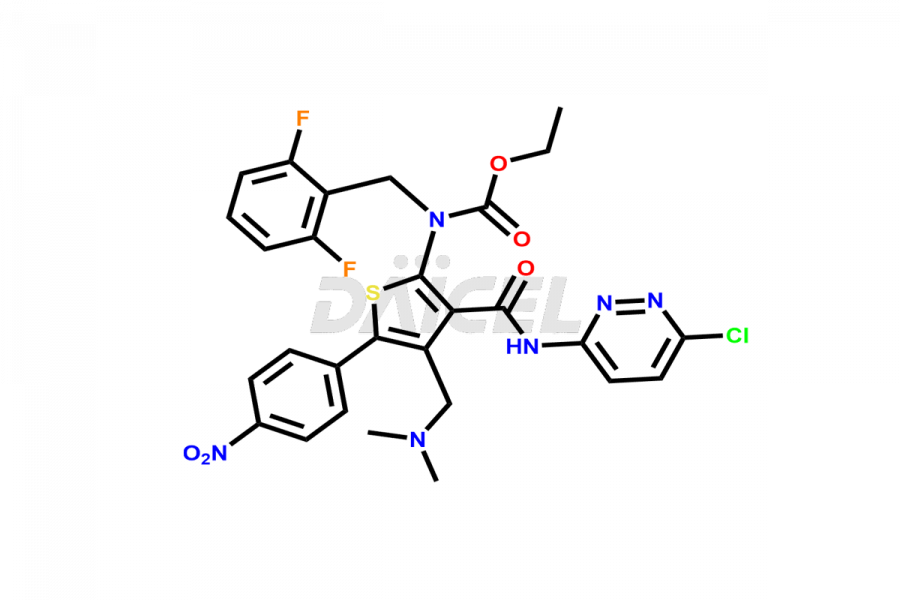
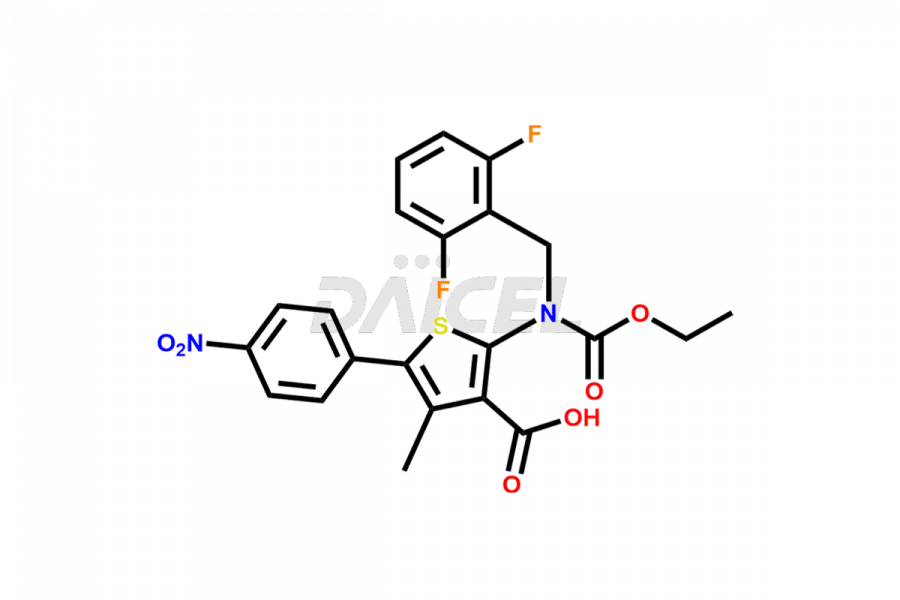
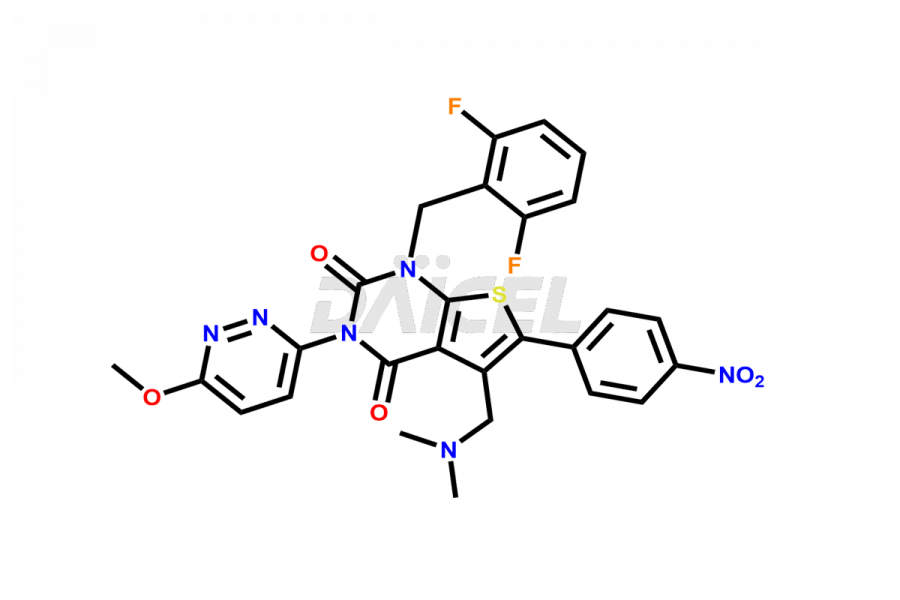
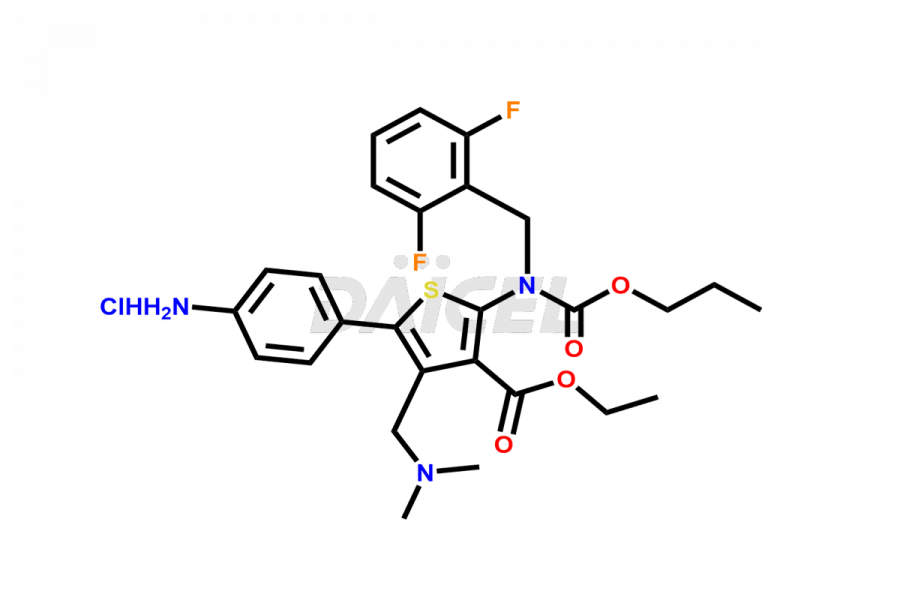
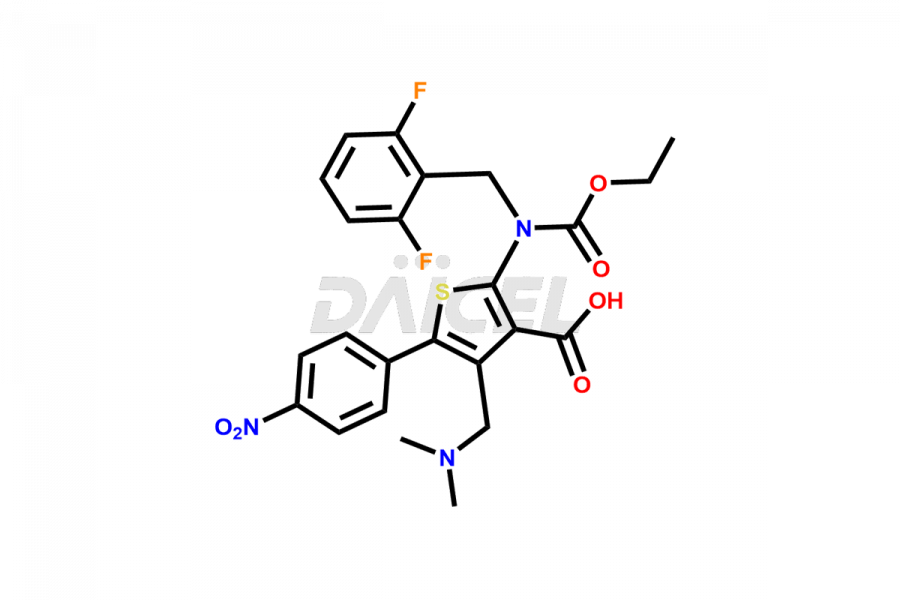
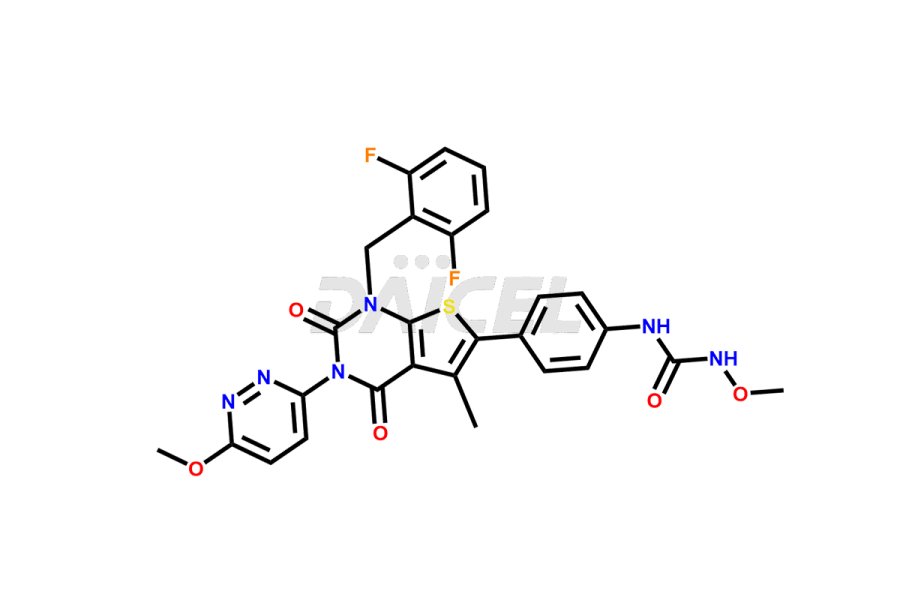
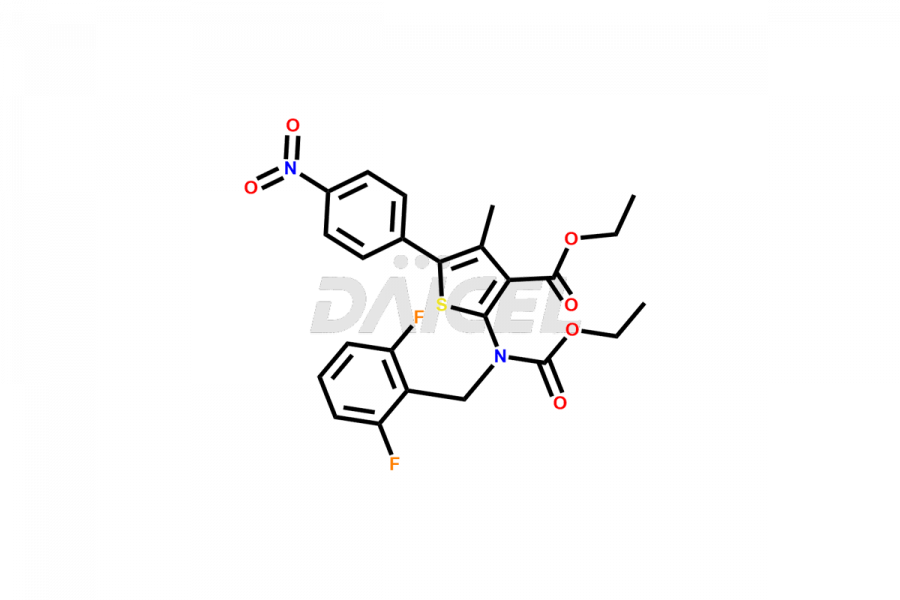
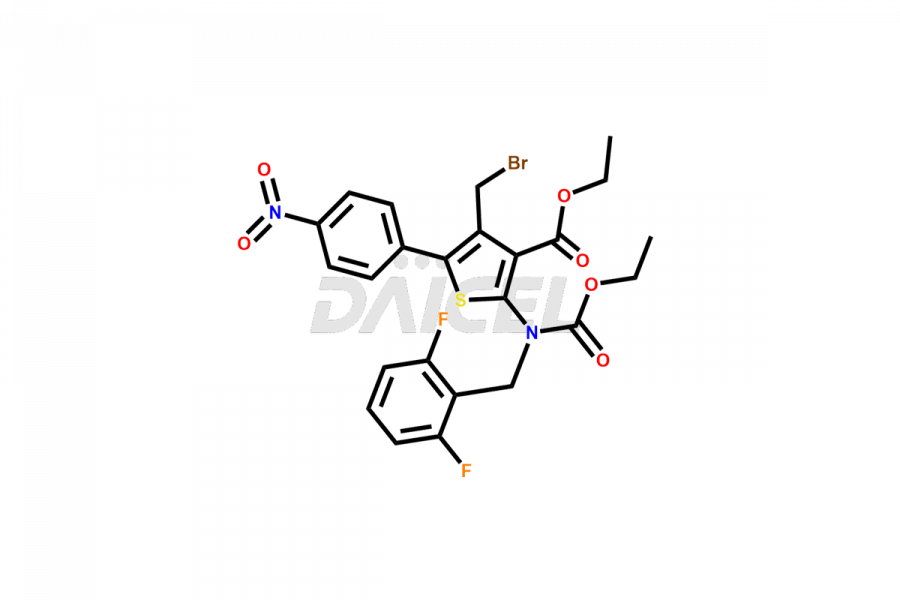
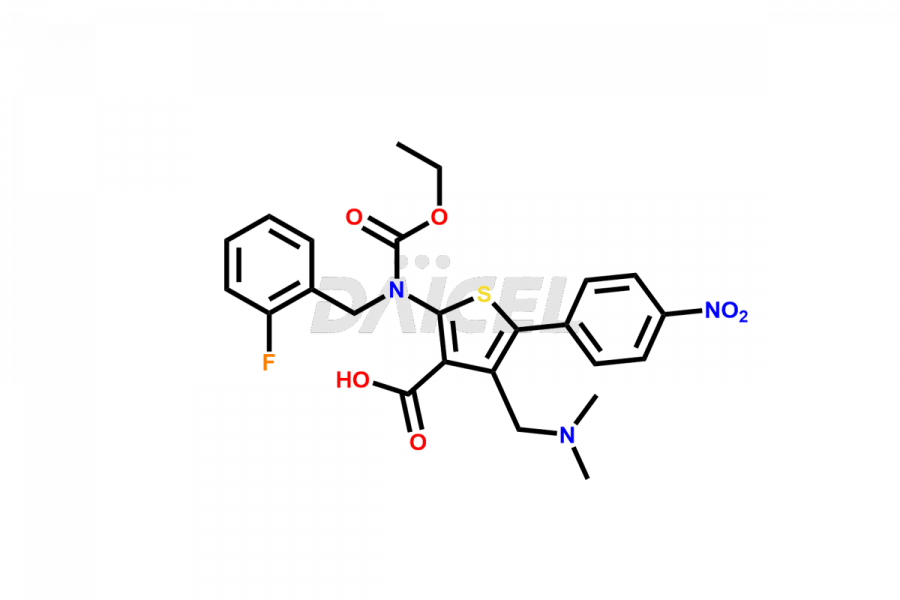
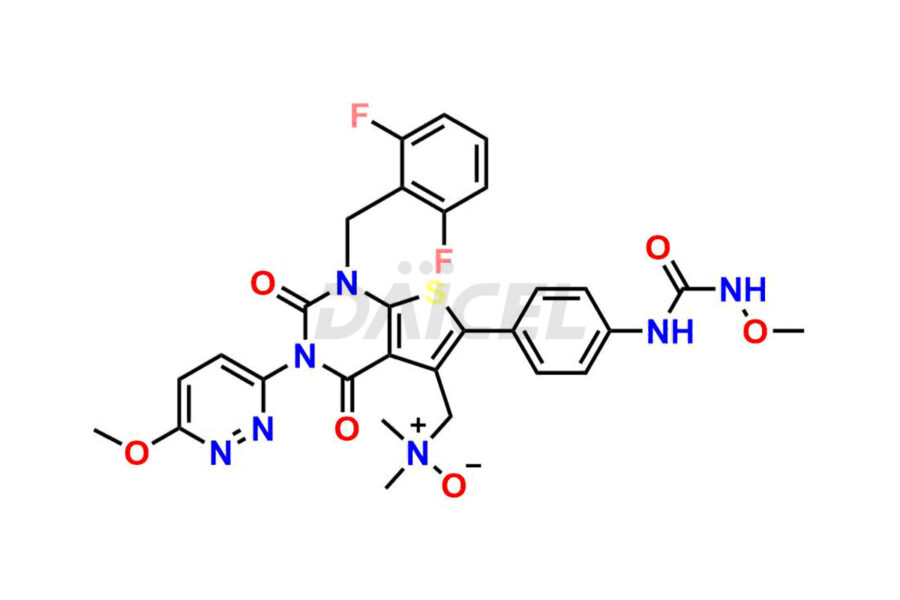
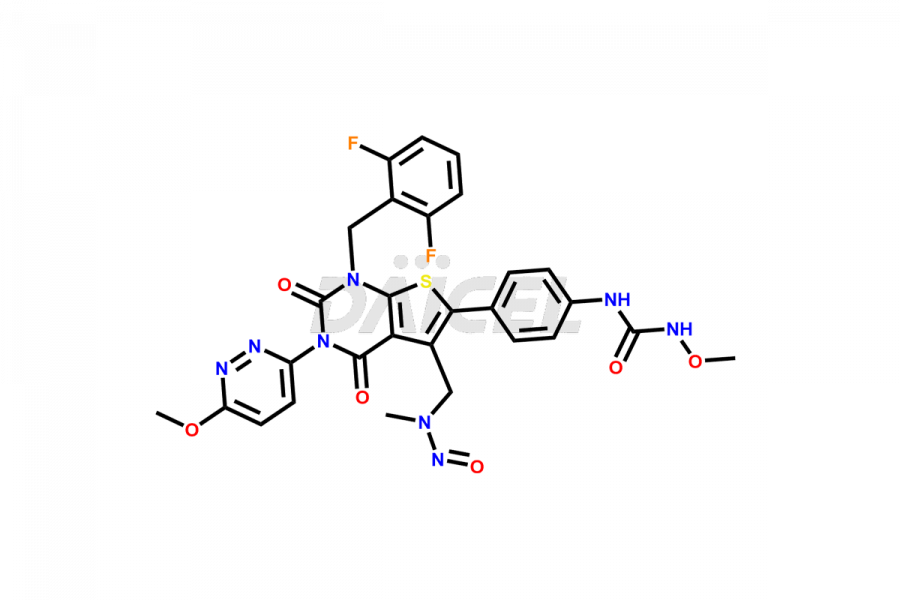
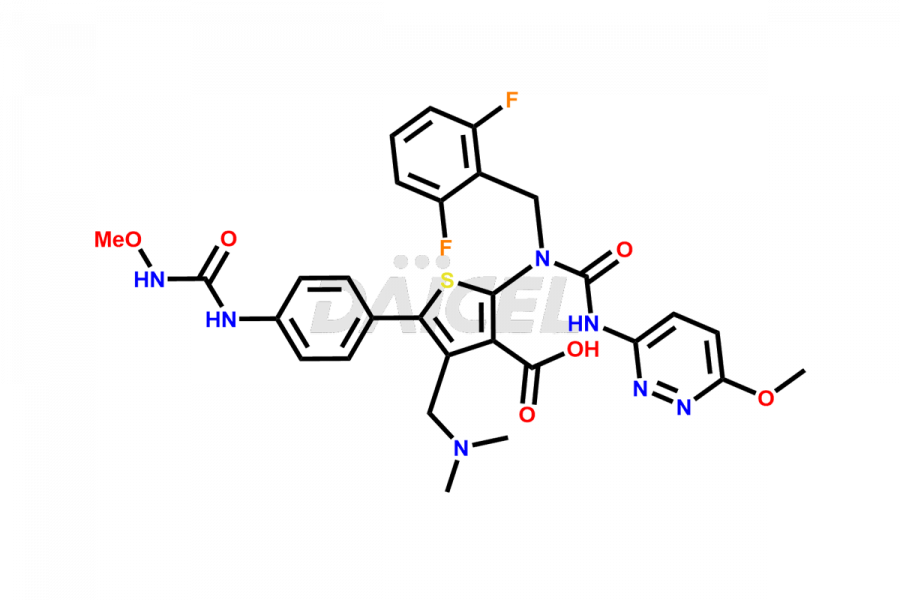
Relugolix Impurities
Daicel supplies relugolix impurity standards for impurity profiling and method development in hormone-related therapies.
General Information
Relugolix Impurities and Relugolix
Daicel Pharma synthesizes more than fifteen high-quality Relugolix impurities, such as Relugolix Amine Impurity, Relugolix Chloro Impurity, Relugolix Desfluoro Impurity, Relugolix Desfluoro ureido acid, Relugolix N-oxide Impurity, Relugolix thiophene acid impurity, and more, are crucial in determining the quality, stability, and biological safety of the active pharmaceutical ingredient, Relugolix. Moreover, Daicel offers custom synthesis of Relugolix impurities and delivers them globally.
Relugolix [CAS: 737789-87-6] is a thienopyrimidine derivative, a non-peptide antagonist that works as a gonadotropin-releasing hormone (GnRH or luteinizing hormone-releasing hormone (LHRH)) receptor antagonist. It has potential antineoplastic activity and treats hormone-responsive conditions, including uterine fibroids and prostate cancer.
Relugolix: Use and Commercial Availability
Relugolix was approved in 2020 by the FDA under the brand name, Orgovyx for treating advanced prostate cancer. It is available as an oral tablet, approved for prostate cancer treatment, providing a more convenient option for patients.

Relugolix Structure and Mechanism of Action
The chemical formula for Relugolix is C29H27F2N7O5S, and its molecular weight is approximately 623.6 g/mol.
Relugolix competitively binds to pituitary GnRH receptors, lowers the release of luteinizing hormone (LH), Follicle Stimulating Hormone (FSH), and thus testosterone.
Relugolix Impurities and Synthesis
Relugolix impurities include related substances such as Relugolix isomer and N-oxide impurity, residual solvents like acetone and methanol, inorganic substances such as lead and arsenic, and degradation products like Relugolix dimer that may form during different stages of synthesis1 and storage of the drug. Hence, it is critical to control the levels of impurities in Relugolix to ensure the quality and safety of the drug.
Daicel offers a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for more than fifteen Relugolix impurity standards2 including Relugolix Amine Impurity, Relugolix Chloro Impurity, Relugolix Desfluoro Impurity, Relugolix Desfluoro ureido acid, Relugolix N-oxide Impurity, Relugolix thiophene acid impurity, etc. The CoA includes complete characterization data, such as 1H NMR, 13C NMR, IR, MASS, and HPLC purity. We also provide 13C-DEPT and CHN on request. We also give a complete characterization report on delivery.
Daicel has the technology and expertise to prepare any unknown Relugolix impurity or degradation product. We also provide labeled compounds to quantify the efficacy of generic Relugolix. Daicel offers Relugolix-D6, a deuterium-labeled standard of Relugolix for bioanalytical research and BA/BE studies with the percentage of isotope data in CoA.
References
FAQ's
References
- Cho, Nobuo; Imada, Takashi; Hitaka, Takenori; Miwa, Kazuhiro; Kusaka, Masami; Suzuki, Nobuhiro, “Preparation of thienopyrimidine derivatives as gonadotropin-releasing hormone antagonists”, Takeda Chemical Industries, Ltd., US7300935B2, Nov 27, 2007
- Zhou, Bugao; Shen, Yuqi; Xu, Guanghui; Hui, Jian; Zhang, Mingyu, “Preparation of relugolix impurity pyridazine derivative” Nanjing F&S Pharmatech Co., Ltd., Jiangsu Litaier Pharmaceutical Co., Ltd., China, CN114478503A, May 13, 2022
Frequently Asked Questions
What types of Relugolix impurities may be present in the drug?
The most common types of Relugolix impurities are related compounds, residual solvents, and heavy metals.
What are the common degradation impurities of Relugolix?
The common Relugolix degradation impurities include acid impurities, oxidative impurities and other impurities.
How are Relugolix impurities detected and quantified?
Impurities in Relugolix are detected and quantified using various analytical techniques such as high-performance liquid chromatography (HPLC), liquid chromatography-mass spectrometry (LC-MS), etc.
How to remove Relugolix impurities during the synthesis of the drug?
During the manufacturing process of Relugolix, various purification techniques such as recrystallization and chromatography help to remove impurities.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.