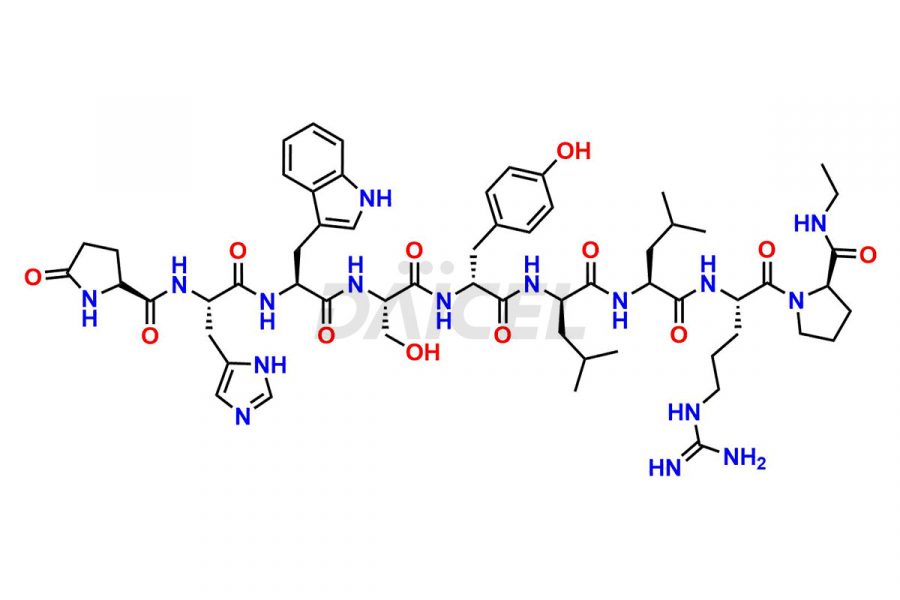
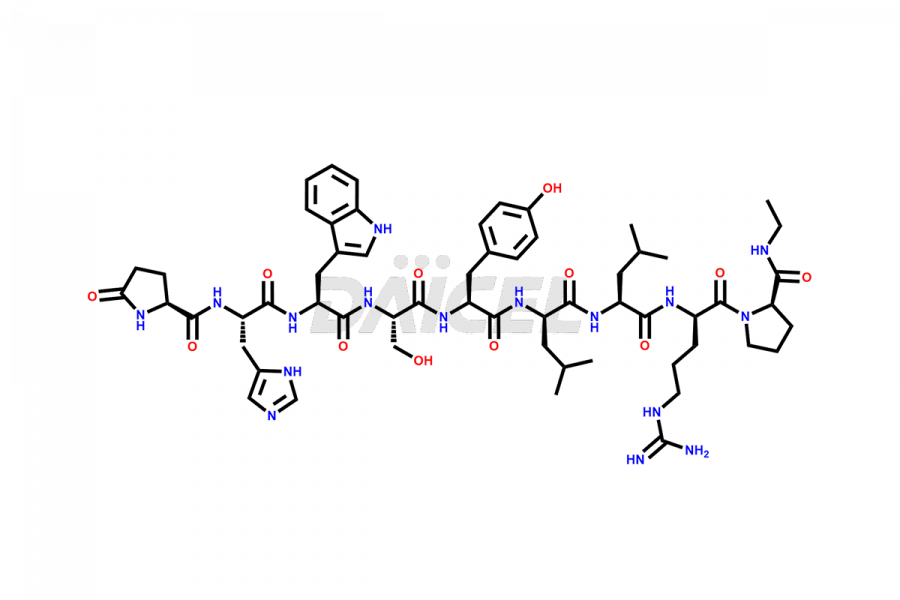
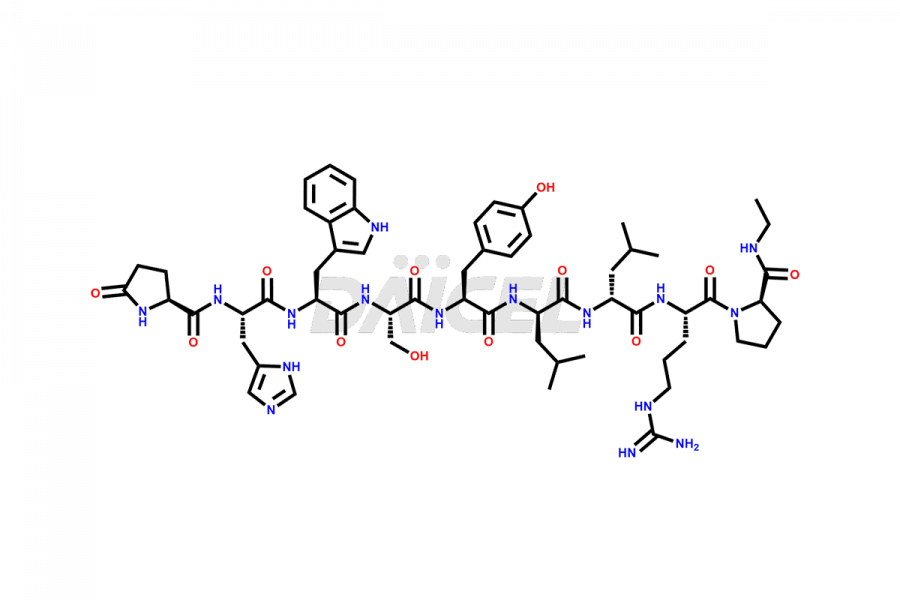
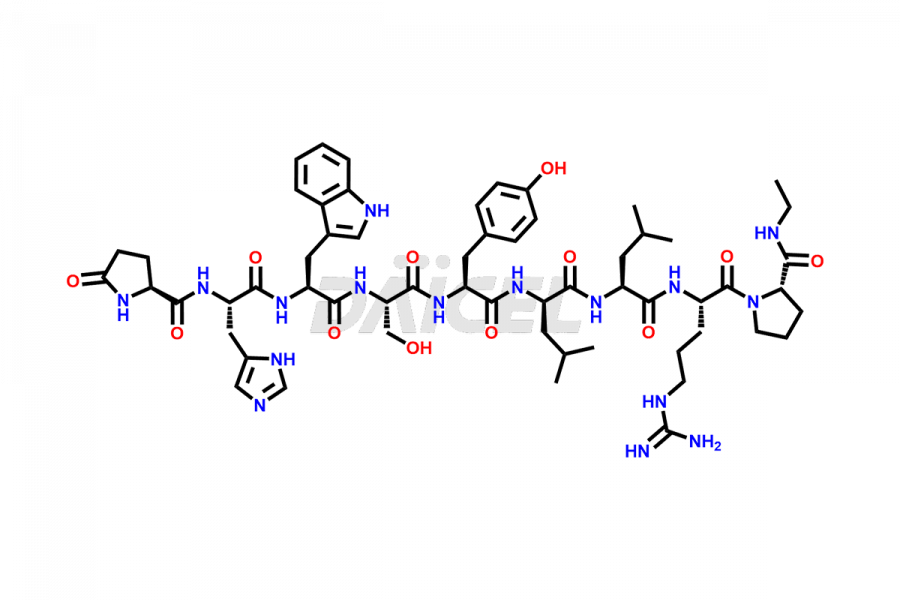
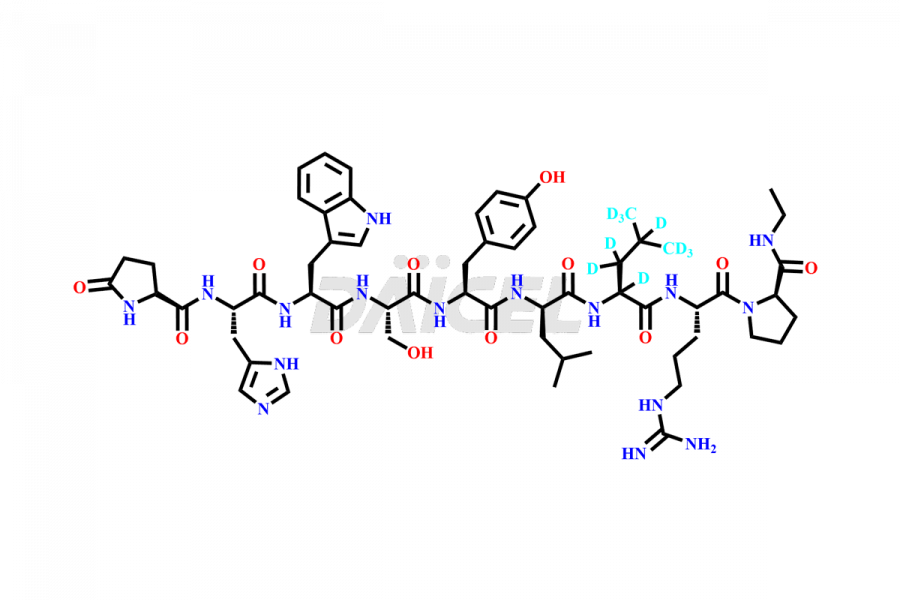
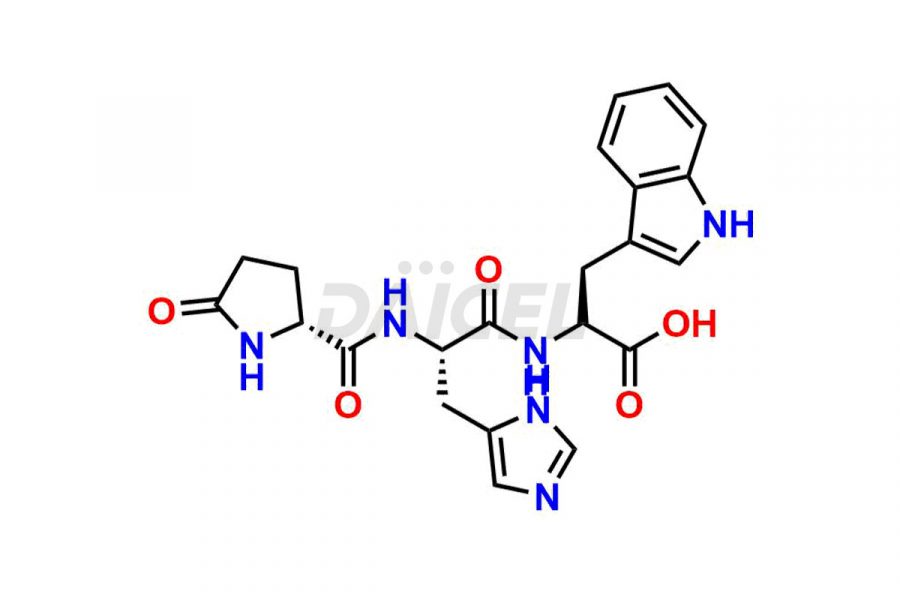
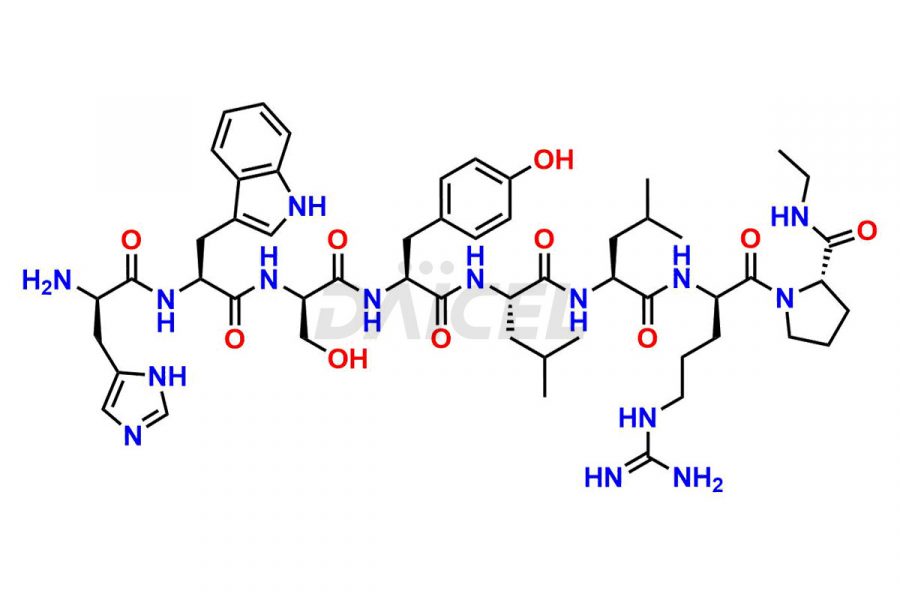
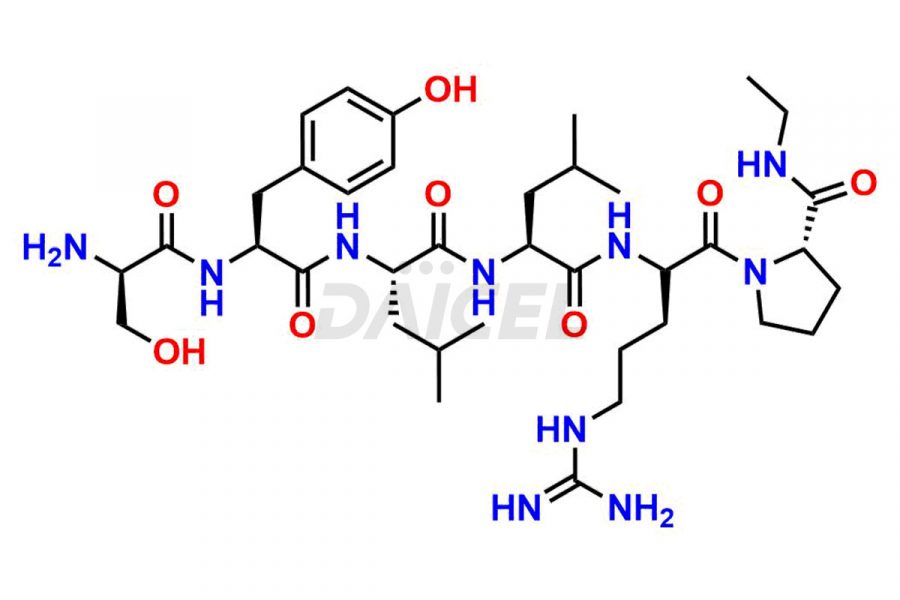
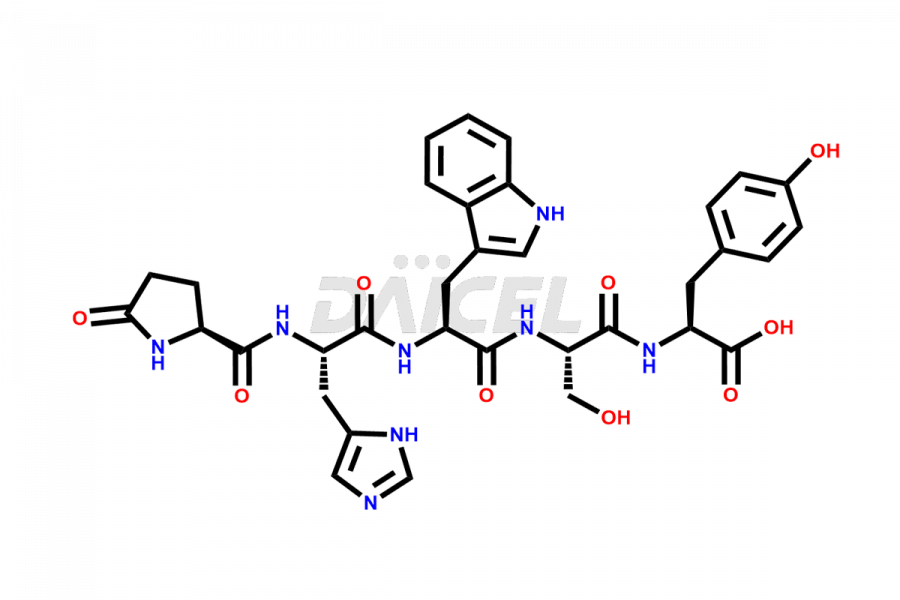
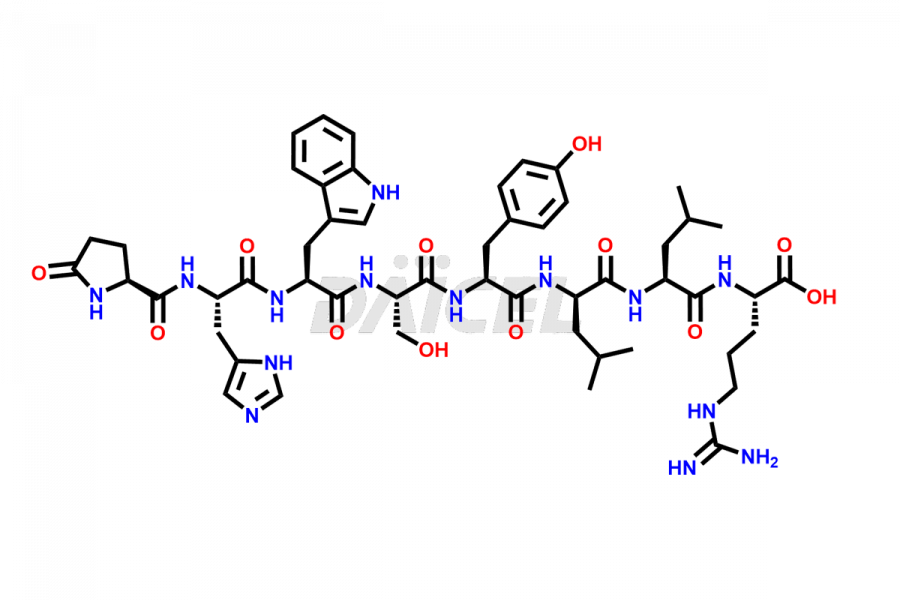
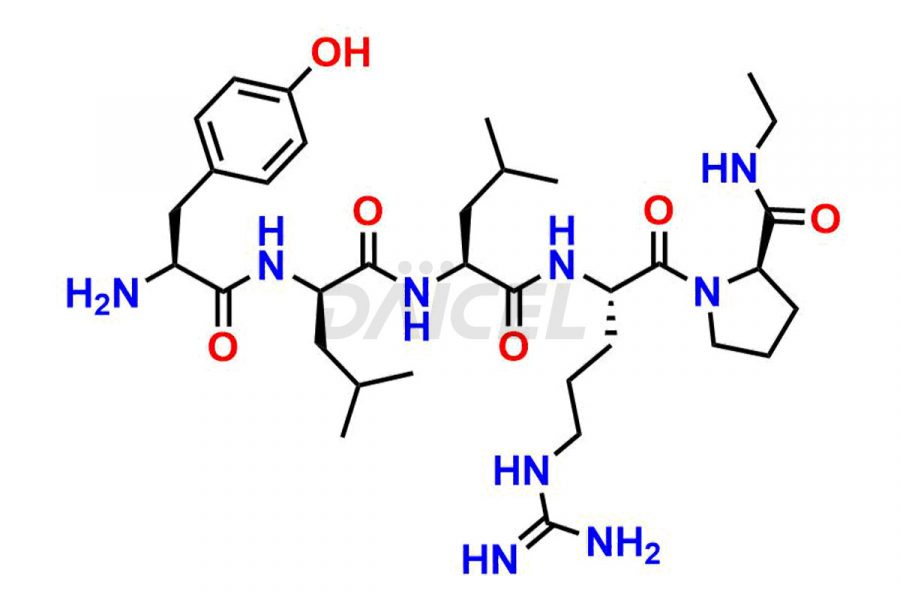
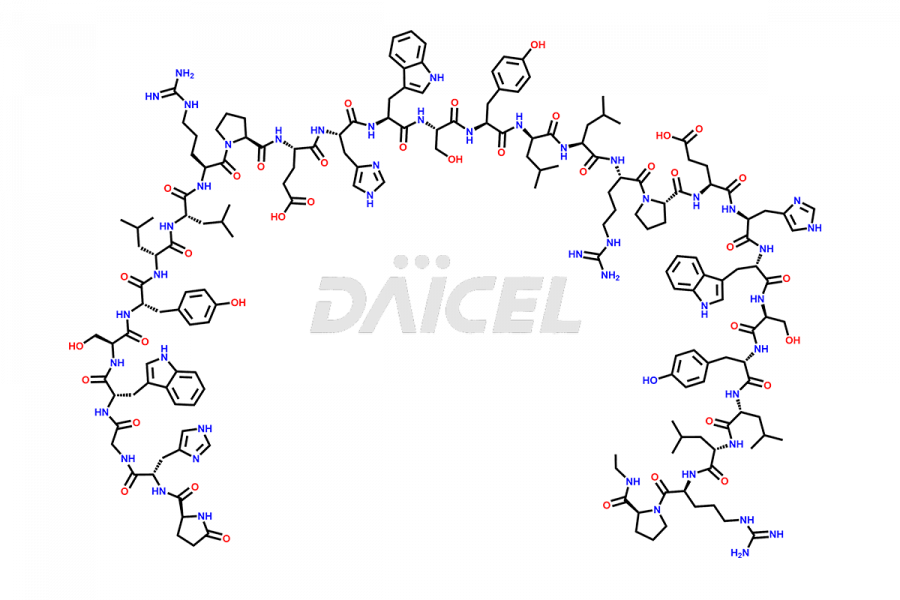
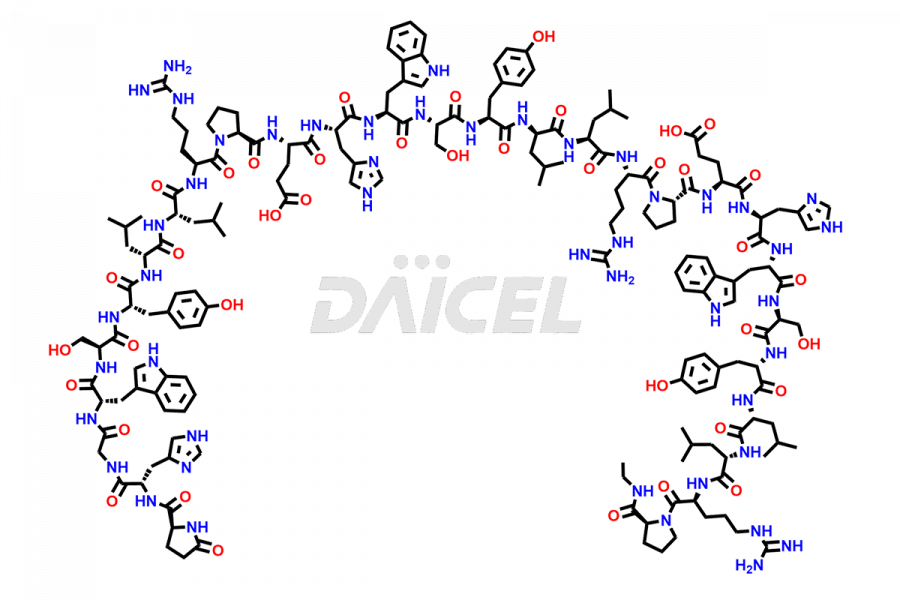
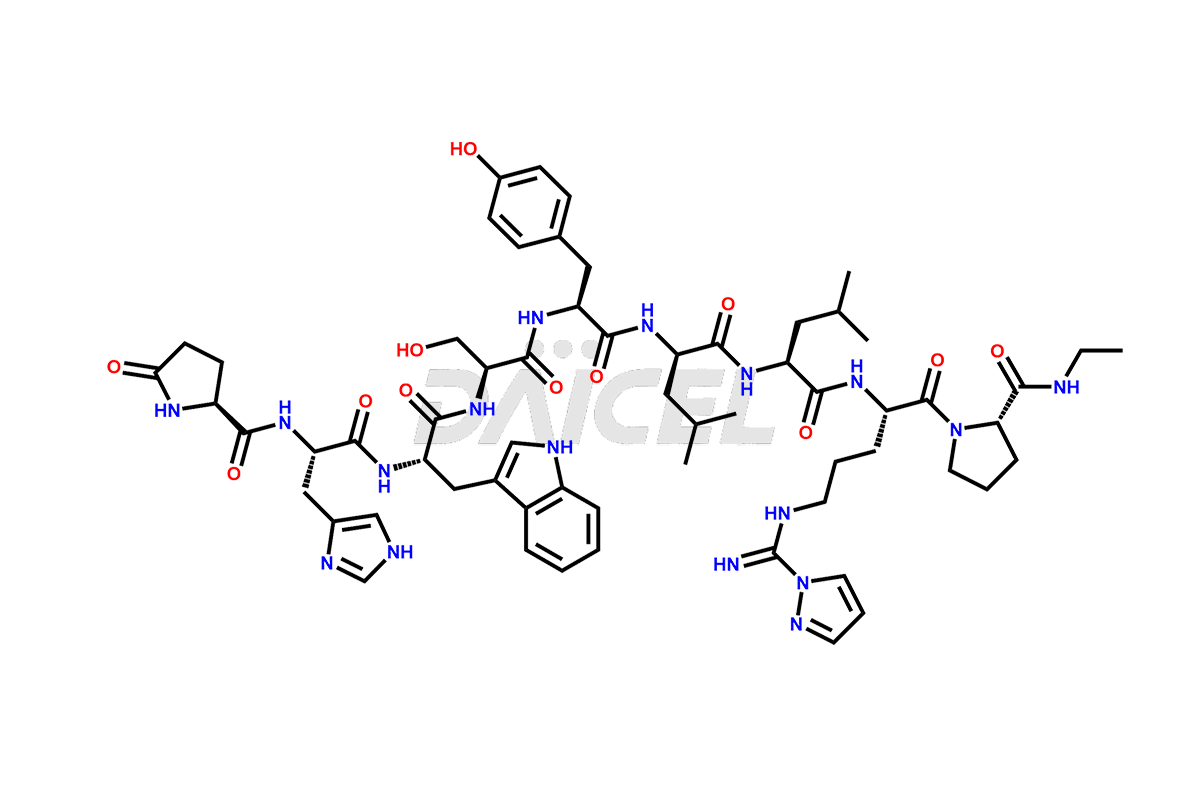
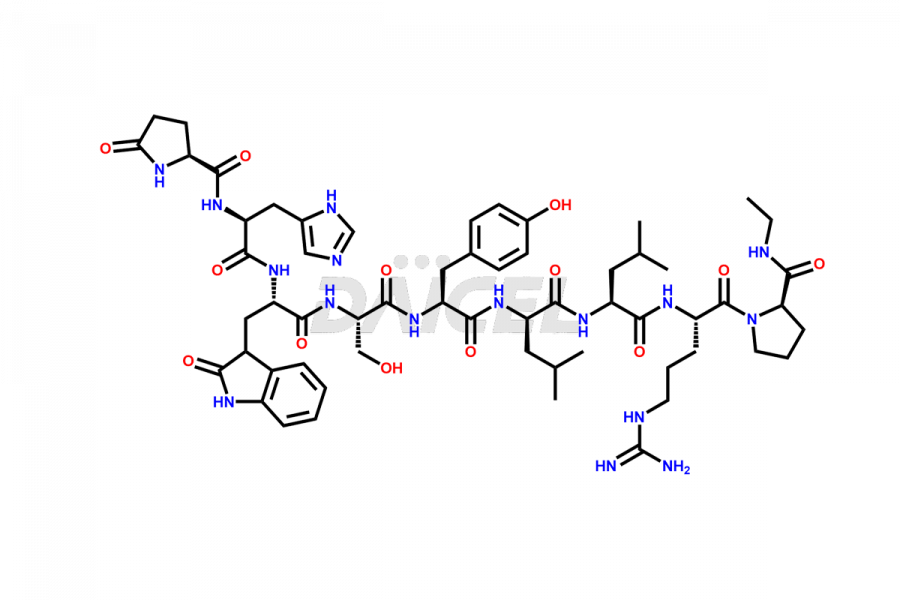
Leuprolide Impurities
Daicel’s leuprolide impurity standards support peptide synthesis control, stability testing, and analytical method validation.
General Information
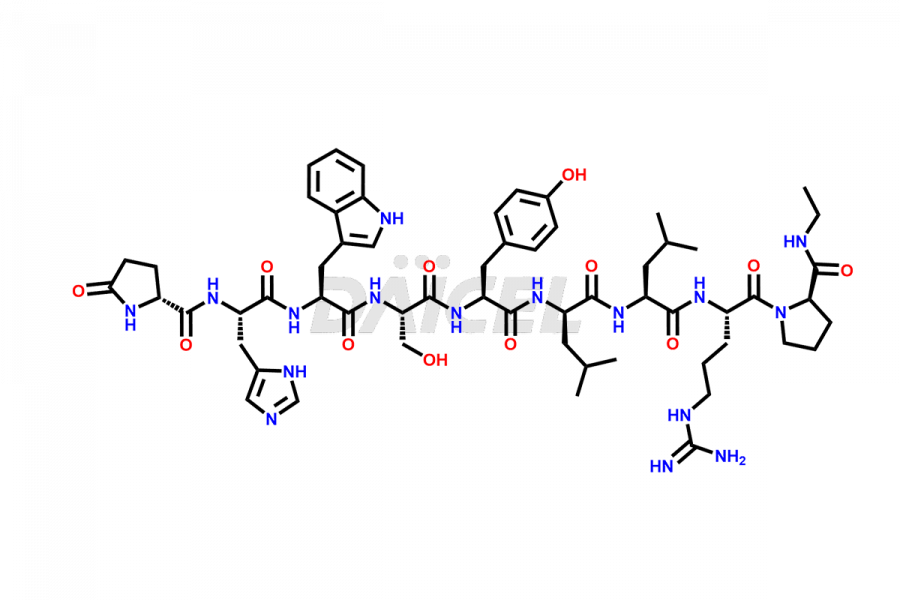
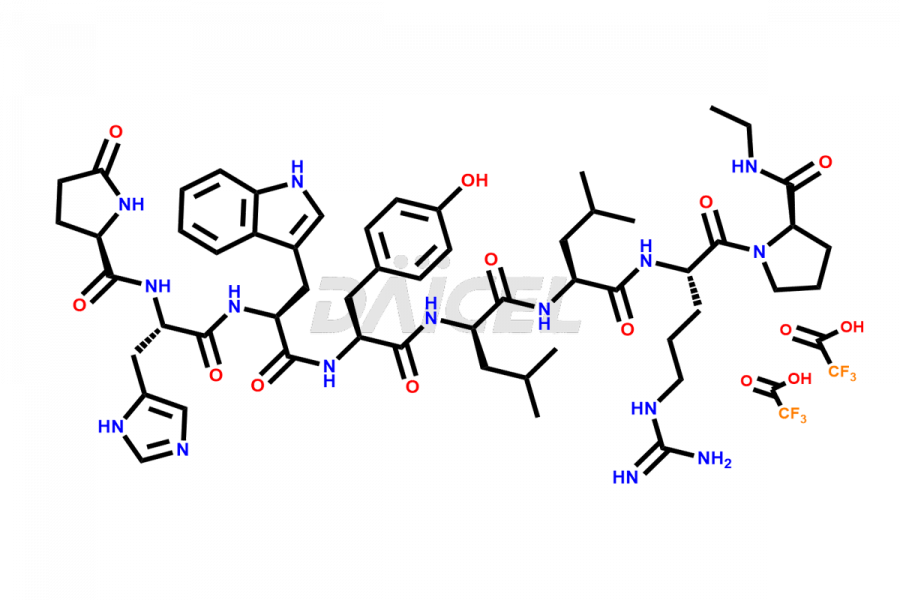
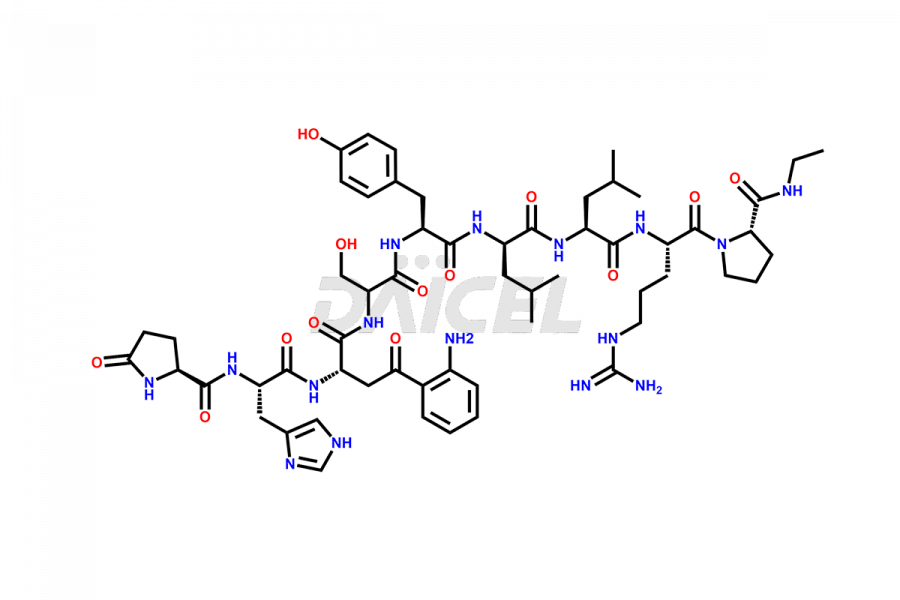
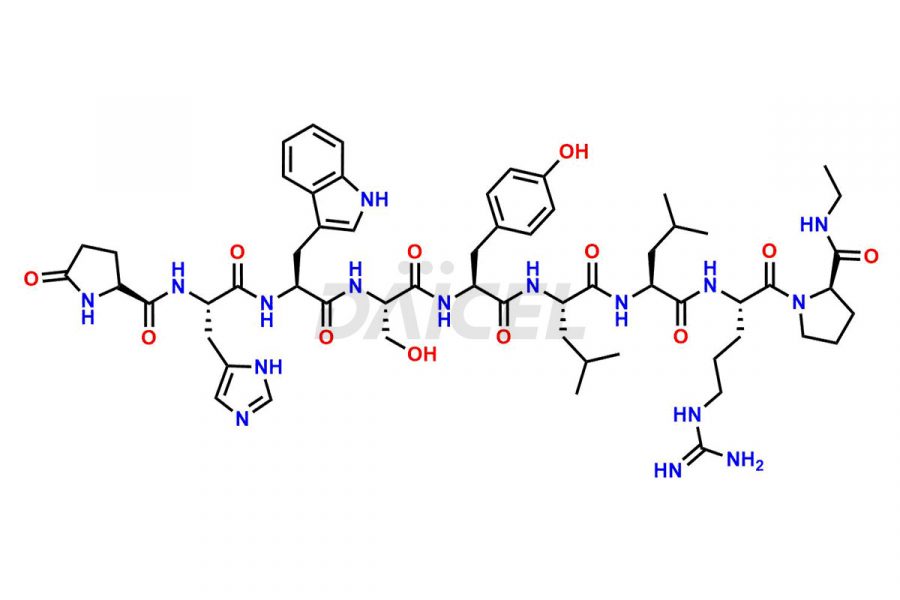
Leuprolide Impurities and Leuprolide
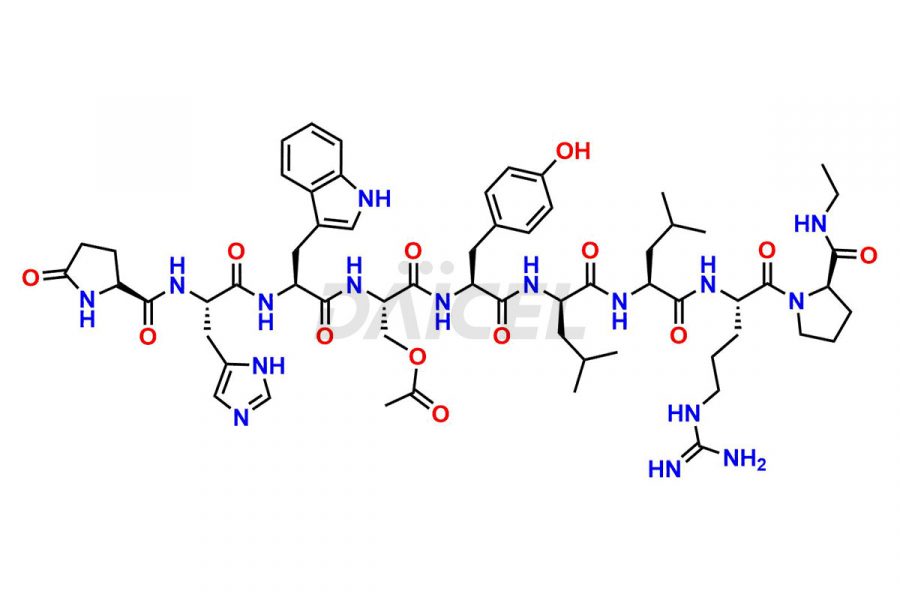
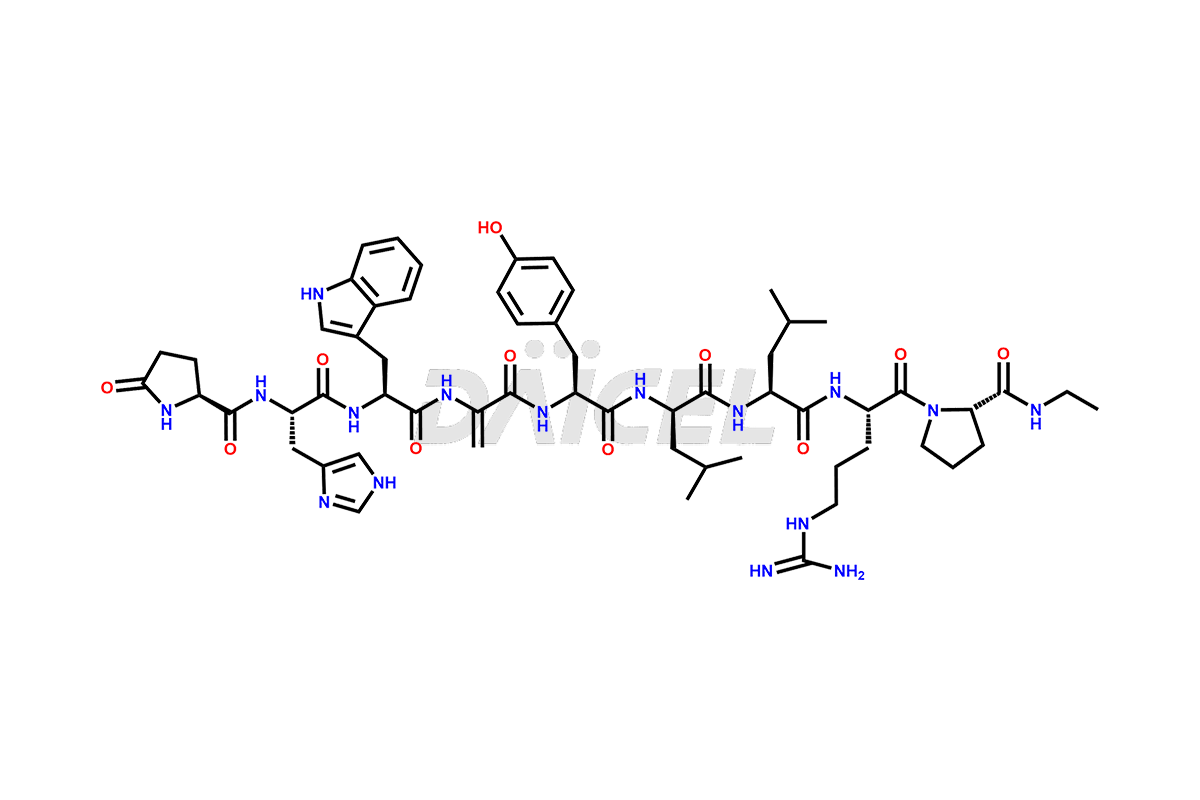
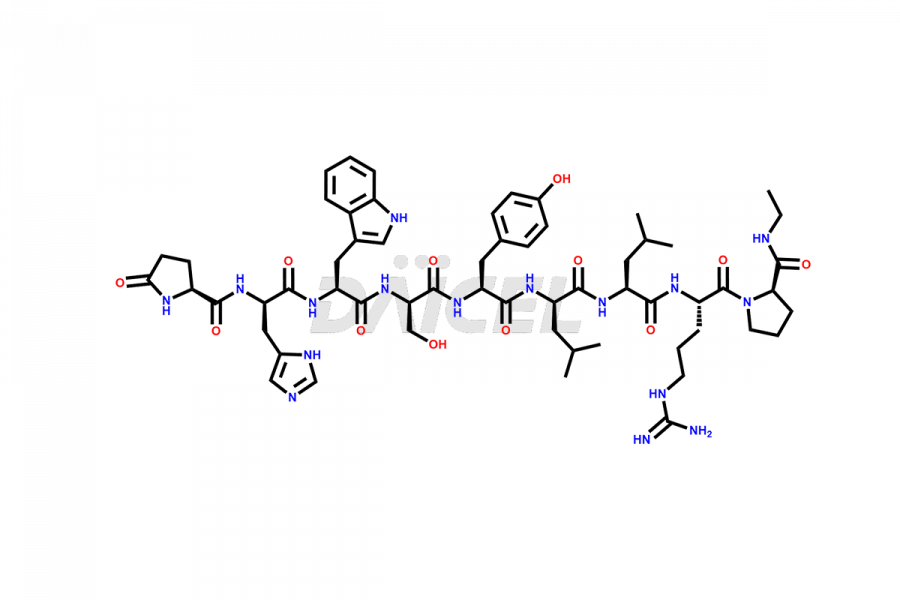
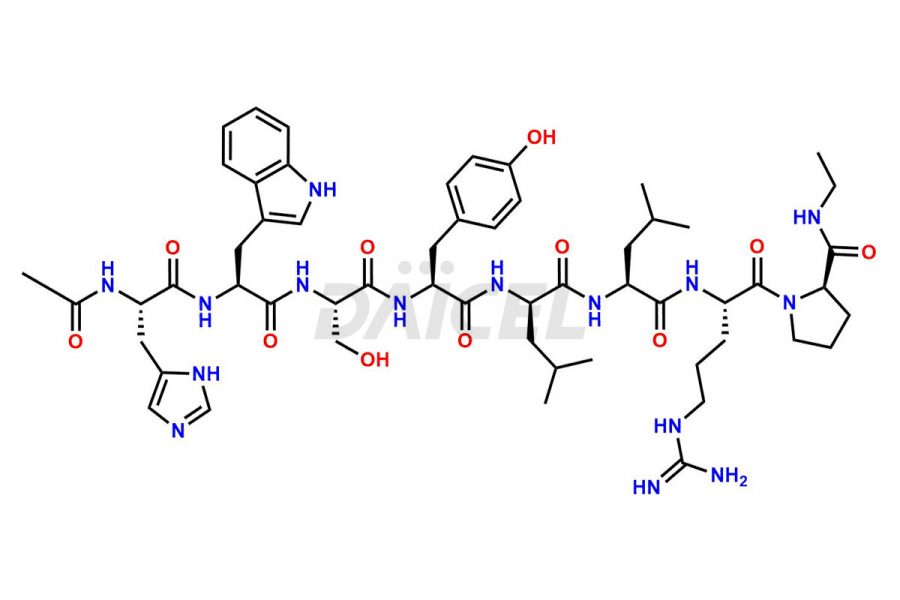
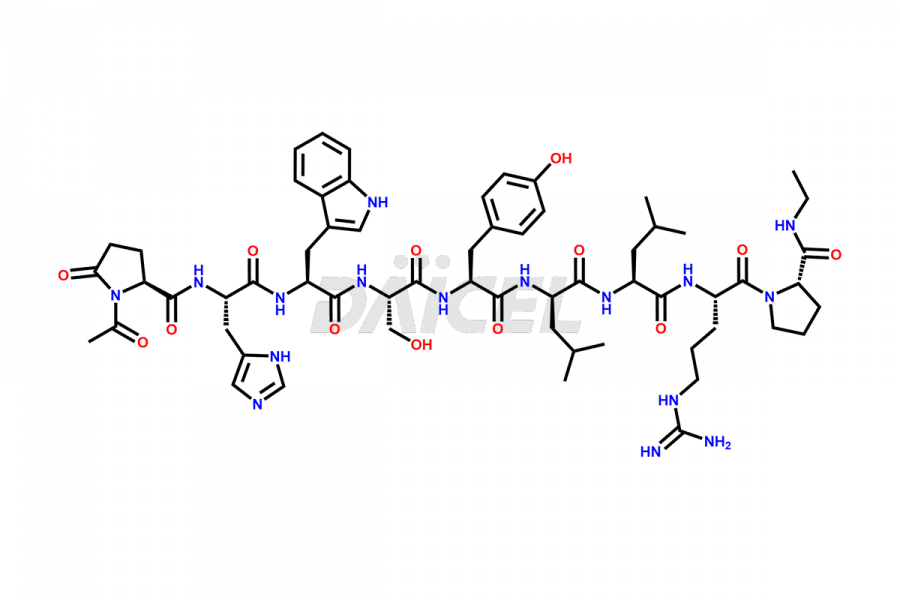
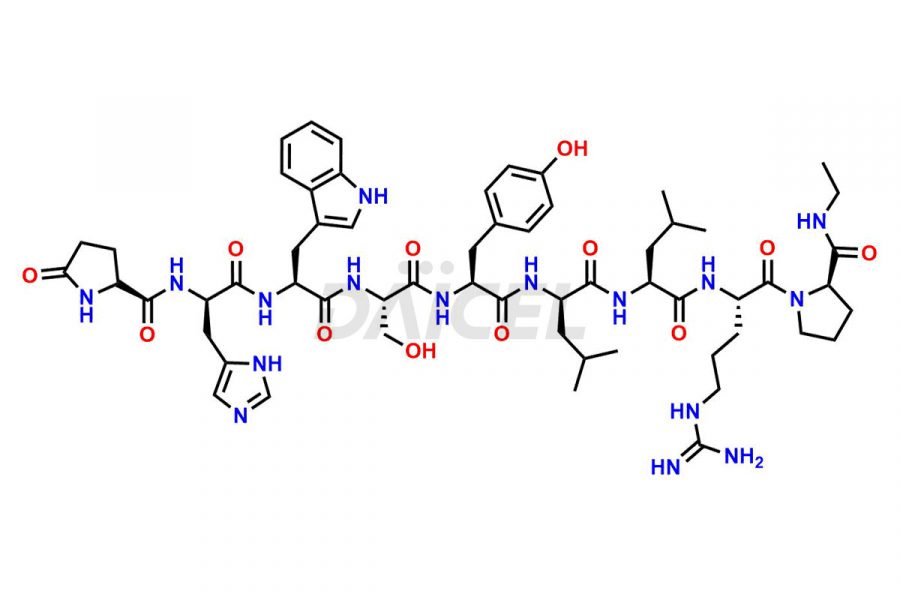
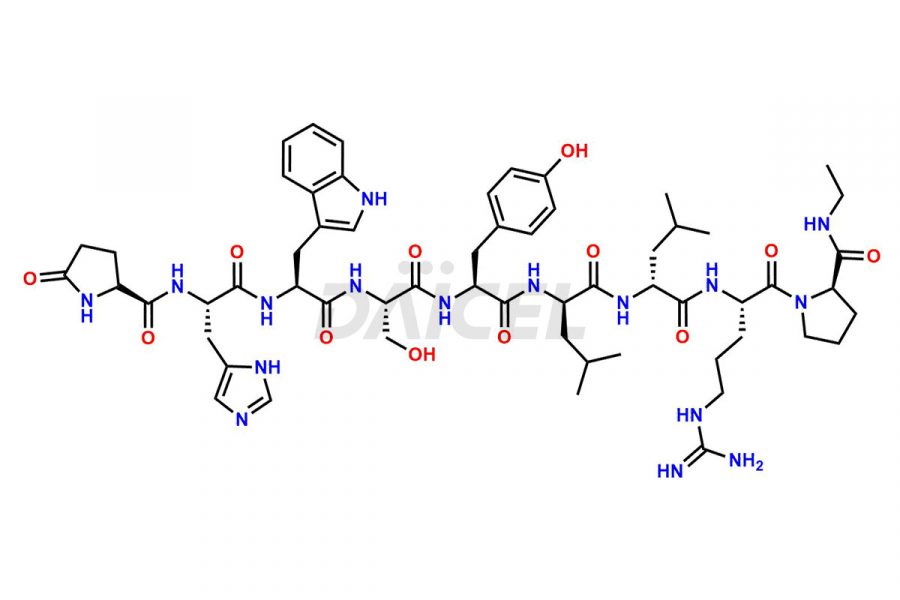
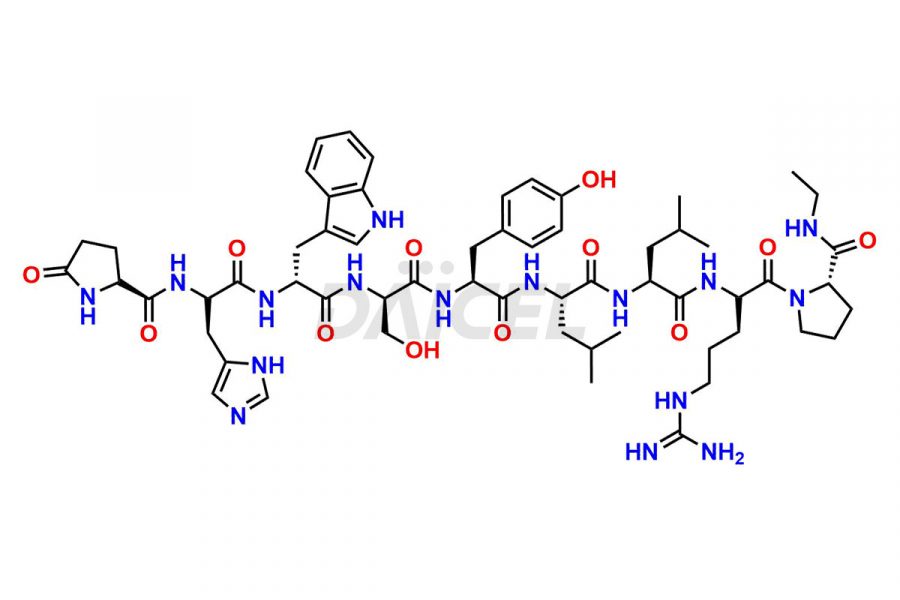
Daicel Pharma synthesizes high-quality Leuprolide impurities, (O Acetyl L Ser) Leuprolide, Ac(2-9) Leuprolide, D His Leuprolide, D Leu Leuprolide, D Ser Leuprolide, D Trp Leuprolide, Des-Ser(4)-Leuprolide, Leuprolide Metabolite(pentapeptide), and more, that help in the quality, stability, and biological safety analysis of Leuprolide. We also offer custom synthesis of Leuprolide impurities and supply worldwide.
Leuprolide [CAS: 53714-56-0] is a synthetic nonapeptide agonist analog of Luteinizing hormone-releasing factor1. It mimics the natural hormone known as gonadotropin-releasing hormone (GnRH). Leuprolide is a powerful GnRH receptor agonist, commonly prescribed in the treatment of prostate cancer, uterine leiomyomata, endometriosis, and central precocious puberty.
Leuprolide: Use and Commercial Availability
Leuprolide is prescribed for advanced prostate cancer and is used for pediatric patients with central precocious puberty (CPP). It is also prescribed in combination with iron supplements for preoperative hematological improvement in anemic patients with uterine leiomyomata (also known as uterine fibroids). Additionally, it is used in combination with oral norethisterone (also called norethindrone) in the initial treatment of endometriosis symptoms.
Leuprolide is available as Leuprolide acetate, which is administrable through an intramuscular or subcutaneous injection or as an implant. It is available under several brand names, such as Lupron, Eligard, Viadur, and Leuprorelin.

Leuprolide Structure and Mechanism of Action
The chemical formula for Leuprolide is C59H84N16O12, and its molecular weight is approximately 1,209.40 g/mol.
Leuprolide acetate suppresses the gonadotrope secretion of Luteinizing hormone (LH) and Follicle-Stimulating hormone (FSH) inhibiting gonadal sex steroid production.
Leuprolide Impurities and Synthesis
Leuprolide impurities may form during the synthesis process2 or storage of the drug. Deamidated Leuprolide, Leuprolide dimer, Leuprolide oxidation products, Leuprolide-related compounds, and residual solvents are some of the most common Leuprolide impurities affecting the efficacy and stability of the drug. Also, these impurities have toxic and immunogenic effects on the patients. Hence, the manufacturers need to perform rigorous analytical testing to detect and quantify these impurities to ensure the quality and safety of Leuprolide.
At Daicel, we provide a Certificate of Analysis (CoA) from a cGMP-compliant analytical facility for more than fifteen Leuprolide impurity standards such as (O Acetyl L Ser) Leuprolide, Ac(2-9) Leuprolide, D His Leuprolide, D Leu Leuprolide, D Ser Leuprolide, D Trp Leuprolide, Des-Ser(4)-Leuprolide, Leuprolide Metabolite(pentapeptide), and more, along with complete characterization data including 1H NMR, 13C NMR, IR, MASS, and HPLC3 purity. Upon request, we provide 13C-DEPT and CHN. Also, we provide a complete characterization report upon delivery. Daicel offers Leuprolide D10 (labelled), which is a deuterium-labeled standard of Leuprolide in bioanalytical research and BA/BE studies with isotope data in CoA.
References
FAQ's
References
- USP-NF on Leuprolide
- Alexander IvchenkoHagi AlonShai ElsterShimon ShushanChaim EidelmanAvi ToviTehila GadiLeah Bar-OzEleonora AltermanGil ZaoviGabriel-Marcus Butilca, Novetide Ltd, “Methods for the production of leuprolide”, EP2119725A1, November 18, 2009
- Sutherland, J. W.; Menon, G. N., , “HPLC of leuprolide acetate in injectable solutions”, Journal of Liquid Chromatography Volume: 10, Issue: 10, Pages: 2281-9,1987
Frequently Asked Questions
How are Leuprolide impurities detected and characterized during the synthesis process?
Leuprolide impurities are detected and characterized using various analytical techniques such as high-performance liquid chromatography (HPLC), liquid chromatography mass spectrometry (LC-MS), and infrared (IR) spectroscopy.
What are the factors influencing the formation of Leuprolide impurities in its synthesis?
The factors that can influence the formation of Leuprolide impurities during its synthesis include the quality and purity of starting materials, reaction conditions, catalysts and reagents used, purification methods, and storage conditions.
How are Leuprolide impurities synthesized?
The synthesis of Leuprolide impurities typically involves solid-phase peptide synthesis techniques, which include protecting and activating amino acid building blocks, assembly of the peptide chain, deprotection and purification of the peptide, and characterization and analysis of the final product
What are the storage conditions required for Leuprolide impurities?
The storage conditions for Leuprolide impurities may vary depending on the specific impurity and its stability. In general, it is recommended to store Leuprolide impurities at temperatures between 2-8°C in a dry and well-ventilated area to prevent degradation and maintain their stability. However, some impurities may require specific storage conditions such as refrigeration or freezing, to ensure their stability over an extended period.
Note: Products protected by valid patents by a manufacturer are not offered for sale in countries having patent protection. The sale of such products constitutes a patent infringement, and its liability is at the buyer's risk.