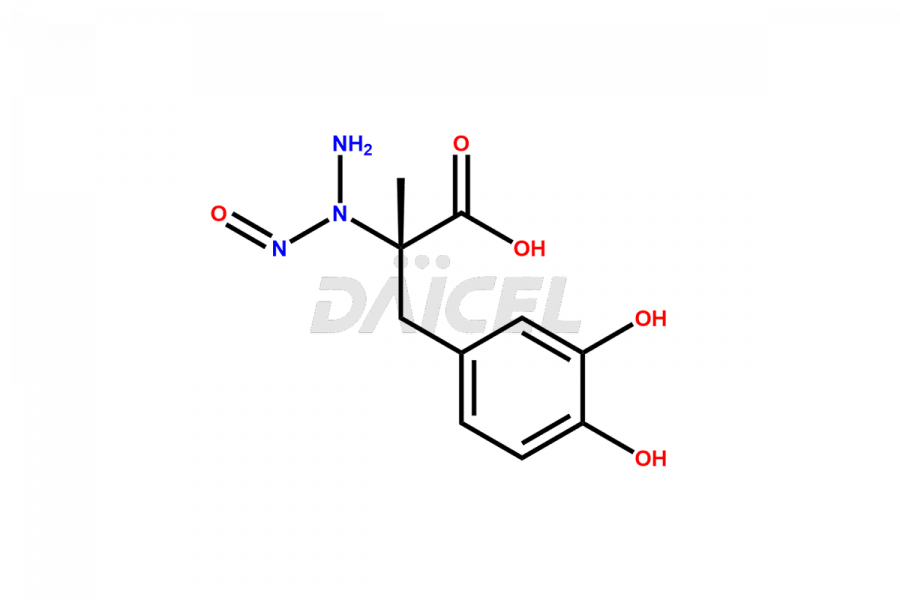
N-Nitroso Carbidopa Acetate Salt
- CAT Number DCTI-C-003658
- CAS NUMBER NA
- MOLECULAR FORMULA Free Acid: C10H13N3O5 Acetate Salt: Chemical Formula: C12H17N3O7-
- MOLECULAR WEIGHT Free Acid: 255.23 Acetate Salt: 315.28
N-Nitroso Carbidopa
- CAT Number DCTI-C-004481
- CAS Number NA
- Molecular Formula C10H13N3O5
- Molecular Weight 255.23